Abstract
Keratinocyte apoptosis induced by UV radiation is a major protective mechanism from skin photocarcinogenesis. The induction of apoptosis by UV radiation, as well as a variety of genotoxic stimuli, involves the activation of PKC-δ by caspase-3-mediated cleavage in its hinge region, thus generating a constitutively active catalytic fragment. To determine the role of PKC-δ cleavage in UV apoptosis signaling, we introduced a caspase-resistant PKC-δ mutant (D330A) into human keratinocytes by retrovirus transduction. Overexpression of PKC-δ(D330A) protected keratinocytes from UV-induced apoptosis and enhanced long-term survival. PKC-δ(D330A) partially prevented the release of cytochrome c from the mitochondria and the loss of Mcl-1, a key antiapoptotic protein downregulated during UV apoptosis. Thus, the cleavage and activation of PKC-δ are critical components of UV-induced apoptosis in human keratinocytes, and the inactivation of PKC-δ can promote the survival of keratinocytes exposed to UV radiation.
Similar content being viewed by others
Introduction
Ultraviolet (UV)B radiation from the sun is the primary carcinogen responsible for the formation of nonmelanoma skin cancer, by far the most common type of cancer in the US. Apoptosis is a major tumor suppressor mechanism that eliminates damaged keratinocytes at risk of becoming transformed from the epidermis. UV radiation triggers apoptosis in human keratinocytes by multiple signaling pathways. UV radiation primarily activates the intrinsic or mitochondrial pathway by causing the release of cytochrome c from the mitochondria.1 Cytochrome c forms a apoptosome complex with Apaf-1 and caspase-9 triggering the activation of caspase-9. In keratinocytes, activated caspase-9 is a critical upstream caspase required for initiating UV apoptosis.2 However, UV radiation also triggers the clustering and activation of membrane receptors such as Fas and tumor necrosis factor receptor.3, 4, 5 Caspase-3 is an effector caspase that can be activated by both caspase-8 and caspase-9. Activation of the effector caspases leads to cleavage of numerous cellular proteins that trigger nuclear breakdown, loss of mitochondrial membrane potential, and cell death.
PKC-δ is one of over 40 known caspase substrates and is cleaved specifically by caspase-3.1, 6 This cleavage takes place in the hinge region (V3) of PKC-δ at the caspase-3 recognition sequence (DMQD330/N). The cleavage of PKC-δ is common to the death effector pathways of diverse apoptotic stimuli, including UV radiation, ionizing radiation, DNA-damaging drugs, TNF-α, and anti-Fas antibody. There is a form of PKC-δ (PKC-δ II) which is insensitive to caspase-3,7 thus cleavage sensitivity can be regulated at the level of RNA splicing. While other PKC isoforms (PKC ɛ, θ, ζ) can undergo proteolysis by caspases, PKC-δ was the only isoform activated by this mechanism in keratinocytes during UV radiation-induced apoptosis.8, 9 PKC-δ itself can also initiate apoptosis in keratinocytes involving direct interaction with the mitochondria and alterations of mitochondrial functions.10, 11 Overexpression of the catalytic domain of PKC-δ is sufficient to cause apoptosis in normal keratinocytes, HaCaT cells, HeLa, NIH3T3 and COS1 cells, suggesting that PKC-δ is an important effector of apoptosis.6, 11, 12 Inhibition of PKC activity by the general PKC inhibitor GF109293X partially blocked UV-induced cleavage and activation of PKC-δ, pro-caspase-3, and pro-caspase-8, and thus rescued keratinocytes from UV-induced apoptosis, indicating that PKC is involved in a positive feedback loop that amplifies the apoptotic response of keratinocytes to UV.11, 13
In this study, we examined the mechanism of protection from UV-induced apoptosis in human keratinocytes by selectively blocking the caspase-3-mediated cleavage of PKC-δ. Our results demonstrate that inhibition of PKC-δ activation by caspase cleavage significantly inhibits apoptosis induced by UV radiation and promotes cell survival. Blocking PKC-δ cleavage inhibited the downregulation of Mcl-1 and the release of cytochrome c from the mitochondria, thus preventing the feedback activation of caspases. This indicates that the positive feedback mediated by cleavage-induced activation of PKC-δ is required for UV-induced apoptosis in normal human keratinocytes.
Results
PKC-δ(D330A) is resistant to cleavage by caspase-3
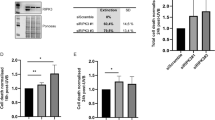
PKC-δ is cleaved by caspase-3 in its hinge region at the recognition motif DMQD330. To create a mutant PKC-δ that is resistant to cleavage and activation by caspase-3, we mutated the aspartic acid at 330 to alanine (D330A) as shown schematically in Figure 1a. The full-length wild-type and mutant forms of PKC-δ were detectable as a 78-kDa band in Figure 1b. Incubation of cell lysates containing the wild-type PKC-δ with active recombinant caspase-3 generated a 43-kDa domain catalytic product. In contrast, the PKC-δ(D330A) mutant was not cleaved by recombinant caspase-3. Weak, nonspecific 45 kDa bands were also detected in all lanes.
Effect of recombinant caspase-3 on PKC-δ and PKC-δ(D330A) cleavage. (a) Schematic representation of PKC-δ and the D330A mutation at the caspase-3 cleavage site in the hinge domain of PKC-δ. (b) Lysates from cells infected with wild-type PKC-δ virus or PKC-δ(D330A) virus were incubated with 200 ng recombinant caspase-3 for 1 h as indicated and analyzed by Western blot for PKC-δ. Note that the wild-type PKC-δ was cleaved by caspase-3, but PKC-δ(D330A) was not
Activation of PKC-δ by UV is blocked by overexpression of PKC-δ(D330A)
To determine if the noncleavable PKC-δ(D330A) is resistant to activation in cells with caspases activated, we overexpressed PKC-δ(D330A) in normal human keratinocytes by retroviral transduction and exposed the cells to UV radiation. As shown in Figure 2, PKC-δ activity was induced over 50-fold 18 h after UV irradiation in vector-infected control cells (Linker), but this induction was inhibited by 64.3% in PKC-δ(D330A)-expressing cells. Thus, PKC-δ(D330A) can act as a dominant inhibitor of PKC-δ activation by UV. In contrast, PKC-δ(D330A) had no effect on the activation of PKC-δ by TPA treatment of cells (Figure 2b), indicating that PKC-δ(D330A) selectively inhibited the caspase-3-dependent PKC-δ activation.
PKC-δ(D330A) inhibits the activation of PKC-δ by UV, but not by TPA. (a) Keratinocytes were infected with control (Linker) or PKC-δ(D330A) virus, and irradiated with 30 mJ/cm2 UV. PKC-δ was immunoprecipitated from the cell lysates 18 h after UV exposure, and the enzyme activity assayed. Note the significant inhibition of UV-induced PKC-δ activity by PKC-δ(D330A) (n=2, P<0.05) with error bars denoting range. (b) Keratinocytes were treated with 2 μM TPA for 15 min. PKC-δ was immunoprecipitated from the cell lysates and the enzyme activity was assayed. This experiment was performed twice with similar results
Normal human keratinocytes are protected from UV-induced apoptosis
Keratinocytes infected with control Linker virus or PKC-δ(D330A) virus had a typical cobblestone appearance as seen in Figure 3a. When Linker virus-infected keratinocytes underwent UV-induced apoptosis, they became rounded, detached and floated in the media. PKC-δ(D330A)-infected keratinocytes were protected from morphological UV-induced cell death as compared to Linker-infected controls, although an approximately 50% decrease in the cell number was observed. This decrease in cell number may be due to a generalized cell cycle arrest induced by UV in the PKC-δ(D330A)-expressing cells, or may reflect the incomplete protection from apoptosis by PKC-δ(D330A), which was also on the order of 50% (Figure 4). PKC-δ(D330A)-infected keratinocytes had more than 10-fold overexpression of the mutant form of PKC-δ as compared to endogenous PKC-δ (Figure 3b).
PKC-δ(D330A) protects normal keratinocytes from UV-induced cell death. (a) Keratinocytes were infected with either LZRS-Linker control virus or PKC-δ(D330A) virus. At 2 days after infection, the cells were exposed to 30 mJ/cm2 of UV. At 18 h after UV exposure, cells were photographed. (b) Total cell lysates were prepared from keratinocytes infected with Linker (L) virus or PKC-δ(D330A) virus, and blotted for PKC-δ and Actin. (c) Equal numbers of keratinocytes were infected with either Linker control, Bcl-xL, or PKC-δ(D330A) virus, and 2 days later exposed to 30 mJ/cm2 UV. After 1 day, the media was changed and the cells were grown in the same wells until day 3 when they were replated into larger dishes. Viable cell counts were taken on the indicated days after irradiation. The asterisks denote statistically significant different viable cell numbers compared to the Linker control at each time point (n=2, *P<0.05) with error bars denoting range. Similar results were obtained in an independent experiment
PKC-δ(D330A) protects keratinocytes from UV-induced apoptosis. (a) Keratinocytes infected with either Linker virus or PKC-δ(D330A) virus were exposed to 30 mJ/cm2 UV and after 18 h, the cells were stained with propidium iodide and analyzed by flow cytometry. The percentage of cells with sub-G1 DNA (apoptotic) is indicated. (b) The sub-G1 DNA content of cells from three independent experiments performed as described in (a) is shown. The error bars denote S.D. (n=3, P<0.05). (c) Keratinocytes infected with either Linker or PKC-δ(D330A) virus were exposed to 30 mJ/cm2 UV. At 18 h after UV exposure, the cells were stained with Annexin V and propidium iodide and analyzed by flow cytometry. (d) The percentage of cells undergoing apoptosis (early plus late) from three independent experiments was quantitated and represented in a bar graph. The error bars denote S.D. (n=3, P<0.05)
The survival of keratinocytes exposed to UV was also enhanced by PKC-δ(D330A) expression. As shown in Figure 3c, the number of viable cells at 1, 3, and 7 days after UV exposure was higher in the cells expressing PKC-δ(D300A) than Linker-infected control cells (*P<0.05). While the cells did not proliferate significantly while remaining in the same dish due to apoptotic cellular debris, the keratinocytes did grow after day 3 upon replating into larger dishes. Note that the growth rate of the cells was not increased, but only the overall long-term survival. As a positive control, Bcl-xL infection also enhanced the survival and growth of UV-irradiated keratinocytes cells. These results indicate that inhibition of PCK-δ cleavage and activation does not simply delay apoptosis, but affords a significant growth advantage.
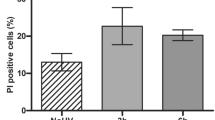
To determine whether PKC-δ(D330A)-infected keratinocytes were protected from apoptosis induced by UV, we performed propidium iodide and Annexin V staining of keratinocytes 18 h after UV exposure. Figures 4a and b show that in keratinocytes infected with PKC-δ(D330A), UV-induced apoptosis was inhibited 62%, as measured by propidium iodide staining from three independent experiments (P<0.05). Annexin V/PI staining was also performed to quantitate early and late apoptotic events. PKC-δ(D330A) was able to protect keratinocytes from UV-induced apoptosis as compared to Linker-infected controls. As shown in Figures 4c and d, the protection from UV-induced apoptosis was 68.1% by Annexin V staining from three independent experiments (P<0.05).
PKC-δ(D330A) inhibits the release of cytochrome c in response to UV
Cytochrome c release from the mitochondria is an early triggering event in UV apoptosis, while PKC-δ cleavage by caspase-3 is a late effector event. As shown in Figure 5, cytochrome c is elevated ∼5-fold in the cytoplasmic fraction following UV irradiation, indicating release from the mitochondria. UV-induced cytochrome c release was partially blocked by overexpressing PKC-δ(D330A). The inhibition of cytochrome c release by PKC-δ(D330A) was reproducible, and an average of four experiments is shown in Figure 5b (n=4, P<0.05). The release of cytochrome c from mitochondria was also blocked by the antiapoptotic Bcl-xL (data not shown).
PKC-δ(D330A) inhibits the release of cytochrome c from mitochondria in response to UV. (a) Keratinocytes infected with either Linker virus or PKC-δ(D330A) were left untreated or exposed to 30 mJ/cm2 UV. After 18 h, the cells were fractionated, and cytosolic fractions were analyzed by Western blotting for cytochrome c and Actin. (b) Cytochrome c release data from four independent experiments as described in (a) were quantitated and are shown in the bar graph. The level of cytosolic cytochrome c in untreated Linker-infected keratinocytes was normalized to 1.0 for each experiment. The error bars denote S.D. (n=4, P<0.05)
PKC-δ(D330A) inhibits caspase-3 activity in response to UV
Since the caspase cleavage-resistant PKC-δ(D330A) was able to reduce cytochrome c release, we determined the activity of caspase-3 using the fluorogenic substrate Ac-DEVD-AFC and performed Western blots for caspases-3 and cleaved caspase-9 (Figure 6). PKC-δ(D330A) significantly inhibited caspase-3 activity (66%) in response to UV exposure. This result was obtained from three independent experiments (P<0.05). PKC-δ(D330A) also partially prevented the cleavage of full-length caspase-3 and cleaved caspase-9 in response to UV. In the upper panel of Figure 6b, the full-length form of caspase-3 was detected as a 32-kDa band. In the middle panel of Figure 6b, the cleaved product of caspase-9 was detected as a 17-kDa band. PKC-δ(D330A) was able to inhibit caspase-9 cleavage since the level of activated caspase-9 was three-fold less in PKC-δ(D330A) than Linker-infected controls. Actin was used as a loading control in the lower panel of Figure 6b. Taken together, these results indicate that PKC-δ cleavage is important for amplification of the caspase cascade in the UV-death effector pathway.
UV-induced caspase-3 activation is blocked by PKC-δ(D330A). (a) Keratinocytes infected with either Linker virus or PKC-δ(D330A) virus were exposed to 30 mJ/cm2 UV and after 18 h, the caspase activity in the cell lysates was assayed using fluorogenic caspase-3 substrate Ac-DEVD-AFC. The results from three independent experiments were quantitated. The error bars denote S.D. (n=3, P<0.05). (b) Cell lysates were analyzed by Western blotting for full-length caspase-3, cleaved caspase-9, and Actin as the loading control
PKC-δ(D330A) protects keratinocytes from UV-induced loss of mitochondrial membrane potential
To determine whether PKC-δ(D330A) was able to protect human keratinocytes from UV-induced loss of mitochondrial membrane potential, we performed Rhodamine 123 staining. The loss of mitochondrial membrane potential is an indicator of cells undergoing the terminal phase of UV-induced apoptosis and is dependent on caspase activity. Figure 7 shows that PKC-δ(D330A) protected keratinocytes from UV-induced loss of mitochondrial membrane potential as compared to Linker-infected controls. As shown in Figure 7a (a representative experiment), 50% of Linker-infected control cells had lost their mitochondrial membrane potential in response to UV compared to 17.6% in PKC-δ(D330A)-infected keratinocytes. Figure 7b shows that the percentage of protection was 57.9% as determined by Rhodamine 123 staining from three independent experiments (P<0.05). Thus, PKC-δ(D330A) protected normal human keratinocytes from UV-induced loss of mitochondrial membrane potential.
PKC-δ(D330A) protects keratinocytes from UV-induced loss of mitochondrial membrane potential. (a) Keratinocytes infected with either Linker virus or PKC-δ(D330A) virus were exposed to 30 mJ/cm2 UV and after 18 h, the cells were stained with Rhodamine 123 and analyzed by flow cytometry. (b) The percent of cells with loss of mitochondrial membrane potential from three independent experiments are quantitated in bar graph form. The error bars denote S.D. (n=3, P<0.05)
PKC-δ(D330A) protects keratinocytes from UV-induced loss of Mcl-1
The UV-induced loss of Mcl-1 is required for release of cytochrome c from mitochondria. To determine if PKC inhibition was able to prevent the downregulation of Mcl-1, keratinocytes were infected with Linker virus, a kinase-inactive PKC-δ(K378A) virus, PKC-δ(D330A) virus, or treated with the general PKC inhibitor GF109203X. At 18 h after UV exposure, Mcl-1 levels were detected by Western blotting. Figure 8a shows that PKC-δ(K378A), PKC-δ(D330A), and GF109203X were able to partially protect keratinocytes from UV-induced downregulation of Mcl-1 as compared to Linker-infected control cells. An extra band was observed at approximately 66 kDa in the PKC-δ Western, which could represent a novel cleavage product. However, this band was not reproducible seen elevated in UV-irradiated cell lysates (Figure 9b). The downregulation of Mcl-1 in keratinocytes in response to UV is partly dependent on caspase activity since zVAD was also able to protect keratinocytes from UV-induced downregulation of Mcl-1, especially at the 18 h time point (Figure 8b).
PKC-δ(D330A) protects keratinocytes from loss of Mcl-1 during UV apoptosis. (a) Keratinocytes were infected with PKC-δ(K378A) virus (K), PKC-δ(D330A) virus (D), Linker virus (L), or treated with the PKC inhibitor GF109203X (GF) as indicated and exposed to UV. After 18 h, lysates were prepared for Mcl-1 Western blotting. Note the reduction in Mcl-1 levels by UV (Linker + UV) is partially blocked by PKC-δ (K378A), PKC-δ(D330A), and GF109203X. Overexpression of the mutant PKC-δ forms was verified by PKCδ Western blot. (b) Mcl-1 protein levels were detected by Western blotting at 0, 3, 6, and 18 h after UV exposure in the presence and absence of zVAD. Note the early (3 and 6 h) and late (18 h) reduction in Mcl-1 levels. zVAD protected from Mcl-1 loss primarily at the 18 h time point
PKC-δ(D330A) prevents UV-induced translocation of the PKC-δ catalytic fragment to mitochondria. (a) Keratinocytes were infected with either Linker (L) virus or PKC-δ(D330A) (D) virus and were left untreated or exposed to UV. After 18 h, mitochondrial lysates were prepared and PKC-δ levels detected by Western blotting. Note that the 43 kDa PKC-δ catalytic fragment is not present in the mitochondria of PKC-δ(D330A)-expressing cells exposed to UV. (b) Total cell lysates prepared from keratinocytes treated as in (a) were blotted for PKC-δ. Note that PKC-δ(D330A) is not cleaved in UV-irradiated cells, and that PKC-δ(D330A) also prevented the cleavage of endogenous PKC-δ
PKC-δ(D330A) prevents the translocation of the endogenous PKC-δ catalytic fragment to the mitochondria
Since mitochondrial translocation of the active PKC-δ catalytic fragment has been implicated in its proapoptotic functions, we examined mitochondrial-enriched fractions from UV-irradiated keratinocytes for the PKC-δ cleavage product. As shown in Figure 9a, the endogenous PKC-δ catalytic fragment from Linker-infected control cells was detected in the mitochondria extracts following UV exposure, but overexpression of PKC-δ(D330A) prevented the appearance of the 43 kDa mitochondrial PKC-δ fragment. In Figure 9b, an analysis of total cell lysates demonstrates that not only is the PKC-δ(D330A) mutant not cleaved in UV-irradiated keratinocytes, but cleavage of the endogenous PKC-δ is blocked in PKC-δ(D330A)-infected cells. Thus, by not being cleaved and inhibiting the cleavage of endogenous PKC-δ, the PKC-δ(D330A) mutant was able to prevent mitochondrial accumulation of the PKC-δ catalytic fragment. Note that the ∼45 kDa bands seen in the PKC-δ(D330A) lysates are present even in the absence of UV, and thus are considered nonspecific bands.
Discussion
In human keratinocytes, the induction of apoptosis is a major cellular proofreading mechanism that eliminates damaged cells and protects from skin carcinogenesis.14, 15 Of the five PKC isoforms expressed in human keratinocytes (α, δ, ɛ, η, ζ), PKC-δ has a clear proapoptotic function11, 13 as well as an apparent tumor suppressor function since transgenic mice overexpressing PKC-δ in the epidermis are resistant to skin tumor promotion by TPA.16 Activation of overexpressed PKC-δ, or expression of the enzymatically active catalytic fragment induces apoptosis in keratinocytes.10, 11 Studies using the pan-PKC inhibitor GF109203X11, 17 have shown that inhibition of total PKC activity rescues cells from UV-induced apoptosis. In this study, we specifically inhibited the caspase-3-dependent cleavage and activation of PKC-δ by overexpressing a cleavage-resistant mutant form of PKC-δ. PKC-δ(D330A) blocked many UV-induced apoptotic end points including morphological cell death (Figure 3), sub-G1 DNA (Figures 4a and b), activation of caspase-3 and caspase-9 (Figure 6), and the loss of mitochondrial membrane potential (Figure 7). Furthermore, PKC-δ(D330A) inhibited the loss of Mcl-1 (Figure 8) and the release of cytochrome c (Figure 5), early events in the UV-apoptotic pathway.
PKC-δ is a widely expressed PKC isoform that is activated allosterically by TPA or DAG generated from lipid hydrolysis via binding to its C1 domain, or by proteolytic cleavage in its hinge region to generate an active catalytic fragment domain.6, 11, 18 Translocation of PKC-δ from the cytosol to the membrane does not occur in keratinocytes undergoing UV apoptosis;13 however, rapid translocation has been observed in JB6 cells suggestive of DAG-mediated activation.19 The PKC-δ(D330A) mutant had a functional C1 domain and could be activated by TPA but not caspase-3 (Figure 2), indicating that activation of PKC-δ by DAG at the C1 domain is not required for UV apoptosis in keratinocytes. The results from this study using the caspase cleavage resistant form of PKC-δ indicate that the activation of PKC-δ by proteolytic cleavage is the relevant mechanism of activation of PKC-δ in keratinocytes undergoing UV apoptosis.
A critical event leading to the activation of PKC-δ following UV exposure is the release of cytochrome c from the mitochondria. Inhibition of PKC-δ cleavage and activation with PKC-δ(D330A) partially inhibited cytochrome c release (Figure 5). The release of cytochrome c following UV exposure is mediated by Bax and Bak, and the early downregulation of the antiapoptotic protein Mcl-1 is also necessary.20, 21 We found that the downregulation of Mcl-1 by UV was partially prevented by PKC-δ(D330A) (Figure 8). Mcl-1 itself is a substrate of caspase-3, and some of the protection from Mcl-1 loss is probably due to inhibition of caspase activation by PKC-δ(D330A) since zVAD also protected from Mcl-1 loss at later time points.22 Thus, PKC-δ cleavage and activation can feed back to reduce the antiapoptotic protein Mcl-1 involved in initiating cytochrome c. However, because PKC-δ cleavage and activation occur after the initial cytochrome c release, protecting from Mcl-1 loss cannot completely block cytochrome c release (Figure 5).
Once cytochrome c is released, it participates in the formation of the apoptosome resulting in activation of the caspase cascade including caspase-3. Active caspase-3 then cleaves PKC-δ to generate the active catalytic fragment, which localizes to the mitochondria.10, 11 PKC-δ is involved in a positive feedback loop with caspase-3 in various cell systems,17, 23, 24, 25 and our results indicate that cleavage of PKC-δ is required. The exact mechanisms involved in the activation of caspase-3 by PKC-δ are not yet known.26 However, we showed that the caspase-3 cleavage resistant PKC-δ(D330A) inhibits the generation and translocation of the catalytic domain of PKC-δ to the mitochondria (Figure 9), and this may disrupt the positive feedback loop between the caspase activation and PKC-δ. Consistent with this, the PKC-δ(D330A) mutant was able to partially prevent caspase activation in response to UV (Figure 6). Thus, our working model positions PKC-δ as a critical caspase-3 substrate, which when cleaved, translocates to the mitochondria to enhance the release of cytochrome c and caspase-9/caspase-3 activation, thereby promoting apoptosis. The caspase-resistant PKC-δ(D330A) inhibits apoptosis by competitively inhibiting endogenous PKC-δ cleavage, thus preventing the formation and mitochondrial translocation of the active PKC-δ catalytic fragment. We estimate that at least 50% of UV-induced apoptosis requires this PKC-δ-positive feedback since we consistently see greater than 50% protection from UV apoptosis when PKC-δ cleavage and activation are prevented (Figures 4 and 7). PKC-δ activation is also associated with the loss of mitochondrial membrane potential. This is a late event in UV apoptosis, and PKC-δ(D330A) was able to protect keratinocytes from UV-induced loss of mitochondrial membrane potential.
Overexpression of PKC-δ in the epidermis can suppress skin tumor formation by TPA in mice,16 and depletion of PKC-δ in rat fibroblasts leads to tumor promotion,27 indicating that PKC-δ has a tumor suppressor function. Since activation of overexpressed full-length PKC-δ can induce apoptosis, the tumor suppression in PKC-δ transgenic mice may be due to the induction of apoptosis. In keratinocytes with rasHa activated, PKC-δ is inhibited by downregulation in human keratinocytes28, 29, 30 or by tyrosine phosphorylation through Src and Fyn in mouse keratinocytes.28, 29, 30 Inhibition of UV-induced apoptosis by PKC-δ(D330A) resulted in enhanced survival and growth of keratinocytes (Figure 3b), and thus cells with reduced or inactive PKC-δ have a selective growth advantage over cells with functional PKC-δ, consistent with the tumor suppressor function of PKC-δ. In light of our results demonstrating that the cleavage/activation of PKC-δ in keratinocytes is an important event required for UV apoptosis, the inactivation of PKC-δ by Ras activation in premalignant keratinocytes could provide a selective growth advantage to keratinocytes repeatedly exposed to genotoxic doses of UV radiation from the sun.
Materials and Methods
Antibodies and chemicals
For the detection of PKC-δ by Western blotting, a carboxyl-terminal PKC-δ antibody was used at a dilution of 1 : 4000 (sc-937, Santa Cruz Biotechnology, Santa Cruz, CA, USA). For cytochrome c detection, an antibody (#630105, Clontech, Palo Alto, CA, USA) at 1 : 500 dilution was used. A cleaved caspase-9 antibody (9501S, Cell Signaling, Beverly, MA, USA) and a caspase-3 antibody (sc-7272, Santa Cruz, CA, USA) at 1 : 500 dilution were used. For the detection of Mcl-1, an Mcl-1 antibody was used at a dilution of 1 : 500 (sc-819, Santa Cruz Biotechnology, Santa Cruz, CA, USA). For caspase assays, the fluorogenic substrate Ac-DEVD-AFC for caspase-3 (Enzyme Systems Products, Livermore, CA, USA) was prepared at 20 mM stock in Me2SO. The PKC inhibitor GF109203X (Alexis Biochemicals, San Diego, CA, USA) was used at 5 μM.
Cell culture and UV treatment
Normal human epidermal keratinocytes were isolated from neonatal foreskins following routine circumcision as previously described.13 Each experiment was performed on the cells isolated from a single foreskin and were used at passage 2. Keratinocytes were irradiated with a Panelite Unit (Ultralite Enterprises, Inc., Lawrenceville, GA, USA) equipped with four UVB bulbs (FS36T12/UVB-VHO), which have the majority of their output in the UVB range (∼65%), with minor output in the UVA and UVC wavelengths (∼34 and ∼1%, respectively). The cells were exposed with the dish lids removed for the indicated doses, with a 30 mJ/cm2 dose requiring about 1-min exposure. The UV dose was monitored with an International Light Inc. (Newburyport, MA, USA) radiometer fitted with a UVB detector. For Figure 3b, the growth of cells after UV irradiation was monitored by counting viable cells using Trypan Blue exclusion.
Retrovirus construction, production, and infection
The PKC-δ(D330A) mutant containing a mutation of Asp330 to Ala was generated using the QuikChange Site-Directed Mutagenesis kit (Stratagene, La Jolla CA, USA). The primers MFD 30: 5′-GCT GGG GAG GAT ATG CAA GCC AAC AGT GGG ACC-3′ and MFD 31: 5′-GGT CCC ACT GTT GGC TTG CAT ATC CTC CCC AGC-3′ were used with the pKS-PKC-δ template.11 The mutated base is underlined. This cleavage resistant mutant form of PKC-δ was cloned into LZRS retroviral vector between the EcoR1 and Not1 sites.31 After cloning, the insert was verified by sequencing and the presence of the mutation confirmed. For retrovirus production, LZRS PKC-δ(D330A) DNA was transfected into Phoenix-Ampho retroviral packaging cell line using calcium phosphate as described.32 Packaging cells were selected and expanded in the presence of 1 μg/ml puromycin, and virus was harvested from confluent dishes cultured for 24–48 h at 32°C. In some experiments, a kinase-inactive PKC-δ (K378A) virus was used. For infection, normal keratinocytes were plated in six-well dishes at 105 cells per well, and the following day viral supernatant was added in the presence of 4 μg/ml polybrene (hexadimethrine bromide, Sigma). The cells were infected by spinning the plates at 300 × g for 1 h at 32°C, and the viral supernatants replaced with fresh Medium 154. The day after infection, cells were washed two times with phosphate-buffered saline and fed with Medium 154.
Cell fractionation and immunoblotting
Total cell lysates for immunoblotting were prepared by lysing cells in 20 mM Tris-HCl, pH 7.5, 1% Triton X-100, 5 mM EDTA, 40 μg/ml leupeptin, 1 μM pepstatin, 1 mM AEBSF. The lysates were spun in a microcentrifuge for 10 min at 4°C, and 20 μg protein from the supernatant was taken for analysis. The proteins were run on SDS polyacrylamide gels, transferred to nitrocellulose, and stained with Ponceau S to ensure protein equal loading. The membranes were blocked with 5% milk in 20 mM Tris-HCl, pH 7.6, 150 mM NaCl (TBS), stained with primary antibody and horseradish peroxidase-conjugated secondary antibody for 1 h each and extensively washed in TBS, 0.05% Tween-20. Proteins were detected using the ECL detection kit (Amersham Life Sciences, Piscataway, NJ, USA).
For cytosol isolation, cells were trypsinized, washed in PBS, and suspended in 10 mM HEPES, pH 7.5, 250 mM sucrose, 10 mM KCl, 1.5 mM MgCl2, 1 mM EGTA, 1 mM EDTA, 1 mM dithiothreitol, and 1 × Complete Protease Inhibitor Cocktail (Boehringer Mannheim). Digitonin was added to 0.05%, and the cells were incubated for 2 min at room temperature. The cells were then spun at 750 × g for 5 min at 4°C to pellet unbroken cells and nuclei, and the supernatant was taken and spun at 10 000 × g at 4°C for 25 min. This supernatant was taken as the cytosolic fraction and the pellet was the mitochondrial fraction.
Caspase assay
For caspase assays, cells were trypsinized and combined with floating cells. The cells were lysed in 2 × caspase-lysis buffer: 25 mM HEPES-NaOH, pH 7.4, 10% sucrose, 0.1% CHAPS, 2 mM EDTA, and 5 mM dithothreitol. Cells were spun for 5 min in a microcentrifuge and then the protein concentration was estimated by the Bradford method. Intotal, 50 μg of total protein was used per sample and mixed with 2 × caspase assay buffer: 25 mM HEPES-NaOH, pH 7.4, 5 mM DTT, and 100 μM of the fluorogenic caspase-3 substrate Ac-DEVD-AFC. After incubation for 18 h at 37°C, the fluorimetric detection of cleaved AFC product was performed on CytoFlour Multi-Well Plate Reader Series 4000 (PerSeptive Biosystems) at 400 nM excitation and 530 nM emission filter.
Flow cytometry
Apoptosis was measured by propidium iodide staining and flow cytometry as previously described.13 Rhodamine 123 staining and flow cytometry to determine loss of mitochondrial membrane potential was performed as previously described.2
Annexin V staining was performed using an Annexin V-FITC kit (Beckman Coulter Inc., Fullerton, CA, USA). At 18 h after UV exposure, the floaters were combined with the cells obtained after trypsinization and spun for 5 min at 20°C. The cell pellets were then washed with ice-cold phosphate-buffered saline with Ca2+ and Mg2+. The cell pellets were then suspended in 100 μl of 1 × diluted binding buffer at 4°C. In all, 1 μl of Annexin V and 5 μl of dissolved propidium iodide were then added per sample and incubated at 4°C in the dark for 10 min. Ice-cold 1 × diluted binding buffer (400) was then added per sample tube and mixed gently. The samples were analyzed by flow cytometry immediately after Annexin V staining.
PKC-δ immunoprecipitation kinase assay
Keratinocytes were infected with LZRS-Linker virus or LZRS-PKC-δ(D330A) virus at 20% confluency. When the cells reached 80–90% confluency, they were UV irradiated with a dose of 30 mJ/cm2 or treated with 2 μM TPA for 15 min. Protein (500 μg) was used per sample for overnight immunoprecipitation with PKC-δ (sc-937, Santa Cruz Biotech, CA, USA) and a kinase reaction was performed as previously described.13
Abbreviations
- UV:
-
ultraviolet
- PKC:
-
protein kinase C
References
Thornberry NA and Lazebnik Y (1998) Caspases: enemies within. Science 281: 1312–1316
Sitailo LA, Tibudan SS and Denning MF (2002) Activation of caspase-9 is required for UV-induced apoptosis of human keratinocytes. J. Biol. Chem. 277: 19346–19352
Rehemtulla A, Hamilton CA, Chinnaiyan AM and Dixit VM (1997) Ultraviolet radiation-induced apoptosis is mediated by activation of CD-95 (Fas/APO-1). J. Biol. Chem. 272: 25783–25786
Aragane Y, Kulms D, Metze D, Wilkes G, Poppelmann B, Luger TA and Schwartz T (1998) Ultraviolet light induces apoptosis via direct activation of CD95 (Fas/APO-1) independently of its ligand CD95L. J. Cell Biol. 140: 171–182
Leverkus M, Yaar M and Gilchrest BA (1997) Fas/Fas ligand interaction contributes to UV-induced apoptosis in human keratinocytes. Exp. Cell Res. 232: 255–262
Ghayur T, Hugunin M, Talanian RV, Ratnofsky S, Quinlan C, Emoto Y, Pandey P, Datta R, Huang Y, Kharbanda S, Allen H, Kamen R, Wong W and Kufe D (1996) Proteolytic activation of protein kinase C delta by an ICE/CED 3-like protease induces characteristics of apoptosis. J. Exp. Med. 184: 2399–2404
Sakurai Y, Onishi Y, Tanimoto Y and Kizaki H (2001) Novel protein kinase C delta isoform insensitive to caspase-3. Biol. Pharm. Bull. 24: 973–977
Emoto Y, Manome Y, Meinhardt G, Kisaki H, Kharbanda S, Robertson M, Ghayur T, Wong WW, Kamen R, Weichselbaum R and Kufe D (1995) Proteolytic activation of protein kinase C delta by an ICE-like protease in apoptotic cells. EMBO J. 14: 6148–6156
Datta R, Kojima H, Yoshida K and Kufe D (1997) Caspase-3-mediated cleavage of protein kinase C theta in induction of apoptosis. J. Biol. Chem. 272: 20317–20320
Li L, Lorenzo PS, Bogi K, Blumberg PM and Yuspa SH (1999) Protein kinase C delta targets mitochondria, alters mitochondrial membrane potential, and induces apoptosis in normal and neoplastic keratinocytes when overexpressed by an adenoviral vector. Mol. Cell. Biol. 19: 8547–8558
Denning MF, Wang Y, Alkan S, Nickoloff BJ and Qin JZ (2002) Caspase activation and disruption of mitochondrial membrane potential during UV radiation-induced apoptosis of human keratinocyte requires activation of protein kinase C. Cell Death Diff. 9: 40–52
Mizuno K, Noda K, Araki T, Imaoka T, Kobayashi Y, Akita Y, Shimonaka M, Kishi S and Ohno S (1997) The proteolytic cleavage of protein kinase C isotypes, which generates kinase and regulatory fragments, correlates with Fas-mediated and 12-O-tetradecanoyl-phorbol-13-acetate-induced apoptosis. Eur. J. Biochem. 250: 7–18
Denning MF, Wang Y, Nickoloff BJ and Wrone-Smith T (1998) Protein kinase C δ is activated by caspase-dependent proteolysis during ultraviolet radiation-induced apoptosis of human keratinocytes. J. Biol. Chem. 273: 29995–30002
Ziegler A, Jonason AS, Leffell DJ, Simon JA, Sharma HW, Kimmelman J, Remington L, Jacks T and Brash DE (1994) Sunburn and p53 in the onset of skin cancer. Nature 372: 773–776
Pena JC, Rudin CM and Thompson CB (1998) A Bcl-xL transgene promotes malignant conversion of chemically initiated skin papillomas. Cancer Res. 58: 2111–2116
Reddig PJ, Dreckschimdt NE, Ahrens H, Simsiman R, Tseng CP, Zou J, Oberly TD and Verma AK (1999) Transgenic mice overexpressing protein kinase C delta in the epidermis are resistant to skin tumor promotion by 12-O-tetradecanoylphorbol-13-acetate. Cancer Res. 59: 5710–5718
Reyland ME, Anderson SM, Matassa AA, Barzen KA and Quissell DO (1999) Protein kinase C delta is essential for etoposide-induced apoptosis in salivary gland acinar cells. J. Biol. Chem. 274: 19115–19123
Stabel S and Parker PJ (1991) Protein kinase C. Pharmacol. Ther. 51: 71–95
Chen N, Ma W, Huang C and Dong Z (1999) Translocation of protein kinase C epsilon and protein kinase C delta to membrane is required for ultraviolet B-induced activation of mitogen-activated protein kinases and apoptosis. J. Biol. Chem. 274: 15389–15394
Wei MC, Zong WX, Cheng EH, Lindsten T, Panoutsakopoulou V, Ross AJ, Roth KA, MacGregor GR, Thompson CB and Korsmeyer SJ (2001) Proapoptotic BAX and BAK: a requisite gateway to mitochondrial dysfunction and death. Science 292: 727–730
Nijhawan D, Fang M, Traer E, Zhong Q, Gao W, Du F and Wang X (2003) Elimination of Mcl-1 is required for the initiation of apoptosis following ultraviolet irradiation. Genes Dev. 17: 1475–1486
Snowden RT, Sun XM, Dyer MJ and Cohen GM (2003) Bisindolylmaleimide IX is a potent inducer of apoptosis in chronic lymphocytic leukaemic cells and activates cleavage of Mcl-1. Leukemia 17: 1981–1989
Anantharam V, Kitazawa M, Wagner J, Kaul S and Kanthasamy AG (2002) Caspase-3-dependent proteolytic cleavage of protein kinase C delta is essential for oxidative stress-mediated dopaminergic cell death after exposure to methylcyclopentadienyl manganese tricarbonyl. J. Neurosci. 22: 1738–1751
Blass M, Kronfeld I, Kazimirsky G, Blumberg PM and Brodie C (2002) Tyrosine phosphorylation of protein kinase C delta is essential for its apoptotic effect in response to etoposide. Mol. Cell. Biol. 22: 182–195
Basu A and Akkaraju G (1999) Regulation of caspase activation and cis-diamminedichloroplatinum(II)-induced cell death by protein kinase C. Biochemistry 38: 4245–4251
Brodie C and Blumberg PM (2003) Regulation of cell apoptosis by protein kinase C delta. Apoptosis 8: 19–27
Lu Z, Hornia A, Jiang YW, Zang Q, Ohno S and Foster DA (1997) Tumor promotion by depleting cells of protein kinase C delta. Mol. Cell Biol. 17: 3418–3428
Denning MF, Dlugosz AA, Threadgill DW, Magnuson T and Yuspa SH (1996) Activation of the epidermal growth factor receptor signal transduction pathway stimulates tyrosine phosphorylation of protein kinase C delta. J. Biol. Chem. 271: 5325–5331
Geiges D, Marks F and Gschwendt M (1995) Loss of protein kinase C delta from human HaCaT keratinocytes upon ras transfection is mediated by TGF alpha. Exp. Cell Res. 219: 299–303
Denning MF, Dlugosz AA, Howett MK and Yuspa SH (1993) Expression of an oncogenic rasHa gene in murine keratinocytes induces tyrosine phosphorylation and reduced activity of protein kinase C delta. J. Biol. Chem. 268: 26079–26081
Kinsella TM and Nolan GP (1996) Episomal vectors rapidly and stably produce high-titer recombinant retrovirus. Hum. Gene Ther. 7: 1405–1413
Qin JZ, Bacon P, Chaturvedi V and Nickoloff BJ (2001) Role of NF-kappaB activity in apoptotic response of keratinocytes mediated by interferon-gamma, tumor necrosis factor-alpha, and tumor-necrosis-factor-related apoptosis-inducing ligand. J. Invest. Dermatol. 117: 898–907
Acknowledgements
We thank all members of the Skin Cancer Research Program for their input into this project, especially Shalini S Tibudan and Drs. Leonid A Sitailo, Jian-Zhong Qin, and Brian J Nickoloff. This work was supported in part by NIH Grant CA83784 (MF Denning).
Author information
Authors and Affiliations
Corresponding author
Additional information
Edited by B Osborne
Rights and permissions
About this article
Cite this article
D'Costa, A., Denning, M. A caspase-resistant mutant of PKC-δ protects keratinocytes from UV-induced apoptosis. Cell Death Differ 12, 224–232 (2005). https://doi.org/10.1038/sj.cdd.4401558
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.cdd.4401558
Keywords
This article is cited by
-
UV induced changes in proteome of rats plasma are reversed by dermally applied cannabidiol
Scientific Reports (2021)
-
SIRT1 activation mediates heat-induced survival of UVB damaged Keratinocytes
BMC Dermatology (2017)
-
Heat-mediated reduction of apoptosis in UVB-damaged keratinocytes in vitro and in human skin ex vivo
BMC Dermatology (2016)
-
Protein Kinase Cδ in Apoptosis: A Brief Overview
Archivum Immunologiae et Therapiae Experimentalis (2012)
-
PKC-δ and -η, MEKK-1, MEK-6, MEK-3, and p38-δ Are Essential Mediators of the Response of Normal Human Epidermal Keratinocytes to Differentiating Agents
Journal of Investigative Dermatology (2010)