Abstract
Highly active anti-retroviral therapy (HAART) is very effective in suppressing HIV-1 replication in patients. However, continuous HAART is required to prevent viral rebound, which may have detrimental effects in various tissues, including persistent neuroinflammation in the central nervous system (CNS). Here, we show that quercetin (3,5,7,3’,4’-pentahydroxy flavones), a natural antioxidant used in Chinese traditional medicines, suppresses the neuroinflammation that is induced by chronic exposure to Zidovudine (azidothymidine, AZT), a nucleoside reverse transcriptase inhibitor (NRTI) that is commonly part of HAART regimens. We found that the up-regulation of pro-inflammatory cytokines and microglial and astrocytic markers induced by AZT (100 mg/kg/day; 8 days) was significantly inhibited by co-administration of quercetin (50 mg/kg/day) in the mouse cortex, hippocampus and spinal cord. We further showed that quercetin attenuated AZT-induced up-regulation of Wnt5a, a key regulator of neuroinflammation. These results suggest that quercetin has an inhibitory effect on AZT-induced neuroinflammation in the CNS, and Wnt5a signaling may play an important role in this process. Our results may further our understanding of the mechanisms of HAART-related neurotoxicity and help in the development of effective adjuvant therapy.
Similar content being viewed by others
Introduction
There are over 36.9 million people living with HIV (PLWH) (WHO, 2015). Highly active antiretroviral therapy (HAART) is a standard anti-HIV treatment, which has effectively transformed this previously deadly viral infection into a manageable chronic disease1. However, in order to suppress viral replication, PLWH need to stay on HAART continuously.
Neurotoxicity is a critical HAART-related side effect in the nervous system2,3,4,5. HAART-related neural damage may directly contribute to the disorders that commonly develop in PLWH6,7. Hence, effective adjuvant therapy is an urgent need to prevent or reverse HAART-related neurotoxicity. Towards this end, it is important to understand the mechanism by which HAART causes toxicity in the nervous system.
Neuroinflammation, indicated by glial activation and pro-inflammatory cytokine up-regulation, is thought to contribute to the pathogenesis of neurotoxicity via various pathways2,8,9,10,11. Previous studies showed that compared with naïve PLWH, HAART-treated PLWH had more active microglia in the hippocampus and basal ganglia, indicating a contribution of HAART to neuroinflammation5. Consistent with this notion, we recently showed that long-term administration of NRTIs [e.g. zidovudine (AZT), lamivudine (3TC) and stavudine (D4T)], the backbone components of HAART regimens, significantly up-regulated pro-inflammatory cytokines in the mouse CNS12.
We are interested in testing the possibility of Chinese traditional medicines as potential HAART adjuvants. Specifically, in this study, we determined the effect of quercetin, a component of Chinese skullcap, on AZT-induced neuroinflammation in the CNS. Quercetin is a natural antioxidant13, has anti-neuroinflammatory activity14,15,16,17, and can inhibit pro-inflammatory cytokine release and microglia activation both in vivo and in vitro18,19,20,21. By immunoblotting and immunohistochemistry analyses of pro-inflammatory cytokines (IL-1β and IL-6) and glial markers (GFAP and CD11b) in different CNS regions from mice, we found that quercetin significantly attenuated AZT-induced up-regulation of the pro-inflammatory cytokines and activation of microglia and astrocytes. In addition, we also found that quercetin inhibited AZT-induced up-regulation of Wnt5a, which is an upstream regulator of NRTI-induced neuroinflammation12. These results suggest that quercetin is a potential HAART adjuvant that can reverse and attenuate the neurotoxicity caused by AZT and probably other NRTIs.
Results
Quercetin inhibits AZT-induced cytokine up-regulation in the CNS
Our previous studies revealed that administration of AZT for 5 days caused the up-regulation of IL-1β, IL-6 and TNF-α in the cortex, the hippocampus and the spinal cord12. These cytokines have been implicated in neuroinflammation and inflammation-related diseases22,23,24,25,26,27,28,29,30,31. Thus, we wanted to test whether quercetin (>95% purity), a natural flavonoid32,33,34,35,36,37 with reported anti-inflammatory activity38,39,40,41,42, would inhibit the up-regulation of these cytokines that was induced by a representative NRTI, AZT. C57BL/6 mice (male, 8 weeks) were injected subcutaneously with 100 mg/kg/day AZT for 5 days or 8 days. CNS tissues (cerebral cortices, hippocampi and spinal cords) were collected for Western blotting at the end of the drug treatment periods. The pilot experiments showed that cytokine up-regulation was more obvious at 8 days. Therefore, we treated C57BL/6 mice (male, 8 weeks) with 100 mg/kg/day AZT with or without 50 mg/kg/day quercetin) co-administration for 8 days.
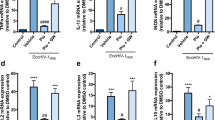
Western blotting showed that quercetin attenuated AZT-induced IL-1β and IL-6 expression significantly in cerebral cortices, hippocampi and spinal cords (Fig. 1). Most notably among these CNS regions, quercetin completely blocked the cytokine up-regulation in cortices (Fig. 1A). Partial but significant inhibitory effects of quercetin were also observed in hippocampi (Fig. 1B) and spinal cords (Fig. 1C). These data indicate that quercetin can suppress the expression of cytokines induced by AZT in different CNS regions.
Quercetin inhibited AZT-induced up-regulation of cytokines in the CNS. Expression levels of IL-1β and IL-6 in the cortices (A), hippocampi (B) and spinal cords (C) of mice treated with AZT and quercetin for 8 days. Data are expressed as means ± SEM from at least 8 mice per group (*p < 0.05, **p < 0.01, ***p < 0.001). The cropped blots displayed in the main are presented in full in Supplementary Figure 1.
Quercetin attenuates activation of astrocytes induced by AZT in the CNS
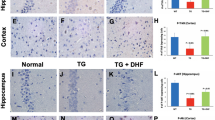
Activation of astrocytes and microglia is a cellular hallmark of neuroinflammation. AZT has been implicated in astrocyte reaction in PLWH5. Reactive astrocytes and microglia are probably the major sources of cytokines in the CNS with neuroinflammation9,43,44,45,46. We next tested the effect of quercetin on activation of astrocytes and microglia. We observed that the AZT treatment led to significant GFAP up-regulation in the cortex (Fig. 2A), the hippocampus (Fig. 2B) and the spinal cord (Fig. 2C), indicating that AZT induced the astrocyte reaction. Importantly, we also observed that the co-administration of quercetin significantly attenuated the GFAP up-regulation in all of the CNS regions examined (Fig. 2A–C). Immunocytochemistry staining experiments confirmed that AZT treatment led to an increase of GFAP-positive cells in the hippocampus (Fig. 2D) and the spinal cord (Fig. 2E) and that quercetin blocked this increase (Fig. 2D,E). These observations suggest that quercetin inhibits AZT-induced astrocyte activation.
Quercetin attenuates the AZT-induced activation of astrocytes in the CNS. Expression levels of GFAP in the cortices (A), hippocampi (B) and spinal cords (C) of mice treated with AZT and quercetin for 8 days. Data are expressed as means ± SEM from at least 8 mice per group (*p < 0.05, **p < 0.01, ***p < 0.001). Immunocytochemistry staining of GFAP in the hippocampi (D) and spinal cords (E) of mice treated with AZT and quercetin for 8 days. Scale bars: 100 μm. The cropped blots are presented in full in Supplementary Figure 2.
The analysis of the expression of microglial marker CD11b by immunoblotting (Fig. 3A–C) and immunocytochemistry (Fig. 3D,E) showed that AZT induced significant microglial activation in the spinal cord (Fig. 3C,E) but not in the cortex or the hippocampus (Fig. 3A,B,D). We also observed that quercetin blocked the microglial activation that was induced by AZT in the spinal cord (p < 0.05) (Fig. 3C,E) without affecting the basal level of CD11b expression in the cortex (Fig. 3A) or the hippocampus (Fig. 3B,D).
Effects of quercetin on microglia. Expression levels of CD11b in the cortices (A), hippocampi (B) and spinal cords (C) of mice treated with AZT and quercetin for 8 days. Data are expressed as means ± SEM from at least 8 mice per group (*p < 0.05, **p < 0.01, ***p < 0.001). Immunocytochemistry staining of CD11b in the hippocampus (D) and the spinal cord (E). Scale bars: 100 μm. The cropped blots are presented in full in Supplementary Figure 3.
Quercetin inhibits Wnt5a up-regulation induced by AZT in the CNS
Previous studies have indicated that Wnt5a is a crucial regulator of neuroinflammation in different experimental systems47,48,49. Because of our recent findings on the NRTI-induced Wnt5a up-regulation in the mouse CNS12, we sought to determine the potential effect of quercetin on AZT-induced Wnt5a expression in the CNS. We found that, consistent with the previous report12, AZT treatment (8 days) induced Wnt5a up-regulation in cerebral cortices (Fig. 4A), hippocampi (Fig. 4B) and spinal cords (Fig. 4C). In contrast, co-treatment with quercetin significantly attenuated the AZT-induced Wnt5a expression (Fig. 4A–C). These results suggest that quercetin can inhibit the AZT-induced Wnt5a up-regulation in the CNS.
Quercetin inhibits the AZT-induced up-regulation of Wnt5a in the CNS. Expression levels of Wnt5a in the cortices (A), the hippocampi (B) and the spinal cords (C) of mice treated with AZT and quercetin for 8 days. Data are expressed as means ± SEM from at least 8 mice per group (*p < 0.05, **p < 0.01, ***p < 0.001). The cropped blots are presented in full in Supplementary Figure 4.
Discussion
In this study, we tested the effect of quercetin on AZT-induced neuroinflammation in the CNS. Although quercetin was reported to effectively inhibit neuroinflammation in various animal models16,20,50,51, our work expanded this inhibitory effect of quercetin to AZT-induced neuroinflammation. Our results show that quercetin inhibits the expression of inflammatory cytokines and astrocyte reaction that are induced by AZT in different CNS regions. Since other NRTIs also cause neuroinflammation12,52, it will be interesting to determine the effect of quercetin in other NRTI and non-NRTI HAART components.
It is currently unclear how quercetin inhibits AZT-induced neuroinflammation in the CNS. Quercetin has a broad inhibitory effect on the overexpressed cyclooxygenase-2 and inducible nitric oxide synthase, which are induced by activated astrocytes and microglia, in addition to blocking neuroinflammation20,53,54. Furthermore, quercetin inhibits the overexpression of β-catenin and interrupts the canonical Wnt pathway, which has important regulatory roles in neuroinflammation55,56,57. Since we showed previously that NRTIs, including AZT, cause neuroinflammation in the CNS in a Wnt5a signaling-dependent manner12, we also looked at Wnt5a expression in this paper. Here, our results indicate that quercetin also attenuates AZT-induced Wnt5a up-regulation (Fig. 4). As the inhibitory effect of quercetin on Wnt5a is similar to that of the Wnt5a antagonist BOX 512, we propose that the mechanisms of quercetin and BOX 5 inhibition on Wnt5a expression are similar. Further experiments will be needed to confirm this. Taken together, we propose that quercetin attenuates AZT-induced neuroinflammation by inhibiting Wnt5a.
Neuroinflammation is implicated in the pathogenesis of various neurological disorders, including neurodegeneration and pathological pain58,59,60,61,62,63,64,65. Thus, NRTI-induced neuroinflammation may crucially contribute to the development of neurological deficits in PLWH who are on HAART. Indeed, ddC, an NRTI, is known to cause pain in animal models by a TNF-α-regulated mechanism66. Our data indicate that systematic administration of quercetin can effectively suppress AZT-induced neuroinflammation in different CNS regions. This indicates that quercetin can sufficiently cross the blood-brain barrier. Taken together, these findings illustrate the exciting possibility that quercetin may be a potential adjuvant that can be co-administered with HAART to protect the CNS.
Materials and Methods
Animals
C57BL/6 mice (male, 8 weeks) were purchased from Shanghai Ling Chang Biological Technology Co., Ltd. Procedures were carried out after the approval from the Zhejiang Sci-Tech University Experimental Animal Ethics Committee, and experiments were in line with relevant standards and regulations to reduce the suffering of the animals.
AZT and quercetin
AZT (Selleck, S2579) and quercetin (Sigma, Q4951-10G) were dissolved in sterilized PBS to a final concentration of 8 mg/ml and 4 mg/ml, respectively, and stored at −20 °C.
Subcutaneous (SC) injection
Mice were randomly divided into three groups (n ≥ 8). Group-1 mice were treated with PBS for 8 days (vehicle), group-2 were treated with AZT (100 mg/kg/day) only (AZT), and group-3 were co-treated with quercetin (50 mg/kg/day) and AZT (100 mg/kg/day) (A + Q). The doses of AZT12 and quercetin67,68,69 were based on previous studies. SC injection was performed at a volume of 0.25 ml/20 g/day. SC sites were sterilized with 75% alcohol wipes before injection. After injection, gentle pressure was applied to the SC injection site with the alcohol wipe for a moment to prevent the drug from leaking back through the injection site.
Western blotting and antibodies
The antibodies used are anti-IL-6 (1:2500, Abcam, ab7737); anti-IL-1β (1:5000, R&D, AF-401-NA); anti-Wnt5a (1:2500, Abcam, ab72583); anti-CD11b (1:2500, Abcam, ab133357); anti-GFAP (1:10000, Millipore, MAB360); anti-GAPDH (1:10000, Abcam, ab181602); anti-α-tubulin (1:10000, Proteintech, 66031-1-lg); Goat Anti-Rabbit IgG H&L (HRP) (1:30000, Abcam, ab97051) and Goat Anti-Mouse IgG H&L (HRP) (1:30000, Abcam, ab97023).
Western blotting and quantification
Mice were anesthetized with inhaled ether and decapitated. Cerebral cortex and hippocampus: Brains were isolated rapidly and then cerebral cortices and hippocampi were dissected carefully. Spinal cord: The whole spines of the mice were exposed, cut near the tail cavity, and pushed out the spinal cord with cold 0.01 M PBS, pH 7.4. Tissues were frozen in liquid nitrogen immediately after collection and stored at −80 °C for western blotting.
Tissues were homogenized in cell lysis buffer (Beyotime, P0013) containing 1 mM PMSF (Sigma, P7626-5G). After centrifugation (12,000 g), the protein concentration in the supernatant was determined with the BCA Protein Assay Kit (BIOMIGA, PW0104) according to manufacturer’s instructions. Boiled total protein (40–60 μg) was loaded on 12% SDS-PA gels. After electrophoresis (120 V for 90–120 min), the protein was blotted onto PVDF membranes (100 V for 90–120 min) at 4 °C. The membrane was blocked with 5% skim milk in TBST (20 mM Tris-HCl, 150 mM NaCl, 0.05% Tween-20) for 2 h at room temperature (RT). Blocked membrane was incubated with primary antibodies in TBST for 2 h at RT. After washing with TBST (3 × 10 min), membranes were incubated with anti-mouse or anti-rabbit secondary antibodies conjugated with HRP in TBST for 1 h at RT and then washed with TBST (3 × 10 min). Protein bands were visualized by Western Bright ECL (Advansta, K-12045-D10) according to the manufacturer’s instructions. GAPDH or tubulin was blotted as a loading control.
Immunohistochemistry
Mice were anesthetized with inhaled ether and perfused transcardially with PBS. Brains were isolated rapidly, and spinal cords were dissected as above. Tissues were fixed in paraformaldehyde (PFA; 4% in 0.1 M PBS) for 40 min, and paraffin sections were prepared as usual.
Paraffin sections were deparaffinized in xylene and rehydrated in ddH2O with different concentrations of ethanol (100%, 95%, 80%, and 70%). Deparaffinized sections were treated with 0.3% H2O2 to block endogenous peroxidase. After incubation with EDTA buffer (pH 8.0) to repair antigen (95–100 °C for 20 min), the sections were incubated with anti-GFAP (1:800) or anti-CD11b (1:200) in 1% BSA for 1 h at RT. Then, the sections were incubated with secondary antibody for 30 min and stained with DAB (Gene Technology). Finally, the sections were rinsed first with hematoxylin and then with ddH2O, dehydrated and mounted for imaging.
References
Ghosh, A. K., Osswald, H. L. & Prato, G. Recent Progress in the Development of HIV-1 Protease Inhibitors for the Treatment of HIV/AIDS. Journal of medicinal chemistry 59, 5172–5208, https://doi.org/10.1021/acs.jmedchem.5b01697 (2016).
Shah, A. et al. Neurotoxicity in the Post-HAART Era: Caution for the Antiretroviral Therapeutics. Neurotox Res 30, 677–697, https://doi.org/10.1007/s12640-016-9646-0 (2016).
Xu, H. et al. Lamivudine/telbivudine-associated neuromyopathy: neurogenic damage, mitochondrial dysfunction and mitochondrial DNA depletion. Journal of clinical pathology 67, 999–1005, https://doi.org/10.1136/jclinpath-2013-202069 (2014).
Vivithanaporn, P., Asahchop, E. L., Acharjee, S., Baker, G. B. & Power, C. HIV protease inhibitors disrupt astrocytic glutamate transporter function and neurobehavioral performance. Aids 30, 543–552, https://doi.org/10.1097/QAD.0000000000000955 (2016).
Anthony, I. C., Ramage, S. N., Carnie, F. W., Simmonds, P. & Bell, J. E. Influence of HAART on HIV-related CNS disease and neuroinflammation. Journal of neuropathology and experimental neurology 64, 529–536 (2005).
Caruana, G. et al. The burden of HIV-associated neurocognitive disorder (HAND) in post-HAART era: a multidisciplinary review of the literature. European review for medical and pharmacological sciences 21, 2290–2301 (2017).
Xu, J. Q. & Ikezu, T. The Comorbidity of HIV-Associated Neurocognitive Disorders and Alzheimer’s Disease: A Foreseeable Medical Challenge in Post-HAART Era. J Neuroimmune Pharm 4, 200–212, https://doi.org/10.1007/s11481-008-9136-0 (2009).
Smith, D. B., Simmonds, P. & Bell, J. E. Brain viral burden, neuroinflammation and neurodegeneration in HAART-treated HIV positive injecting drug users. Journal of neurovirology 20, 28–38, https://doi.org/10.1007/s13365-013-0225-3 (2014).
Harry, G. J. & Kraft, A. D. Neuroinflammation and microglia: considerations and approaches for neurotoxicity assessment. Expert opinion on drug metabolism & toxicology 4, 1265–1277, https://doi.org/10.1517/17425255.4.10.1265 (2008).
Rehman, S. U., Shah, S. A., Ali, T., Chung, J. I. & Kim, M. O. Anthocyanins Reversed D-Galactose-Induced Oxidative Stress and Neuroinflammation Mediated Cognitive Impairment in Adult Rats. Molecular neurobiology 54, 255–271, https://doi.org/10.1007/s12035-015-9604-5 (2017).
Doty, K. R., Guillot-Sestier, M. V. & Town, T. The role of the immune system in neurodegenerative disorders: Adaptive or maladaptive? Brain research 1617, 155–173, https://doi.org/10.1016/j.brainres.2014.09.008 (2015).
Wu, T. et al. Nucleoside reverse transcriptase inhibitors (NRTIs) induce proinflammatory cytokines in the CNS via Wnt5a signaling. Sci Rep 7, 4117, https://doi.org/10.1038/s41598-017-03446-w (2017).
Costa, L. G., Garrick, J. M., Roque, P. J. & Pellacani, C. Mechanisms of Neuroprotection by Quercetin: Counteracting Oxidative Stress and More. Oxidative medicine and cellular longevity 2016, 2986796, https://doi.org/10.1155/2016/2986796 (2016).
Suganthy, N., Devi, K. P., Nabavi, S. F., Braidy, N. & Nabavi, S. M. Bioactive effects of quercetin in the central nervous system: Focusing on the mechanisms of actions. Biomedicine & pharmacotherapy = Biomedecine & pharmacotherapie 84, 892–908, https://doi.org/10.1016/j.biopha.2016.10.011 (2016).
Rinwa, P. & Kumar, A. Quercetin suppress microglial neuroinflammatory response and induce antidepressent-like effect in olfactory bulbectomized rats. Neuroscience 255, 86–98, https://doi.org/10.1016/j.neuroscience.2013.09.044 (2013).
Testa, G. et al. Loading into Nanoparticles Improves Quercetin’s Efficacy in Preventing Neuroinflammation Induced by Oxysterols. PloS one 9, https://doi.org/10.1371/journal.pone.0096795 (2014).
Spencer, J. P. E., Vafeiadou, K., Williams, R. J. & Vauzour, D. Neuroinflammation: Modulation by flavonoids and mechanisms of action. Molecular aspects of medicine 33, 83–97, https://doi.org/10.1016/j.mam.2011.10.016 (2012).
Mehta, V., Parashar, A. & Udayabanu, M. Quercetin prevents chronic unpredictable stress induced behavioral dysfunction in mice by alleviating hippocampal oxidative and inflammatory stress. Physiol Behav 171, 69–78, https://doi.org/10.1016/j.physbeh.2017.01.006 (2017).
Simonyi, A. et al. Inhibition of microglial activation by elderberry extracts and its phenolic components. Life Sci 128, 30–38, https://doi.org/10.1016/j.lfs.2015.01.037 (2015).
Bournival, J., Plouffe, M., Renaud, J., Provencher, C. & Martinoli, M. G. Quercetin and Sesamin Protect Dopaminergic Cells from MPP +-Induced Neuroinflammation in a Microglial (N9)-Neuronal (PC12) Coculture System. Oxidative Medicine & Cellular Longevity 2012, 921941, https://doi.org/10.1155/2012/921941 (2012).
Kao, T. K. et al. Inhibition of nitric oxide production by quercetin in endotoxin/cytokine-stimulated microglia. Life Sci 86, 315–321, https://doi.org/10.1016/j.lfs.2009.12.014 (2010).
Saijo, K., Crotti, A. & Glass, C. K. Nuclear Receptors, Inflammation, and Neurodegenerative Diseases. Advances in Immunology, Vol 106 106, 21–59, https://doi.org/10.1016/S0065-2776(10)06002-5 (2010).
Sarray, S. et al. Serum IL-6, IL-10, and TNF alpha levels in pediatric sickle cell disease patients during vasoocclusive crisis and steady state condition. Cytokine 72, 43–47, https://doi.org/10.1016/j.cyto.2014.11.030 (2015).
Jing, X. et al. Diagnostic Potential of Differentially Expressed Homer1, IL-1 beta, and TNF-alpha in Coronary Artery Disease. Int J Mol Sci 16, 535–546, https://doi.org/10.3390/ijms16010535 (2015).
Gupta, V., Garg, R. K. & Khattri, S. Levels of IL-8 and TNF-alpha decrease in Parkinson’s disease. Neurological research 38, 98–102, https://doi.org/10.1080/01616412.2015.1133026 (2016).
O’Carroll, S. J. et al. Pro-inflammatory TNFalpha and IL-1beta differentially regulate the inflammatory phenotype of brain microvascular endothelial cells. J Neuroinflammation 12, 131, https://doi.org/10.1186/s12974-015-0346-0 (2015).
Yin, L. et al. Manganese exposure facilitates microglial JAK2-STAT3 signaling and consequent secretion of TNF-a and IL-1beta to promote neuronal death. Neurotoxicology, https://doi.org/10.1016/j.neuro.2017.04.001 (2017).
Gu, Y. et al. Role of TNF in mast cell neuroinflammation and pain. Journal of biological regulators and homeostatic agents 29, 787–791 (2015).
Rodgers, J., Bradley, B., Kennedy, P. G. & Sternberg, J. M. Central Nervous System Parasitosis and Neuroinflammation Ameliorated by Systemic IL-10 Administration in Trypanosoma brucei-Infected Mice. PLoS neglected tropical diseases 9, e0004201, https://doi.org/10.1371/journal.pntd.0004201 (2015).
Yu, A. et al. IL-17A promotes microglial activation and neuroinflammation in mouse models of intracerebral haemorrhage. Molecular immunology 73, 151–157, https://doi.org/10.1016/j.molimm.2016.04.003 (2016).
Latta, C. H. et al. Determining the role of IL-4 induced neuroinflammation in microglial activity and amyloid-beta using BV2 microglial cells and APP/PS1 transgenic mice. J Neuroinflamm 12, https://doi.org/10.1186/s12974-015-0243-6 (2015).
Bhuiyan, M. N., Mitsuhashi, S., Sigetomi, K. & Ubukata, M. Quercetin inhibits advanced glycation end product formation via chelating metal ions, trapping methylglyoxal, and trapping reactive oxygen species. Bioscience, biotechnology, and biochemistry 81, 882–890, https://doi.org/10.1080/09168451.2017.1282805 (2017).
Nam, J. S. et al. Application of Bioactive Quercetin in Oncotherapy: From Nutrition to Nanomedicine. Molecules 21, E108, https://doi.org/10.3390/molecules21010108 (2016).
Li, X. et al. Quercetin Protects Mice from ConA-Induced Hepatitis by Inhibiting HMGB1-TLR Expression and Down-Regulating the Nuclear Factor Kappa B Pathway. Inflammation 39, 96–106, https://doi.org/10.1007/s10753-015-0227-9 (2016).
Waseem, M. & Parvez, S. Neuroprotective activities of curcumin and quercetin with potential relevance to mitochondrial dysfunction induced by oxaliplatin. Protoplasma 253, 417–430, https://doi.org/10.1007/s00709-015-0821-6 (2016).
Liu, C. M. et al. Quercetin protects mouse liver against nickel-induced DNA methylation and inflammation associated with the Nrf2/HO-1 and p38/STAT1/NF-kappaB pathway. Food and chemical toxicology: an international journal published for the British Industrial Biological Research Association 82, 19–26, https://doi.org/10.1016/j.fct.2015.05.001 (2015).
Mao, W., Schuler, M. A. & Berenbaum, M. R. Disruption of quercetin metabolism by fungicide affects energy production in honey bees (Apis mellifera). Proceedings of the National Academy of Sciences of the United States of America 114, 2538–2543, https://doi.org/10.1073/pnas.1614864114 (2017).
Gerin, F. et al. The Effects of Quercetin on Acute Lung Injury and Biomarkers of Inflammation and Oxidative Stress in the Rat Model of Sepsis. Inflammation 39, 700–705, https://doi.org/10.1007/s10753-015-0296-9 (2016).
Yang, J. et al. Quercetin Protects Obesity-Induced Hypothalamic Inflammation by Reducing Microglia-Mediated Inflammatory Responses via HO-1 Induction. Nutrients 9, https://doi.org/10.3390/nu9070650 (2017).
Ruiz-Miyazawa, K. W. et al. Quercetin inhibits gout arthritis in mice: induction of an opioid-dependent regulation of inflammasome. Inflammopharmacology https://doi.org/10.1007/s10787-017-0356-x (2017).
Galleggiante, V. et al. Dendritic Cells Modulate Iron Homeostasis and Inflammatory Abilities Following Quercetin Exposure. Current pharmaceutical design 23, 2139–2146, https://doi.org/10.2174/1381612823666170112125355 (2017).
Chen, H. et al. Quercetin ameliorates imiquimod-induced psoriasis-like skin inflammation in mice via the NF-kappaB pathway. International immunopharmacology 48, 110–117, https://doi.org/10.1016/j.intimp.2017.04.022 (2017).
Carson, M. J., Thrash, J. C. & Walter, B. The cellular response in neuroinflammation: The role of leukocytes, microglia and astrocytes in neuronal death and survival. Clinical neuroscience research 6, 237–245, https://doi.org/10.1016/j.cnr.2006.09.004 (2006).
Rajkowska, G. & Miguel-Hidalgo, J. J. Gliogenesis and glial pathology in depression. CNS & neurological disorders drug targets 6, 219–233 (2007).
Muller, N. & Ackenheil, M. Psychoneuroimmunology and the cytokine action in the CNS: implications for psychiatric disorders. Progress in neuro-psychopharmacology & biological psychiatry 22, 1–33 (1998).
Brites, D. & Fernandes, A. Neuroinflammation andDepression: Microglia Activation, Extracellular Microvesicles and microRNA Dysregulation. Frontiers in cellular neuroscience 9, 476, https://doi.org/10.3389/fncel.2015.00476 (2015).
Li, B. et al. Wingless-type mammary tumor virus integration site family, member 5A (Wnt5a) regulates human immunodeficiency virus type 1 (HIV-1) envelope glycoprotein 120 (gp120)-induced expression of pro-inflammatory cytokines via the Ca2+/calmodulin-dependent protein kinase II (CaMKII) and c-Jun N-terminal kinase (JNK) signaling pathways. J Biol Chem 288, 13610–13619, https://doi.org/10.1074/jbc.M112.381046 (2013).
Li, B. et al. WNT5A signaling contributes to Abeta-induced neuroinflammation and neurotoxicity. PloS one 6, e22920, https://doi.org/10.1371/journal.pone.0022920 (2011).
Bhatt, P. M. & Malgor, R. Wnt5a: a player in the pathogenesis of atherosclerosis and other inflammatory disorders. Atherosclerosis 237, 155–162, https://doi.org/10.1016/j.atherosclerosis.2014.08.027 (2014).
Kumar, B. et al. Retinal neuroprotective effects of quercetin in streptozotocin-induced diabetic rats. Exp Eye Res 125, 193–202, https://doi.org/10.1016/j.exer.2014.06.009 (2014).
Jung, S. H., Murphy, E. A., McClellan, J. L., Carmichael, M. D. & Davis, J. M. The dietary flavonoid quercetin decreases neuroinflammation in a mouse model of Alzheimer’s disease. Faseb Journal 24 (2010).
Zheng, W. et al. IL-10 mediated by herpes simplex virus vector reduces neuropathic pain induced by HIV gp120 combined with ddC in rats. Molecular pain 10, 49, https://doi.org/10.1186/1744-8069-10-49 (2014).
Pany, S., Pal, A. & Sahu, K. Neuroprotective effect of quercetin in neurotoxicity induced rats: Role of neuroinflammation in neurodegeneration. Asian Journal of Pharmaceutical & Clinical Research 7 (2014).
Bahar, E., Kim, J. Y. & Yoon, H. Quercetin Attenuates Manganese-Induced Neuroinflammation by Alleviating Oxidative Stress through Regulation of Apoptosis, iNOS/NF-κB and HO-1/Nrf2 Pathways. Int J Mol Sci 18, e2841 (2017).
Gudrun, P. et al. Impact of Quercetin and EGCG on Key Elements of the Wnt Pathway in Human Colon Carcinoma Cells. Journal of Agricultural & Food Chemistry 54, 7075 (2006).
Kim, H. et al. Regulation of Wnt signaling activity for growth suppression induced by quercetin in 4T1 murine mammary cancer cells. International Journal of Oncology 43, 1319–1325 (2013).
Novo, M. C. T. et al. Blockage of Wnt/β-catenin signaling by quercetin reduces survival and proliferation of B-1 cells in vitro. Immunobiology 220, 60 (2015).
Hong, H., Kim, B. S. & Im, H. I. Pathophysiological Role of Neuroinflammation in Neurodegenerative Diseases and Psychiatric Disorders. Int Neurourol J 20, S2–S7, https://doi.org/10.5213/inj.1632604.302 (2016).
Ransohoff, R. M. How neuroinflammation contributes to neurodegeneration. Science 353, 777–783, https://doi.org/10.1126/science.aag2590 (2016).
Gao, H. M. et al. HMGB1 acts on microglia Mac1 to mediate chronic neuroinflammation that drives progressive neurodegeneration. The Journal of neuroscience: the official journal of the Society for Neuroscience 31, 1081–1092, https://doi.org/10.1523/JNEUROSCI.3732-10.2011 (2011).
Qin, L. et al. Systemic LPS causes chronic neuroinflammation and progressive neurodegeneration. Glia 55, 453–462, https://doi.org/10.1002/glia.20467 (2007).
Maixner, D. W. & Weng, H. R. The Role of Glycogen Synthase Kinase 3 Beta in Neuroinflammation and Pain. Journal of pharmaceutics & pharmacology 1, 001, https://doi.org/10.13188/2327-204X.1000001 (2013).
Zhou, Y. Q. et al. Interleukin-6: an emerging regulator of pathological pain. J Neuroinflamm 13, https://doi.org/10.1186/S12974-016-0607-6 (2016).
Luo, X., Wang, X. M., Xia, Z. Y., Chung, S. K. & Cheung, C. W. CXCL12/CXCR4 axis: an emerging neuromodulator in pathological pain. Rev Neuroscience 27, 83–92, https://doi.org/10.1515/revneuro-2015-0016 (2016).
Jha, M. K., Jeon, S., Jin, M., Lee, W. H. & Suk, K. Acute Phase Protein Lipocalin-2 Is Associated with Formalin-induced Nociception and PathologicalPain. Immune network 13, 289–294, https://doi.org/10.4110/in.2013.13.6.289 (2013).
Zheng, X. et al. TNF alpha is involved in neuropathic pain induced by nucleoside reverse transcriptase inhibitor in rats. Brain Behavior and Immunity 25, 1668–1676, https://doi.org/10.1016/j.bbi.2011.06.010 (2011).
Eder Marcolin, B. S.-M. et al.. Quercetin treatment ameliorates inflammation and fibrosis in mice with nonalcoholic steatohepatitis. Journal of Nutrition 142, 1821–1828.
Tang, D. et al. Quercetin prevents LPS-induced high-mobility group box 1 release and proinflammatory function. American Journal of Respiratory Cell & Molecular Biology 41, 651–660 (2009).
Xi, L. et al. Quercetin Protects Mice from ConA-Induced Hepatitis by Inhibiting HMGB1-TLR Expression and Down-Regulating the Nuclear Factor Kappa B Pathway. Inflammation 39, 96–106 (2016).
Acknowledgements
This work was supported by the Zhejiang Provincial Natural Science Foundation (No. LY13H090015) to J.S. and NIH grants (R01NS079166, R01NS095747 and R01DA036165) to S.J.T.
Author information
Authors and Affiliations
Contributions
J.H.S., W.P.Z., Y.L.H. and S.J.T. conceived and designed the study. Y.Y., T.W. and X.K.L. performed the experiments. Y.Y. and S.J.T. discussed and analyzed the results. Y.Y. and S.J.T. wrote the manuscript.
Corresponding authors
Ethics declarations
Competing Interests
The authors declare no competing interests.
Additional information
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Yang, Y., Liu, X., Wu, T. et al. Quercetin attenuates AZT-induced neuroinflammation in the CNS. Sci Rep 8, 6194 (2018). https://doi.org/10.1038/s41598-018-24618-2
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-018-24618-2
This article is cited by
-
Progress in Pathological and Therapeutic Research of HIV-Related Neuropathic Pain
Cellular and Molecular Neurobiology (2023)
-
Withania somnifera (L.) Dunal ameliorates neurodegeneration and cognitive impairments associated with systemic inflammation
BMC Complementary and Alternative Medicine (2019)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.