Abstract
Study design:
Experimental, controlled trial, animal study.
Objective:
To assess morphologic changes in different cartilage plates after spinal cord injury and identify the localization of these alterations.
Setting:
Saitama, Japan.
Methods:
A total of 16 Wistar rats were used. Eight rats underwent a spinal cord injury and eight rats had no intervention as control. The cartilage alterations of the knee joint were evaluated with radiography and histomorphometric analysis. To quantify cartilage alterations, we selected the histologic characteristics: thickness of the articular cartilage, number of chondrocytes, matrix staining to toluidine blue as a reflection of proteoglycan content and surface irregularity.
Results:
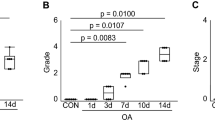
No differences in knee joints were found between the groups by radiography. In the medial knee joint, cartilage thickness of spinal-cord-injured knees increased at the anterior femoral region and decreased at the tibial and posterior femoral regions; however, in the lateral knee, that of spinal cord injuries did not change compared with control knees. Spinal cord injuries decreased the number of chondrocytes, especially at the anterior femoral regions. Matrix staining increased partially at the tibial regions. Surface irregularity of spinal-cord-injured knees was comparable to that of control knees in all cartilage plates.
Conclusion:
The present findings exhibit characteristics of the cartilage after spinal cord injury. These alterations were different in nature between the medial and lateral regions. Future studies should assess separately different cartilage plates, to overestimate these severities when the changes at the medial knee were examined and to underestimate when the changes at the lateral knee were examined.
Similar content being viewed by others
Introduction
Spinal cord injury (SCI) causes unloading and restricted movement of the lower limb joints for substantial periods of time.1, 2, 3 The joints after SCI have been described as undergoing contractures,4, 5, 6, 7 osteoarthritis,2 alterations of the periarticular connective tissue,4, 5 joint space narrowing and periarticular osteoporosis.8 Several studies have also been reported the influence of SCI on the articular cartilage of the knee joint. Enneking and Horowitz4 histologically observed the sagittal section in a human knee joint with SCI of 3 and 1–1/2 years' duration and found that the articular cartilage was normal. Unlike this report, Vanwanseele et al.2, 3 identified the progressive thinning of the human knee cartilage after SCI. Furthermore, we have recently found the changes of the cartilage thickness, the number of chondrocytes and the cartilage matrix staining in the medial knee cartilage of rats with SCI.9
Cartilage alterations after SCI have been described in relation to alterations obtained on various animal models with the suppression of mechanical forces by unloading and immobilization. The results of these animal studies have indicated that changes during immobilization may differ between different cartilage plates of the knee,10 and showed different patterns of alterations between the superficial and deep cartilage, based on the different mechanical conditions specific for each site.11 Taken together, we have previously verified the influence of mechanical forces on the articular cartilage after SCI and proposed that cartilage alterations would not be explained by only a suppression of mechanical forces;9 however, because of the high interregion variability in morphologic cartilage properties, a previous experiment in only the medial knee cartilage did not allow us to reliably negate the influence of mechanical forces.
The goal of this study was to identify the localization of cartilage alterations after SCI in different cartilage plates, including the lateral knee joint. To reach our goal, we quantified histologic articular cartilage alterations in a rat model with SCI.
Materials and methods
Experimental design
This study was carried out in accordance with the guide for the institutional committee of laboratory animals. Sixteen female Wistar rats (CLEA Japan Inc., Tokyo, Japan), 8 weeks old, weighing 170–194 g, were randomly divided into control and SCI groups. The right and the left knee joint of each animal served as different samples. Total 32 knees were assessed relative to the time after intervention, and they were 4, 8, 10, and 12 weeks.
Surgical procedures
Surgical procedures and postoperative care conformed to our previous reports.5, 6, 7, 9 The eight SCI animals were anesthetized by intraperitoneal administration of 40 mg kg−1 sodium pentobarbital. After the spinal cord was exposed by a laminectomy of the T8 vertebra, it was completely transected at the level of T8. The eight rats in the control group had no intervention.
Radiographic evaluation
At the end of the experimental period, while under general anesthesia, radiographs of the knee were taken with a digital microradiography unit (μFX-1000; Fuji Photo Film Co., Tokyo, Japan). They were exposed to the Imaging Plate (Fuji Photo Film Co.) for 10 s at 40 kV and 100 mA. The digital radiographs were obtained with an imaging analyzer (BAS-1800; Fuji Photo Film Co.).
Tissue preparation and staining
After the radiography, the animals were perfused from ascending aorta with 4% paraformaldehyde in phosphate-buffered saline at pH 7.4. The knee joints including the patella and joint capsule were resected and kept in the same fixative for an additional 18 h at 4 °C. The fixed specimens were decalcified with 10% ethylenediaminetetraacetic acid in 0.1 M tris(hydroxymethyl)aminomethane hydrochloric acid (pH 7.4) for 42–89 days at 4 °C. After decalcification, the specimens were embedded in paraffin. Standardized 5 μm serial sections were obtained at the medial and lateral mid-condylar level in the sagittal plane and then were stained with safranin O fast green or toluidine blue.
Quantitative histology
Determination of measurement sites
Femoral and tibial cartilage alterations at the medial and lateral mid-condylar level were determined at the eight regions with the methods of O'Connor (Figure 1).12 In addition, the superficial cartilage was defined as the area from the cartilage surface down to a depth of 30 μm and the deep cartilage was from the tidemark up to a depth of 30 μm.11
Diagrams of eight articular cartilage regions in femoral condyle and tibial plateau of the right knee joint. Femoral articular surface (a). Tibial articular surface (b). Cartilage regions were defined according to their positions in embedded joints where the knee joint was positioned at an angle of 125°. The anterior femoral (FA) and anterior tibial (TA) regions were defined as the regions of articular cartilage located between the inner edges of the anterior and posterior meniscal horns. The edge of the posterior femoral (FP) region was located 20 μm beyond the outer edge of the posterior meniscal horn, and the posterior tibial (TP) cartilage was located adjacent to the posterior horn of the meniscus. Knee flexion contractures develop in our models with SCI, as we have reported previously.5, 6, 7 Therefore, the FA cartilage corresponds to unapposed regions (no contact between cartilage surfaces) of the flexed knee. The FP, TA and TP regions are located at apposed regions (where the articular cartilages of two bones contact each other).9
Cartilage thickness
Cartilage thickness was defined as the distance between the cartilage surface and the osteochondral junction.9, 11, 12 Histologic sections were digitized by a × 4 microscope objective with a light microscope and a camera. At each region (anterior femoral, FA; posterior femoral, FP; anterior tibial, TA and posterior tibial, TP regions), a 1 mm long stretch of the cartilage surface was defined and the area of the cartilage under this stretch was measured, following the osteochondral junction. The mean thickness of the cartilage was calculated by dividing the area by 1 mm.
Number of cells
The number of chondrocytes at each of the eight regions was counted separately in the superficial and deep cartilage. The standardized rectangular field (30 μm deep and 400 μm long) was superimposed over the histologic sections. We counted manually chondrocytes within the rectangular field.9
Cartilage matrix staining
Under standardized light conditions, the histologic sections stained with toluidine blue were digitized as per the measurement of cartilage thickness. Rectangles (30 μm deep and 400 μm long) were superimposed to the superficial and deep cartilage. Intense metachromatic areas at 10 evenly spread points within the standardized rectangular field were measured automatically: on digitized images, intense metachromatic areas were the darkest on a gray scale, where black is 0 and white is 255.11 The average staining intensity of the 10 points constituted the matrix staining.
Cartilage surface irregularity
Quantitative histologic measures as established by Trudel et al.13 were modified and used in determining the irregularity of articular cartilage surface after SCI. The cartilage contour files were opened with LabVIEW 8.5 (National Instruments Japan Co., Tokyo, Japan) using programs developed by the author. We measured the distances between a reference point and each pixel in digitized images of a cartilage contour line and then calculated the absolute value of the second derivative of these distances. Portions of the cartilage contour line that exceeded a threshold of 8.2 μm per pixel2 were considered irregular.
Statistical analysis
Statistical analyses were conducted with SPSS 15.0J for Windows (SPSS Japan Inc., Tokyo, Japan). All values in the text and table are presented here as median with interquartile ranges. Differences between the control and SCI groups were compared at each time point by Mann–Whitney U-test. We assessed over time the differences within each group by using the Kruskal–Wallis test with post hoc Steel–Dwass tests. An α of less than 0.05 was chosen as the significance level for all statistical analyses.
Results
Radiographic findings
No narrowing of the joint space, overgrowth of the epiphyses, and osteophyte were observed in knee joints after SCI, and there was not a different appearance from the control knees (Figure 2). However, bone atrophy of the femur and the tibia in the SCI group, especially the proximal tibia after 8 weeks, were detected compared to that of the control group.
Cartilage thickness
The mean cartilage thickness for the FA, FP, TA and TP regions of the femur and tibia at the medial and lateral level are shown in Table 1. At the medial mid-condylar region of the knee, articular cartilage at the FA region was significantly thicker in the SCI group than in the control group at all time points (P<0.05; Figure 3). Conversely, the FP cartilage was thinner in SCI rats than in control rats at all time points (P=0.02; Figure 3), the TA cartilage was thinner at 4 and 8 weeks (P=0.04), and TP cartilage was thinner at 4 and 12 weeks (P<0.05). At the lateral regions of the knee, in contrast with the medial regions, neither femoral nor tibial cartilage of the SCI knees presented any significant differences in thickness compared with control knees at any time point from 4 to 12 weeks (P>0.05; Figure 3).
Photomicrographs of the medial (10 weeks) and lateral (12 weeks) femoral articular cartilage in rats. At the medial mid-condylar histologic sections, the anterior femoral (FA) cartilage was thicker in the SCI group than in the control group. The thickness at the medial posterior femoral (FP) region decreased, and the subchondral bone penetrating into the cartilage was prominent. No differences between the groups were observed at the lateral regions. Toluidine blue staining. Scale bar=200 μm.
Number of chondrocytes
Table 2 shows the results of the number of chondrocytes at the FA, FP, TA and TP regions in the superficial and deep cartilage. At the medial histologic sections, the FA cartilage in SCI knees contained significantly fewer chondrocytes than in control knees at 8 and 10 weeks in the superficial cartilage and at 4, 8 and 12 weeks in the deep cartilage (all P<0.03). SCI also decreased the number of chondrocytes at the TA region (P=0.019) and the TP region (P=0.02) (8 weeks in the superficial cartilage and 10 weeks in the deep cartilage, respectively). The number of chondrocytes in the SCI group increased only at 4 weeks in the FP superficial cartilage when compared with the control group (P=0.02). At the lateral regions, we counted significantly fewer chondrocytes in SCI knees at 4, 8 and 10 weeks in the FA superficial cartilage (P<0.05).
Matrix staining intensity
Cartilage matrix of SCI knees stained significantly more than the matrix of control knees at the medial TA region of deep cartilage (4 weeks, 26.3 (23.1, 29.2) vs 35.8 (33.1, 37.8); 10 weeks, 24.8 (23.3, 25.5) vs 30.3 (28.3, 32.3), P=0.04), at the medial TP region of deep cartilage (4 weeks, 27.3 (24.4, 29.3) vs 37.8 (35.9, 41.0), P=0.02) and at the lateral TP region of superficial cartilage (4 weeks, 22.6 (21.8, 23.3) vs 27.6 (25.5, 28.9), P=0.04). At the medial FP region, SCI matrices stained less than did that of the control knees only at 10 weeks in the superficial cartilage (49.7 (47.0, 53.6) vs 33.9 (32.4, 34.4), P=0.02).
Surface irregularity
SCI did not affect the smooth surface of articular cartilage when compared with control group at any time point after intervention (P>0.05; results not shown).
Discussion
The objective of this study was to quantify the morphologic changes of knee cartilage after SCI and identify the localization of cartilage alterations. Our findings indicate that cartilage alterations after SCI may differ between different cartilage plates and differ from alterations obtained on animal models with the suppression of mechanical forces.
Radiologic assessment revealed that no narrowing of the joint space was observed in the SCI group, whereas bone atrophy of the femur and the tibia was detected compared to that of the control group. This result is congruent with the bony changes reported in SCI animal studies.14 Only two prior radiographic studies have attempted to evaluate the influence of paralysis on the human articular cartilage in the lower extremity joints.8, 15 In one study, radiographic evidence of narrowing of the hip joint space by >50% in 25 of 200 patients with flaccid paralysis was identified.15 The other study showed joint space narrowing and periarticular osteoporosis in patients with neuromuscular disorders.8 However, radiography cannot directly visualize cartilage, nor can it discriminate between different cartilage plates in the knee.2, 3
We have found cartilage thickness changes only in the medial knee, without changes in the lateral knee, after SCI. The mean thickness of the medial knee joint after SCI increased at unapposed regions (FA region) and decreased at apposed regions (FP, TA and TP regions). On the basis of mechanical forces, evidence has been reported that cartilage becomes thicker at weight-bearing regions of a joint16 and atrophies at regions subjected to decreased loading.17 Results of this study are in contrast with these studies. The thinning of the human cartilage in paralytic joints has been advocated over the years.2, 3, 8, 15 Vanwanseele et al.2, 3 measured the thickness of the human knee cartilage after SCI using three-dimensional magnetic resonance imaging. After 6 months of injury, the mean cartilage thickness was less in the medial tibia but not in the lateral tibia of SCI patients compared with age-matched healthy volunteers.3 Although this thinning is compatible with those found in this study, we further confirmed contrasting differences between the FA and FP regions of the medial knee joint.
We have shown that the number of chondrocytes decreases especially at the FA region (unapposed regions) in both the superficial and deep cartilage of the medial knee joint and in the superficial cartilage of the lateral knee after SCI. Thaxter et al.18 reported fewer chondrocytes at apposed regions but not at unapposed regions after unloading with immobilization. Trudel et al.11 found that based on the different mechanical conditions immobilized knees harbored fewer chondrocytes in the superficial cartilage at apposed regions and in the deep cartilage at unapposed regions. In either case, these results are not also in line with ours.
Decreased matrix density with toluidine blue staining strongly correlates with a decrease in proteoglycan content.11 Depletion of proteoglycan is a common feature of cartilage alterations observed in various animal models with the suppression of mechanical forces.11 Earlier, we reported that cartilage matrix of medial SCI tibia stained less than that of control at 8 and 12 weeks in the whole uncalcified cartilage combined with the superficial and deep cartilage.9 However, in this study, we measured separately the superficial and deep cartilage, and found that SCI increased the proteoglycan content in the deep cartilage of the medial tibial regions at 4 and 10 weeks. Experimentally, excessive forces were detrimental to cartilage, but milder forces led to early secretion of aggrecan mRNA.19 In the SCI rat, the mechanical forces related to spasticity (involuntary movements and agonist–antagonist co-contractions)5, 6, 7 may be milder forces for cartilage.
Surface irregularity is one consistent feature of cartilage alterations resulting from various diseases associated with the suppression of mechanical forces.13 Trudel et al.13 designed the methods applied here to quantify cartilage surface irregularity and found that irregularities appeared after only 2 weeks of immobilization, continued to increase 4 and 8 weeks after intervention and plateaued thereafter. However, at both the medial and lateral region of the knee joint after SCI, no surface irregularities of cartilage have been observed.
Finsterbush and Friedman20 compared articular changes after peripheral denervation with those after immobilization and found specific changes caused by neurectomy. The present findings exhibit characteristics of the articular cartilage after SCI and would not be explained by only a suppression of mechanical forces. Further studies will therefore have to clarify the pathogeneses of cartilage alterations, including an influence through nerve tissues.
SCI caused cartilage alterations that were very different in nature between the medial and lateral knee, and that were more severe at the medial regions and were milder at the lateral regions. Although our results can be applied to humans with SCI remains controversial, different patterns of alterations between different cartilage plates may cause an alteration in the congruity of the joint surfaces, and thus cause changes in the stress distribution throughout the joint. Why cartilage alterations differ between different cartilage plates, however, is unclear but warrants examination. Future research should assess separately different cartilage plates, to overestimate these severities when the changes at the medial knee were examined and to underestimate when the changes at the lateral knee were examined.
References
Whiteneck GG, Charlifue SW, Frankel HL, Fraser MH, Gardner BP, Gerhart KA et al. Mortality, morbidity, and psychosocial outcomes of persons spinal cord injured more than 20 years ago. Paraplegia 1992; 30: 617–630.
Vanwanseele B, Eckstein F, Knecht H, Spaepen A, Stussi E . Longitudinal analysis of cartilage atrophy in the knees of patients with spinal cord injury. Arthritis Rheum 2003; 48: 3377–3381.
Vanwanseele B, Eckstein F, Knecht H, Stussi E, Spaepen A . Knee cartilage of spinal cord-injured patients displays progressive thinning in the absence of normal joint loading and movement. Arthritis Rheum 2002; 46: 2073–2078.
Enneking WF, Horowitz M . The intra-articular effects of immobilization on the human knee. J Bone Joint Surg Am 1972; 54: 973–985.
Moriyama H, Yoshimura O, Kawamata S, Takemoto H, Saka Y, Tobimatsu Y . Alteration of knee joint connective tissues during contracture formation in spastic rats after an experimentally induced spinal cord injury. Connect Tissue Res 2007; 48: 180–187.
Moriyama H, Yoshimura O, Sunahori H, Tobimatsu Y . Comparison of muscular and articular factors in the progression of contractures after spinal cord injury in rats. Spinal Cord 2006; 44: 174–181.
Moriyama H, Yoshimura O, Sunahori H, Nitta H, Imakita H, Saka Y et al. Progression and direction of contractures of knee joints following spinal cord injury in the rat. Tohoku J Exp Med 2004; 204: 37–44.
Richardson ML, Helms CA, Vogler III JB, Genant HK . Skeletal changes in neuromuscular disorders mimicking juvenile rheumatoid arthritis and hemophilia. AJR Am J Roentgenol 1984; 143: 893–897.
Moriyama H, Yoshimura O, Kawamata S, Takayanagi K, Kurose T, Kubota A et al. Alteration in articular cartilage of rat knee joints after spinal cord injury. Osteoarthritis Cartilage 2008; 16: 392–398.
Leroux MA, Cheung HS, Bau JL, Wang JY, Howell DS, Setton LA . Altered mechanics and histomorphometry of canine tibial cartilage following joint immobilization. Osteoarthritis Cartilage 2001; 9: 633–640.
Trudel G, Himori K, Uhthoff HK . Contrasting alterations of apposed and unapposed articular cartilage during joint contracture formation. Arch Phys Med Rehabil 2005; 86: 90–97.
O'Connor KM . Unweighting accelerates tidemark advancement in articular cartilage at the knee joint of rats. J Bone Miner Res 1997; 12: 580–589.
Trudel G, Himori K, Goudreau L, Uhthoff HK . Measurement of articular cartilage surface irregularity in rat knee contracture. J Rheumatol 2003; 30: 2218–2225.
Okumura H . Experimental osteoporosis in the rat: immobilization osteoporosis. In: Takahashi E (ed). Handbook of Bone Morphometry, 2nd edn. Nishimura: Niigata, 1991, pp 134–141.
Pool Jr WH . Cartilage atrophy. Radiology 1974; 112: 47–50.
Johnson LC . Kinetics of osteoarthritis. Lab Invest 1959; 8: 1223–1241.
Bullough PG . The geometry of diarthrodial joints, its physiologic maintenance, and the possible significance of age-related changes in geometry-to-load distribution and the development of osteoarthritis. Clin Orthop Relat Res 1981; 156: 61–66.
Thaxter TH, Mann RA, Anderson CE . Degeneration of immobilized knee joints in rats; histological and autoradiographic study. J Bone Joint Surg Am 1965; 47: 567–585.
Sironen R, Elo M, Kaarniranta K, Helminen HJ, Lammi MJ . Transcriptional activation in chondrocytes submitted to hydrostatic pressure. Biorheology 2000; 37: 85–93.
Finsterbush A, Friedman B . The effect of sensory denervation on rabbits' knee joints. A light and electron microscopic study. J Bone Joint Surg Am 1975; 57: 949–956.
Acknowledgements
This study was supported in part by Grants-in-Aid for Young Scientists (19700443) from the Japanese Ministry of Education, Culture, Sports, Science, and Technology.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Moriyama, H., Nishihara, K., Hosoda, M. et al. Contrasting alteration patterns of different cartilage plates in knee articular cartilage after spinal cord injury in rats. Spinal Cord 47, 218–224 (2009). https://doi.org/10.1038/sc.2008.96
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sc.2008.96
Keywords
This article is cited by
-
Effects of hypertonia on contracture development in rat spinal cord injury
Spinal Cord (2019)
-
Effects of aging and exercise training on the histological and mechanical properties of articular structures in knee joints of male rat
Biogerontology (2012)
-
Severe Spinal Cord Injury Causes Immediate Multi-cellular Dysfunction at the Chondro-Osseous Junction
Translational Stroke Research (2011)