Abstract
Survivin inhibits apoptosis and promotes mitosis. We determined whether nuclear or cytoplasmic localisation of survivin predicts survival of 48 patients with resected non-small-cell lung cancer (NSCLC). Patients with nuclear staining of survivin had significantly worse survival (relative risk: 3.9, P=0.02). Therefore, survivin may be a biomarker for NSCLC.
Similar content being viewed by others
Main
Survivin was initially identified as an inhibitor of apoptosis. Its expression is undetectable in most terminally differentiated normal tissues, but is at high levels in various malignancies as well as embryonic and fetal tissues (Salvesen and Duckett, 2002). Survivin expression is upregulated in all phases of cell cycle, and the cancer-specific activity of survivin promoter was detected both in vivo and in vitro (Bao et al, 2002). Deletion of survivin resulted in a catastrophic defect of microtubule assembly, with absence of mitotic spindles, disorganised tubulin aggregates and multinucleation, in the survivin knockout mice (Uren et al, 2000). Colocalisation of survivin with Aurora-B and the inner centromere protein (INCENP) suggests that these proteins interact throughout mitosis and are essential for chromosome condensation and segregation as well as the completion of cytokinesis (Adams et al, 2001). Several studies have shown that survivin is a prognostic indicator for poor survival in several malignancies (Adida et al, 2000; Chakravarti et al, 2002; Kennedy et al, 2003; Trieb et al, 2003). Survivin protein level examined by immunostaining was associated with vascular invasion and poor survival (Ikehara et al, 2002). However, the predictive value of survivin mRNA examined by RT–PCR is contradictory between two independent studies (Monzo et al, 1999, Falleni et al, 2003). Survivin staining was found in both nucleus and cytoplasm of non-small-cell lung cancer (NSCLC) (Falleni et al, 2003). Nuclear survivin rather than the cytoplasmic staining was shown to be predictive of poor survival in patients with oesophageal cancers (Grabowski et al, 2003). Since survivin has both nuclear and cytoplasmic targets and is involved in regulation of mitosis and apoptosis, we determined the predictive value of nuclear vs cytoplasmic staining of survivin among patients with resected NSCLC.
Methods
Patients
Archived tissue blocks from 1999 to 2002 were retrieved from the files of Vanderbilt University and the Nashville Veterans Affairs Medical Center pathology departments, according to the approved IRB protocol (010178). For all tissue blocks, the H&E-stained sections were reviewed by two pathologists who specialise in lung cancer. Table 1 summarises the clinical and molecular characteristics of 48 patients with NSCLC. Patients underwent surgical resection with hilar and mediastinal lymph node sampling. None of these patients received neoadjuvant chemotherapy or radiotherapy. Clinical data was obtained from the tumour registry and hospital charts at the Vanderbilt Medical Center.
Immunohistochemistry
Paraffin-embedded material was available in a set of 48 individual tumours for evaluation of nuclear and cytoplasmic staining of survivin. These studies were carried out using a standard avidin–biotin–peroxidase complex technique, with a mouse monoclonal antibody against survivin (Santa Cruz Biotech, Santa Cruz, California, sc-17779). Three separate sections from each case were examined by the immunohistochemistry method. Staining was assessed in 5–10 high-powered fields at × 400 magnification. Survivin immunoreactivity was evaluated semiquantitatively based on the intensity of staining. It was scored as: 1+ (weak); 2+ (moderate); and 3+ (intense). Cases with no or weak staining were considered negative, whereas those with moderate to intense staining were considered as positive. The highest score among the three tissue sections was entered for statistical analyses.
Statistical analysis
For lifetime data analyses, the possible risk factors, for example, nuclear survivin, were compared for survival with the Kaplan–Meier estimates and log-rank tests. The proportional hazards model was used for adjusted tests of significance and estimates of odds ratios. The unadjusted and adjusted 95% confidence intervals (CI) of survival were calculated and reported for univariate and multivariate statistical models. All tests of significance were two sided, and differences were considered statistically significant when P-value was <0.05. SAS version 8.2 and S-Plus 6 were used for all analyses.
Results
Cytoplasmic and nuclear staining of survivin in the tissue samples of NSCLC
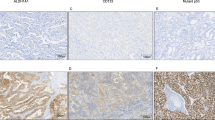
We have performed immunohistochemistry analyses on tissue cores containing NSCLCs from 48 patients who underwent surgical resections at Vanderbilt University Medical Center from 1999 to 2002. Figure 1 shows a tissue core stained with the sc-17779 mouse monoclonal antibody. As shown, prominent nuclear staining was observed. A total of 32 cases (67%) were scored as positive for nuclear staining, whereas 39 cases (83%) were scored as positive for cytoplasmic staining. A totl of 19 cases (44%) had positive staining in both cytoplasm and nucleus.
Nuclear survivin is associated with poor survival in resected NSCLC
In order to determine whether immunohistochemistry of survivin staining has any prognostic value, we examined the association between nuclear or cytoplasmic staining of survivin with overall survival among the 48 patients with resected NSCLC. No difference in survival was detected when the data was analysed by patients’ age or sex, tumour histology or grade, or lymph node status. However, T stage was significantly associated with poor survival as shown in Table 1. As for survivin staining, nuclear survivin positivity was significantly associated with poor survival (P=0.01, median survival of 248 days, 95% CI: 68–458 days), whereas cytoplasmic staining of survivin was not, as shown in Figure 2. This was confirmed by multivariate analyses, which demonstrated a relative risk of 3.9 in the patients with positive nuclear survivin (P=0.02), as shown in Table 2.
Discussion
In this study, we have found that the nuclear presence of survivin was associated with poor survival in patients with NSCLC. Both mRNA and protein levels of survivin were shown to predict unfavourable survival in patients with resected NSCLC (Monzo et al, 1999; Ikehara et al, 2002). Since survivin has dual function in apoptosis and mitosis depending upon its cellular localisation, the predictive value of nuclear vs cytoplasmic staining of survivin has been investigated in a number of malignancies. When nuclear or cytoplasmic staining of survivin was scored separately, nuclear staining was linked with favourable prognosis in gastric cancer (Okada et al, 2001), breast cancer (Kennedy et al, 2003) and osteosarcoma (Trieb et al, 2003), whereas cytoplasmic survivin was not found to be prognostic. In contrast, a recent study in oesophageal cancers showed that nuclear survivin was associated with poor survival (Grabowski et al, 2003). Our results in NCSLC also demonstrated a poor prognostic value of nuclear survivin. The prognostic difference of nuclear survivin among these studies appears to be tumoor specific. Relative importance of nuclear survivin in mitosis may vary among different tumour types and may predict differently the responses to various cancer type-specific therapies, which ultimately determine the overall survival of cancer patients. Nuclear and cytoplasmic pools of survivin have their distinct roles (Fortugno et al, 2002). It has been shown that survivin splice variants had different subcellular localisations (Mahotka et al, 2002). Survivin-delta Ex3 is preferentially localised in the nucleus, whereas survivin and survivin 2B isoforms are found in the cytoplasm. However, survivin-2B is nonantiapoptotic. It is not surprising that the cytoplasmic level is not prognostic since the IHC staining is unable to discriminate the splicing variants and represents the combined level of two survivin variants with opposing effects on apoptosis. The RNA level of survivin 2B (nonantiapoptotic) detected by RT–PCR was, however, found decreased in advanced stages of renal (Mahotka et al, 2002) and gastric cancers (Krieg et al, 2002).
In the nucleus, survivin was shown to interact with aurora B kinase and INCENP, which play essential roles in chromosomal segregation during the exit of mitosis (Honda et al, 2003). Knockdown and inhibition of survivin resulted in multinucleated and polyploid cells, which is a phenotype of mitotic arrest (Uren et al, 2000). Therefore, strong expression of survivin in the nucleus may represent increased mitotic events. On the other hand, in the cytoplasm, survivin inhibits apoptosis by blocking caspase activity. So far, it has not been demonstrated that the cytoplasmic survivin alone predicts clinical outcome. This may result from that the measurement of cytoplasmic survivin includes the combined level of two functionally opposing variants. Alternatively, other antiapoptotic molecules such as bcl-2 proteins may be more important players in lung cancers. However, a dominant negative mutant of survivin, T34A, has been shown to be effective in treating xenografts of breast cancer by freeing up caspase 9 and thus promoting apoptosis (Wall et al, 2003). This suggests that survivin remains a viable therapeutic target in certain cancers (Altieri, 2003).
In summary, nuclear presence of survivin may be an important prognostic marker for patients with resected NSCLC. Larger population studies are needed to confirm the value of nuclear staining of survivin as a prognostic marker. Further investigation should evaluate the strategies of intervening survivin function for therapeutics in lung cancer.
Change history
16 November 2011
This paper was modified 12 months after initial publication to switch to Creative Commons licence terms, as noted at publication
References
Adams RR, Carmena M, Earnshaw WC (2001) Chromosomal passengers and the (aurora) ABCs of mitosis. Trends Cell Biol 11: 49–54
Adida C, Haioun C, Gaulard P, Lepage E, Morel P, Briere J, Dombret H, Reyes F, Diebold J, Gisselbrecht C, Salles G, Altieri DC, Molina TJ (2000) Prognostic significance of survivin expression in diffuse large B-cell lymphomas. Blood 96: 1921–1925
Altieri DC (2003) Validating survivin as a cancer therapeutic target. Nat Rev Cancer 3: 46–54
Bao R, Connolly DC, Murphy M, Green J, Weinstein JK, Pisarcik DA, Hamilton TC (2002) Activation of cancer-specific gene expression by the survivin promoter. J Natl Cancer Inst 94: 522–528
Chakravarti A, Noll E, Black PM, Finkelstein DF, Finkelstein DM, Dyson NJ, Loeffler JS (2002) Quantitatively determined survivin expression levels are of prognostic value in human gliomas. J Clin Oncol 20: 1063–1068
Falleni M, Pellegrini C, Marchetti A, Oprandi B, Buttitta F, Barassi F, Santambrogio L, Coggi G, Bosari S (2003) Survivin gene expression in early-stage non-small cell lung cancer. J Pathol 200: 620–626
Fortugno P, Wall NR, Giodini A, O’Connor DS, Plescia J, Padgett KM, Tognin S, Marchisio PC, Altieri DC (2002) Survivin exists in immunochemically distinct subcellular pools and is involved in spindle microtubule function. J Cell Sci 115: 575–585
Grabowski P, Kuhnel T, Muhr-Wilkenshoff F, Heine B, Stein H, Hopfner M, Germer CT, Scherubl H (2003) Prognostic value of nuclear survivin expression in oesophageal squamous cell carcinoma. Br J Cancer 88: 115–119
Honda R, Korner R, Nigg EA (2003) Exploring the functional interactions between aurora B, INCENP, and survivin in mitosis. Mol Biol Cell 14: 3325–3341
Ikehara M, Oshita F, Kameda Y, Ito H, Ohgane N, Suzuki R, Saito H, Yamada K, Noda K, Mitsuda A (2002) Expression of survivin correlated with vessel invasion is a marker of poor prognosis in small adenocarcinoma of the lung. Oncol Rep 9: 835–838
Kennedy SM, O’Driscoll L, Purcell R, Fitz-Simons N, McDermott EW, Hill AD, O’Higgins NJ, Parkinson M, Linehan R, Clynes M (2003) Prognostic importance of survivin in breast cancer. Br J Cancer 88: 1077–1083
Krieg A, Mahotka C, Krieg T, Grabsch H, Muller W, Takeno S, Suschek CV, Heydthausen M, Gabbert HE, Gerharz CD (2002) Expression of different survivin variants in gastric carcinomas: first clues to a role of survivin-2B in tumour progression. Br J Cancer 86: 737–743
Mahotka C, Liebmann J, Wenzel M, Suschek CV, Schmitt M, Gabbert HE, Gerharz CD (2002) Differential subcellular localization of functionally divergent survivin splice variants. Cell Death Differ 9: 1334–1342
Monzo M, Rosell R, Felip E, Astudillo J, Sanchez JJ, Maestre J, Martin C, Font A, Barnadas A, Abad A (1999) A novel anti-apoptosis gene: re-expression of survivin messenger RNA as a prognosis marker in non-small-cell lung cancers. J Clin Oncol 17: 2100–2104
Okada E, Murai Y, Matsui K, Isizawa S, Cheng C, Masuda M, Takano Y (2001) Survivin expression in tumor cell nuclei is predictive of a favorable prognosis in gastric cancer patients. Cancer Lett 163: 109–116
Salvesen GS, Duckett CS (2002) IAP proteins: blocking the road to death's door. Nat Rev Mol Cell Biol 3: 401–410
Trieb K, Lehner R, Stulnig T, Sulzbacher I, Shroyer KR (2003) Survivin expression in human osteosarcoma is a marker for survival. Eur J Surg Oncol 29: 379–382
Uren AG, Wong L, Pakusch M, Fowler KJ, Burrows FJ, Vaux DL, Choo KH (2000) Survivin and the inner centromere protein INCENP show similar cell-cycle localization and gene knockout phenotype. Curr Biol 10: 1319–1328
Wall NR, O’Connor DS, Plescia J, Pommier Y, Altieri DC (2003) Suppression of survivin phosphorylation on Thr34 by flavopiridol enhances tumor cell apoptosis. Cancer Res 63: 230–235
Acknowledgements
This study was supported in part by Vanderbilt Discovery Grant and Vanderbilt Physician Scientist Grant VICC CCSG5, R01-CA88076, R01-CA89674 and the Vanderbilt Lung Cancer SPORE, P50-CA90949.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
From twelve months after its original publication, this work is licensed under the Creative Commons Attribution-NonCommercial-Share Alike 3.0 Unported License. To view a copy of this license, visit http://creativecommons.org/licenses/by-nc-sa/3.0/
About this article
Cite this article
Lu, B., Gonzalez, A., Massion, P. et al. Nuclear survivin as a biomarker for non-small-cell lung cancer. Br J Cancer 91, 537–540 (2004). https://doi.org/10.1038/sj.bjc.6602027
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.bjc.6602027
Keywords
This article is cited by
-
Evaluation of hTERT, KRT7, and survivin in urine for noninvasive detection of bladder cancer using real‐time PCR
BMC Urology (2021)
-
RETRACTED ARTICLE: The Impact of Survivin on Prognosis and Clinicopathology of Glioma Patients: A Systematic Meta-Analysis
Molecular Neurobiology (2015)
-
Nuclear survivin expression: a prognostic factor for the response to taxane–platinum chemotherapy in patients with advanced non-small cell lung cancer
Medical Oncology (2014)
-
Expression of survivin and patients survival in non-small cell lung cancer: a meta-analysis of the published studies
Molecular Biology Reports (2013)
-
Prognostic Relevance of Survivin in Pancreatic Endocrine Tumors
World Journal of Surgery (2012)