Abstract
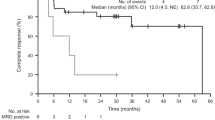
Long-term results of both pretreated and previously untreated patients (pts) with hairy cell leukemia (HCL) using uniformly a single 7-day course of 2-chlorodeoxyadenosine (2-CdA) by continuous infusion are reported. In addition, the probability of obtaining another response with this drug in pts who relapsed after 2-CdA treatment will be addressed. A total of 44 consecutive pts (34 males, 10 females) with a median age of 57 years (range 33–77) at the time of initiation of 2-CdA treatment were analyzed. In all, 11 pts were pretreated with either splenectomy (n=6), interferon α (n=9) or deoxycoformycin (dCF) (n=3) or all procedures in sequence. Two pts treated with dCF did not respond to dCF, but only 2-CdA. The median time to the start of 2-CdA treatment of the 11 pretreated pts was 47 months (mo) (10–160). Out of 44, 43 (98%) achieved complete response (CR) (13 pts with residual disease-RD), one pt reached a good partial response with a single cycle of 2-CdA. Out of 44 pts, 13 had no nonhematologic toxicities at all. Toxicities (WHO grade I–IV) were mainly of grade I and II, in one pt grade IV infectious complication. Bone marrow biopsies were performed at the time of recovery of hematopoiesis, thereafter at 2–3 mo intervals, thereafter at 6 mo, and finally annually in 35 pts. The median follow-up is 8.5 years (0.1–12.2). Disease-free survival from the start of 2-CdA treatment is 36% at 12 years (median 8.4 years), 17/44 pts relapsed. Nine of these pts were treated with 2-CdA again, eight achieved a second CR (median 2.5 yrs), one pt did not respond. Eight of our cohort had a second malignancy before receiving 2-CdA. Six pts died in CR due to the second malignancy. The overall survival at 12 years after the start of 2-CdA treatment is 79%. 2-CdA is a safe and effective treatment of HCL inducing complete remissions in the majority of pts with only a single cycle of 2-CdA, and a paucity of toxicities. Responses are durable and long-lasting. Pts who relapsed following treatment with 2-CdA responded to subsequent retreatment with 2-CdA.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Bouroncle BA, Wiseman BK, Doan CA . Leukemic reticuloendotheliosis. Blood 1958; 13: 609–630.
Golomb HM, Catovsky D, Golde DW . Hairy cell leukemia: a clinical review based on 71 cases. Ann Intern Med 1978; 89: 677–682.
Carson DA, Wasson DB, Beutler E . Antileukemic and immunosuppressive activity of 2-chloro-2-deoxy-adenosine. Proc Natl Acad Sci USA 1984; 81: 2232–2236.
Piro LD, Carrera CJ, Carson DA, Beutler E . Lasting remissions in hairy cell leukemia induced by a single infusion of 2-chlorodeoxyadenosine. New Engl J Med 1990; 322: 1117–1121.
Tallman MS, Hakimian D, Rademaker AW, Zanzig C, Wollins E, Rose E et al. Relapse of hairy cell leukemia after 2-chlorodeoxyadenosine: long-term follow-up of the Northwestern University experience. Blood 1996; 88: 1954–1959.
Hoffman MA, Janson D, Rose E, Rai KR . Treatment of hairy-cell leukemia with Cladribine: response, toxicity, and long-term follow-up. JCO 1997; 15: 1138–1142.
Lauria F, Rondelli D, Zinzani PL, Bocchia M, Marotta G, Salvucci M et al. Remission in patients with hairy cell leukemia treated with 2-CdA: a 5-year survey. Leukemia 1997; 11: 629–632.
Saven A, Burian C, Koziol JA, Piro LD . Long-term follow-up of patients with hairy cell leukemia after Cladribine treatment. Blood 1998; 92: 1918–1926.
Jehn U, Bartl R, Dietzfelbinger H, Vehling-Kaiser U, Wolf-Hornung B, Hill W et al. Long-term outcome of hairy cell leukemia (HCL) treated with 2-chlorodeoxyadenosine (2-CdA). Ann Hematol 1999; 78: 139–144.
Bartl R, Frisch B, Hill W, Burkhardt R, Sommerfeld W, Sund M . Bone marrow histology in hairy cell leukemia: identification of subtypes and their prognostic significance. Am J Clin Pathol 1983; 79: 531–545.
Anonymous. Consensus resolution. Proposed criteria for evaluation of response to treatment inhairy cell leukemia. 1987; 1: 405–408.
Hakimian D, Tallman MS, Kiley C, Peterson LAC . Detection of minimal residual disease by immunostaining of bone marrow biopsies after 2-chorodeoxyadenosine for hairy cell leukemia. Blood 1993; 82: 1798–1802.
Kaplan EL, Meier P . Non-parametric estimation from incomplete observations. JAMA 1958; 53: 457–481.
Estey EH, Kurzrock R, Kantarjian HM, O'Brien SM, McCredie KB, Beran M et al. Treatment of hairy cell leukemia with 2-chloro-deoxyadenosine (2-CdA). Blood 1992; 79: 882–887.
Tallman MS, Hakimian D, Variakojis D, Koslow D, Sisney GA, Rademaker AW et al. A single cycle of 2-chlorodeoxyadenosine results in complete remission in the majority of patients with hairy cell leukemia. Blood 1992; 80: 2203–2209.
Thaler J, Dietze O, Faber V, Greil R, Denz H, Ho AD et al. Monoclonal antibody B-ly 7: a sensitive marker for detection of minimal residual disease in hairy cell leukemia. Leukemia 1990; 4: 170–175.
Burns GF, Cawley JC, Worman CP . Multiple heavy chain isotypes on the surface of the cells of hairy cell leukemia. Blood 1978; 52: 1132–1136.
Thomas DA, O'Brien S, Bueso-Ramos C, Faderl ST, Keating MJ, Giles FJ et al. Rituximab in relapsed or refractory hairy cell leukemia. Blood 2003; 102: 3906–3911.
Wheaton S, Tallman MS, Hakimian D, Peterson L . Minimal residual disease may predict bone marrow relapse in patients with hairy cell leukemia treated with 2-chlorodeoxyadenosine. Blood 1996; 87: 1556–1560.
Kurzrock R, Strom SS, Estey E, O'Brien S, Keating MJ, Jiang H et al. Second cancer risk in hairy cell leukemia: analysis of 350 patients. JCO 1997; 15: 1803–1810.
Cheson BD, Vena DA, Barett J, Freidlin B . Second malignancies as a consequence of nucleoside analog therapy for chronic lymphoid leukemias. JCO 1999; 17: 2454–2460.
Federico M, Zinzani PL, Frassoldati A, Vinceti M, Mode A, Annino L et al. Risk of second cancer in patients with hairy cell leukemia: long term follow-up. JCO 2002; 20: 638–646.
Schlaifer D, Rigal-Huguet F, Pris J . Secondary neoplasms in two patients treated with purine analogues. Nouo Rev Fr Hematol 1994; 36: 321 (letter).
Ho AD, Thaler J, Mandelli F, Lauria F, Zittoun R, Willemze R, et al., for the European Organization for the Research and Treatment of Cancer Leukemia Cooperative Group. Response to pentostatin in hairy-cell-leukemia refractory to interferon alpha. J Clin Oncol 1989; 7: 1533–1538.
Kraut EH, Bouroncle BA, Grever MR . Low-dose deoxycoformycin in the treatment of hairy cell leukemia. Blood 1986; 68: 1119–1122.
Spiers ADS, Moore D, Cassileth PA, Harrington D, Cummings F, Neiman R et al. Remissions in hairy-cell-leukemia with pentostatin. N Engl J Med 1987; 316: 825–830.
Nieva J, Bethel K, Saven A . Phase 2 study of rituximab in the treatment of cladribin-failed patients with hairy cell leukemia. Blood 2003; 102: 810–813.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Jehn, U., Bartl, R., Dietzfelbinger, H. et al. An update: 12-year follow-up of patients with hairy cell leukemia following treatment with 2-chlorodeoxyadenosine. Leukemia 18, 1476–1481 (2004). https://doi.org/10.1038/sj.leu.2403418
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.leu.2403418
Keywords
This article is cited by
-
Hairy Cell Leukemia (HCL) and HCL Variant: Updates and Spotlights on Therapeutic Advances
Current Oncology Reports (2022)
-
Analysis of a cohort of 279 patients with hairy-cell leukemia (HCL): 10 years of follow-up
Blood Cancer Journal (2020)
-
Retrospective Analysis of Hairy Cell Leukemia Patients Treated with Different Modalities as First Line: Real-Life Experience Over 20 years
Indian Journal of Hematology and Blood Transfusion (2019)
-
Hairy cell leukemia: short review, today’s recommendations and outlook
Blood Cancer Journal (2014)
-
Update on the Biology and Treatment Options for Hairy Cell Leukemia
Current Treatment Options in Oncology (2014)