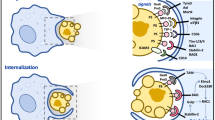
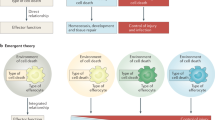
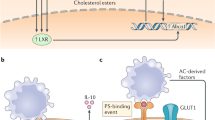
Abstract
Multiple modes of cell death have been identified, each with a unique function and each induced in a setting-dependent manner. As billions of cells die during mammalian embryogenesis and daily in adult organisms, clearing dead cells and associated cellular debris is important in physiology. In this Review, we present an overview of the phagocytosis of dead and dying cells, a process known as efferocytosis. Efferocytosis is performed by macrophages and to a lesser extent by other ‘professional’ phagocytes (such as monocytes and dendritic cells) and ‘non-professional’ phagocytes, such as epithelial cells. Recent discoveries have shed light on this process and how it functions to maintain tissue homeostasis, tissue repair and organismal health. Here, we outline the mechanisms of efferocytosis, from the recognition of dying cells through to phagocytic engulfment and homeostatic resolution, and highlight the pathophysiological consequences that can arise when this process is abrogated.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$189.00 per year
only $15.75 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Galluzzi, L. et al. Molecular mechanisms of cell death: recommendations of the Nomenclature Committee on Cell Death 2018. Cell Death Differ. 25, 486–541 (2018). A useful set of definitions of diverse modes of cell death based on mechanisms rather than morphology.
Bergmann, A. & Steller, H. Apoptosis, stem cells, and tissue regeneration. Sci. Signal. 3, re8 (2010).
Suzanne, M. & Steller, H. Shaping organisms with apoptosis. Cell Death Differ. 20, 669–675 (2013).
Fuchs, Y. & Steller, H. Programmed cell death in animal development and disease. Cell 147, 742–758 (2011).
Blander, J. M. Death in the intestinal epithelium-basic biology and implications for inflammatory bowel disease. FEBS J. 283, 2720–2730 (2016).
Cummings, R. J. et al. Different tissue phagocytes sample apoptotic cells to direct distinct homeostasis programs. Nature 539, 565–569 (2016). A demonstration of the role of clearance of dead intestinal epithelial cells by intestinal macrophages and dendritic cells in controlling inflammation of the tissue.
Eckhart, L., Lippens, S., Tschachler, E. & Declercq, W. Cell death by cornification. Biochim. Biophys. Acta 1833, 3471–3480 (2013).
Florey, O., Krajcovic, M., Sun, Q. & Overholtzer, M. Entosis. Curr. Biol. 20, R88–R89 (2010).
Overholtzer, M. et al. A nonapoptotic cell death process, entosis, that occurs by cell-in-cell invasion. Cell 131, 966–979 (2007).
Segawa, K. et al. Phospholipid flippases enable precursor B cells to flee engulfment by macrophages. Proc. Natl Acad. Sci. USA 115, 12212–12217 (2018). Genetic ablation of the phospholipid flippase ATP11C results in loss of developing B cells due to their engulfment.
Metayer, L. E., Vilalta, A., Burke, G. A. A. & Brown, G. C. Anti-CD47 antibodies induce phagocytosis of live, malignant B cells by macrophages via the Fc domain, resulting in cell death by phagoptosis. Oncotarget 8, 60892–60903 (2017).
Kerr, J. F., Wyllie, A. H. & Currie, A. R. Apoptosis: a basic biological phenomenon with wide-ranging implications in tissue kinetics. Br. J. Cancer 26, 239–257 (1972).
Majno, G. & Joris, I. Apoptosis, oncosis, and necrosis. An overview of cell death. Am. J. Pathol. 146, 3–15 (1995).
Wyllie, A. H., Kerr, J. F. & Currie, A. R. Cell death: the significance of apoptosis. Int. Rev. Cytol. 68, 251–306 (1980).
Lauber, K. et al. Apoptotic cells induce migration of phagocytes via caspase-3-mediated release of a lipid attraction signal. Cell 113, 717–730 (2003).
Elliott, M. R. et al. Nucleotides released by apoptotic cells act as a find-me signal to promote phagocytic clearance. Nature 461, 282–286 (2009).
Truman, L. A. et al. CX3CL1/fractalkine is released from apoptotic lymphocytes to stimulate macrophage chemotaxis. Blood 112, 5026–5036 (2008).
Elliott, M. R., Koster, K. M. & Murphy, P. S. Efferocytosis signaling in the regulation of macrophage inflammatory responses. J. Immunol. 198, 1387–1394 (2017).
Gude, D. R. et al. Apoptosis induces expression of sphingosine kinase 1 to release sphingosine-1-phosphate as a “come-and-get-me” signal. FASEB J. 22, 2629–2638 (2008).
Weigert, A. et al. Apoptotic cells promote macrophage survival by releasing the antiapoptotic mediator sphingosine-1-phosphate. Blood 108, 1635–1642 (2006).
Peter, C. et al. Release of lysophospholipid ‘find-me’ signals during apoptosis requires the ATP-binding cassette transporter A1. Autoimmunity 45, 568–573 (2012).
Luo, B. et al. Erythropoeitin signaling in macrophages promotes dying cell clearance and immune tolerance. Immunity 44, 287–302 (2016).
Kumar, H., Kawai, T. & Akira, S. Pathogen recognition by the innate immune system. Int. Rev. Immunol. 30, 16–34 (2011).
Akira, S., Uematsu, S. & Takeuchi, O. Pathogen recognition and innate immunity. Cell 124, 783–801 (2006).
Scaffidi, P., Misteli, T. & Bianchi, M. E. Release of chromatin protein HMGB1 by necrotic cells triggers inflammation. Nature 418, 191–195 (2002).
Shi, Y., Evans, J. E. & Rock, K. L. Molecular identification of a danger signal that alerts the immune system to dying cells. Nature 425, 516–521 (2003).
Shi, J., Gao, W. & Shao, F. Pyroptosis: gasdermin-mediated programmed necrotic cell death. Trends Biochem. Sci. 42, 245–254 (2017).
Liu, X., Zou, H., Slaughter, C. & Wang, X. DFF, a heterodimeric protein that functions downstream of caspase-3 to trigger DNA fragmentation during apoptosis. Cell 89, 175–184 (1997).
Enari, M. et al. A caspase-activated DNase that degrades DNA during apoptosis, and its inhibitor ICAD. Nature 391, 43–50 (1998).
Sakahira, H., Enari, M. & Nagata, S. Cleavage of CAD inhibitor in CAD activation and DNA degradation during apoptosis. Nature 391, 96–99 (1998).
Arandjelovic, S. & Ravichandran, K. S. Phagocytosis of apoptotic cells in homeostasis. Nat. Immunol. 16, 907–917 (2015).
Zhang, Q. et al. Circulating mitochondrial DAMPs cause inflammatory responses to injury. Nature 464, 104–107 (2010).
Hawes, M. C., Wen, F. & Elquza, E. Extracellular DNA: a bridge to cancer. Cancer Res. 75, 4260–4264 (2015).
Yang, Y. G., Lindahl, T. & Barnes, D. E. Trex1 exonuclease degrades ssDNA to prevent chronic checkpoint activation and autoimmune disease. Cell 131, 873–886 (2007).
Paludan, S. R. & Bowie, A. G. Immune sensing of DNA. Immunity 38, 870–880 (2013).
Motwani, M., Pesiridis, S. & Fitzgerald, K. A. DNA sensing by the cGAS-STING pathway in health and disease. Nat. Rev. Genet. 20, 657–674 (2019).
Tanaka, Y. & Chen, Z. J. STING specifies IRF3 phosphorylation by TBK1 in the cytosolic DNA signaling pathway. Sci. Signal. 5, ra20 (2012).
Kawane, K. et al. Requirement of DNase II for definitive erythropoiesis in the mouse fetal liver. Science 292, 1546–1549 (2001).
Gao, D. et al. Activation of cyclic GMP-AMP synthase by self-DNA causes autoimmune diseases. Proc. Natl Acad. Sci. USA 112, E5699–E5705 (2015).
Baum, R. et al. STING contributes to abnormal bone formation induced by deficiency of DNase II in mice. Arthritis Rheumatol. 69, 460–471 (2017).
Ahn, J., Gutman, D., Saijo, S. & Barber, G. N. STING manifests self DNA-dependent inflammatory disease. Proc. Natl Acad. Sci. USA 109, 19386–19391 (2012).
Cunha, L. D. et al. AIM2 ngages Acative but unprocessed caspase-1 to induce noncanonical activation of the NLRP3 inflammasome. Cell Rep. 20, 794–805 (2017).
Lugrin, J. & Martinon, F. The AIM2 inflammasome: sensor of pathogens and cellular perturbations. Immunol. Rev. 281, 99–114 (2018).
Stros, M. HMGB proteins: interactions with DNA and chromatin. Biochim. Biophys. Acta 1799, 101–113 (2010).
Yang, H., Wang, H., Chavan, S. S. & Andersson, U. High mobility group box protein 1 (HMGB1): the prototypical endogenous danger molecule. Mol. Med. 21, S6–S12 (2015).
Bertheloot, D. & Latz, E. HMGB1, IL-1alpha, IL-33 and S100 proteins: dual-function alarmins. Cell Mol. Immunol. 14, 43–64 (2017).
Bonaldi, T. et al. Monocytic cells hyperacetylate chromatin protein HMGB1 to redirect it towards secretion. EMBO J. 22, 5551–5560 (2003).
Kazama, H. et al. Induction of immunological tolerance by apoptotic cells requires caspase-dependent oxidation of high-mobility group box-1 protein. Immunity 29, 21–32 (2008).
Venereau, E. et al. Mutually exclusive redox forms of HMGB1 promote cell recruitment or proinflammatory cytokine release. J. Exp. Med. 209, 1519–1528 (2012).
Wen, Q., Liu, J., Kang, R., Zhou, B. & Tang, D. The release and activity of HMGB1 in ferroptosis. Biochem. Biophys. Res. Commun. 510, 278–283 (2019).
Tirone, M. et al. High mobility group box 1 orchestrates tissue regeneration via CXCR4. J. Exp. Med. 215, 303–318 (2018). HMGB1, released on tissue injury, promotes the repair of liver and muscle via interaction with the chemokine receptor CXCR4.
Rock, K. L. & Kono, H. The inflammatory response to cell death. Annu. Rev. Pathol. 3, 99–126 (2008).
Chen, C. J. et al. Identification of a key pathway required for the sterile inflammatory response triggered by dying cells. Nat. Med. 13, 851–856 (2007).
Eigenbrod, T., Park, J. H., Harder, J., Iwakura, Y. & Nunez, G. Cutting edge: critical role for mesothelial cells in necrosis-induced inflammation through the recognition of IL-1 alpha released from dying cells. J. Immunol. 181, 8194–8198 (2008).
Martin, S. J. Cell death and inflammation: the case for IL-1 family cytokines as the canonical DAMPs of the immune system. FEBS J. 283, 2599–2615 (2016). In this useful commentary, the idea that IL-1 family cytokines function as the ‘true’ DAMPs, released on cell death, is convincingly argued.
Idzko, M., Ferrari, D. & Eltzschig, H. K. Nucleotide signalling during inflammation. Nature 509, 310–317 (2014).
Chiu, Y. H. et al. A quantized mechanism for activation of pannexin channels. Nat. Commun. 8, 14324 (2017).
Chekeni, F. B. et al. Pannexin 1 channels mediate ‘find-me’ signal release and membrane permeability during apoptosis. Nature 467, 863–867 (2010).
Qu, Y. et al. Pannexin-1 is required for ATP release during apoptosis but not for inflammasome activation. J. Immunol. 186, 6553–6561 (2011).
Wang, Q. et al. Pyroptotic cells externalize eat-me and release find-me signals and are efficiently engulfed by macrophages. Int. Immunol. 25, 363–372 (2013).
Yang, D., He, Y., Munoz-Planillo, R., Liu, Q. & Nunez, G. Caspase-11 requires the pannexin-1 channel and the purinergic P2X7 pore to mediate pyroptosis and endotoxic shock. Immunity 43, 923–932 (2015).
Chen, J., Zhao, Y. & Liu, Y. The role of nucleotides and purinergic signaling in apoptotic cell clearance - implications for chronic inflammatory diseases. Front. Immunol. 5, 656 (2014).
Segawa, K. & Nagata, S. An apoptotic ‘eat me’ signal: phosphatidylserine exposure. Trends Cell Biol. 25, 639–650 (2015).
Suzuki, J., Denning, D. P., Imanishi, E., Horvitz, H. R. & Nagata, S. Xk-related protein 8 and CED-8 promote phosphatidylserine exposure in apoptotic cells. Science 341, 403–406 (2013).
Kloditz, K. & Fadeel, B. Three cell deaths and a funeral: macrophage clearance of cells undergoing distinct modes of cell death. Cell Death Discov. 5, 65 (2019).
Zargarian, S. et al. Phosphatidylserine externalization, “necroptotic bodies” release, and phagocytosis during necroptosis. PLoS Biol. 15, e2002711 (2017).
Ousingsawat, J., Schreiber, R. & Kunzelmann, K. TMEM16F/anoctamin 6 in ferroptotic cell death. Cancers 11, 625 (2019).
Fujii, T., Sakata, A., Nishimura, S., Eto, K. & Nagata, S. TMEM16F is required for phosphatidylserine exposure and microparticle release in activated mouse platelets. Proc. Natl Acad. Sci. USA 112, 12800–12805 (2015).
Suzuki, J., Umeda, M., Sims, P. J. & Nagata, S. Calcium-dependent phospholipid scrambling by TMEM16F. Nature 468, 834–838 (2010).
Hochreiter-Hufford, A. & Ravichandran, K. S. Clearing the dead: apoptotic cell sensing, recognition, engulfment, and digestion. Cold Spring Harb. Perspect. Biol. 5, a008748 (2013).
Wu, Y., Tibrewal, N. & Birge, R. B. Phosphatidylserine recognition by phagocytes: a view to a kill. Trends Cell Biol. 16, 189–197 (2006).
Geng, K. et al. Requirement of gamma-carboxyglutamic acid modification and phosphatidylserine binding for the activation of tyro3, Axl, and Mertk receptors by growth arrest-specific 6. Front. Immunol. 8, 1521 (2017).
Lee, H. N. et al. Dendritic cells expressing immunoreceptor CD300f are critical for controlling chronic gut inflammation. J. Clin. Invest. 127, 1905–1917 (2017).
Parks, B. W. et al. CD36, but not G2A, modulates efferocytosis, inflammation, and fibrosis following bleomycin-induced lung injury. J. Lipid Res. 54, 1114–1123 (2013).
Tait, J. F. & Smith, C. Phosphatidylserine receptors: role of CD36 in binding of anionic phospholipid vesicles to monocytic cells. J. Biol. Chem. 274, 3048–3054 (1999).
Szondy, Z., Sarang, Z., Kiss, B., Garabuczi, E. & Koroskenyi, K. Anti-inflammatory mechanisms triggered by apoptotic cells during their clearance. Front. Immunol. 8, 909 (2017).
Xu, W. et al. IL-10-producing macrophages preferentially clear early apoptotic cells. Blood 107, 4930–4937 (2006).
Fadok, V. A. et al. Macrophages that have ingested apoptotic cells in vitro inhibit proinflammatory cytokine production through autocrine/paracrine mechanisms involving TGF-beta, PGE2, and PAF. J. Clin. Invest. 101, 890–898 (1998).
Freire-de-Lima, C. G. et al. Apoptotic cells, through transforming growth factor-beta, coordinately induce anti-inflammatory and suppress pro-inflammatory eicosanoid and NO synthesis in murine macrophages. J. Biol. Chem. 281, 38376–38384 (2006).
Voll, R. E. et al. Immunosuppressive effects of apoptotic cells. Nature 390, 350–351 (1997).
Kim, S. J., Gershov, D., Ma, X., Brot, N. & Elkon, K. B. I-PLA(2) activation during apoptosis promotes the exposure of membrane lysophosphatidylcholine leading to binding by natural immunoglobulin M antibodies and complement activation. J. Exp. Med. 196, 655–665 (2002).
Gardai, S. J. et al. Cell-surface calreticulin initiates clearance of viable or apoptotic cells through trans-activation of LRP on the phagocyte. Cell 123, 321–334 (2005).
Ogden, C. A. et al. C1q and mannose binding lectin engagement of cell surface calreticulin and CD91 initiates macropinocytosis and uptake of apoptotic cells. J. Exp. Med. 194, 781–795 (2001).
Oldenborg, P. A. et al. Role of CD47 as a marker of self on red blood cells. Science 288, 2051–2054 (2000).
Barkal, A. A. et al. CD24 signalling through macrophage Siglec-10 is a target for cancer immunotherapy. Nature 572, 392–396 (2019). Identification of the CD24–SIGLEC10 interaction as a ‘don’t-eat-me’ signal and its possible value in cancer therapy.
Brown, S. et al. Apoptosis disables CD31-mediated cell detachment from phagocytes promoting binding and engulfment. Nature 418, 200–203 (2002).
Abram, C. L. & Lowell, C. A. Shp1 function in myeloid cells. J. Leukoc. Biol. 102, 657–675 (2017).
Barkal, A. A. et al. Engagement of MHC class I by the inhibitory receptor LILRB1 suppresses macrophages and is a target of cancer immunotherapy. Nat. Immunol. 19, 76–84 (2018). Identification of the MHC class I–LILRB1 interaction as a ‘don’t-eat-me’ signal and its possible value in cancer therapy.
Willingham, S. B. et al. The CD47-signal regulatory protein alpha (SIRPa) interaction is a therapeutic target for human solid tumors. Proc. Natl Acad. Sci. USA 109, 6662–6667 (2012).
Werfel, T. A. & Cook, R. S. Efferocytosis in the tumor microenvironment. Semin. Immunopathol. 40, 545–554 (2018).
Weiskopf, K. et al. Engineered SIRPalpha variants as immunotherapeutic adjuvants to anticancer antibodies. Science 341, 88–91 (2013).
Chao, M. P. et al. Calreticulin is the dominant pro-phagocytic signal on multiple human cancers and is counterbalanced by CD47. Sci. Transl Med. 2, 63ra94 (2010).
Azuma, Y., Nakagawa, H., Dote, K., Higai, K. & Matsumoto, K. Decreases in CD31 and CD47 levels on the cell surface during etoposide-induced Jurkat cell apoptosis. Biol. Pharm. Bull. 34, 1828–1834 (2011).
Richards, D. M. & Endres, R. G. The mechanism of phagocytosis: two stages of engulfment. Biophys. J. 107, 1542–1553 (2014).
Rosales, C. & Uribe-Querol, E. Phagocytosis: a fundamental process in immunity. Biomed. Res. Int. 2017, 9042851 (2017).
Ma, Z. et al. Regulation of Rac1 activation by the low density lipoprotein receptor-related protein. J. Cell Biol. 159, 1061–1070 (2002).
Park, D. et al. BAI1 is an engulfment receptor for apoptotic cells upstream of the ELMO/Dock180/Rac module. Nature 450, 430–434 (2007).
Green, D. R., Oguin, T. H. & Martinez, J. The clearance of dying cells: table for two. Cell Death Differ. 23, 915–926 (2016).
Elliott, M. R. & Ravichandran, K. S. The dynamics of apoptotic cell clearance. Dev. Cell 38, 147–160 (2016).
Sevajol, M. et al. The C-terminal polyproline-containing region of ELMO contributes to an increase in the life-time of the ELMO-DOCK complex. Biochimie 94, 823–828 (2012).
Waterborg, C. E. J. et al. Protective role of the MER tyrosine kinase via efferocytosis in rheumatoid arthritis models. Front. Immunol. 9, 742 (2018).
Nienhuis, H. L. et al. AGE and their receptor RAGE in systemic autoimmune diseases: an inflammation propagating factor contributing to accelerated atherosclerosis. Autoimmunity 42, 302–304 (2009).
Marie-Anais, F., Mazzolini, J., Herit, F. & Niedergang, F. Dynamin-actin cross talk contributes to phagosome formation and closure. Traffic 17, 487–499 (2016).
Stenmark, H. Rab GTPases as coordinators of vesicle traffic. Nat. Rev. Mol. Cell Biol. 10, 513–525 (2009).
Rink, J., Ghigo, E., Kalaidzidis, Y. & Zerial, M. Rab conversion as a mechanism of progression from early to late endosomes. Cell 122, 735–749 (2005).
Chavrier, P., Parton, R. G., Hauri, H. P., Simons, K. & Zerial, M. Localization of low molecular weight GTP binding proteins to exocytic and endocytic compartments. Cell 62, 317–329 (1990).
Vieira, O. V. et al. Modulation of Rab5 and Rab7 recruitment to phagosomes by phosphatidylinositol 3-kinase. Mol. Cell Biol. 23, 2501–2514 (2003).
Flannagan, R. S., Jaumouille, V. & Grinstein, S. The cell biology of phagocytosis. Annu. Rev. Pathol. 7, 61–98 (2012).
Rubino, M., Miaczynska, M., Lippe, R. & Zerial, M. Selective membrane recruitment of EEA1 suggests a role in directional transport of clathrin-coated vesicles to early endosomes. J. Biol. Chem. 275, 3745–3748 (2000).
Vieira, O. V. et al. Distinct roles of class I and class III phosphatidylinositol 3-kinases in phagosome formation and maturation. J. Cell Biol. 155, 19–25 (2001).
Murray, J. T., Panaretou, C., Stenmark, H., Miaczynska, M. & Backer, J. M. Role of Rab5 in the recruitment of hVps34/p150 to the early endosome. Traffic 3, 416–427 (2002).
Kissing, S. et al. Vacuolar ATPase in phagosome-lysosome fusion. J. Biol. Chem. 290, 14166–14180 (2015).
Lukacs, G. L., Rotstein, O. D. & Grinstein, S. Determinants of the phagosomal pH in macrophages. In situ assessment of vacuolar H+-ATPase activity, counterion conductance, and H+ “leak”. J. Biol. Chem. 266, 24540–24548 (1991).
Harrison, R. E., Bucci, C., Vieira, O. V., Schroer, T. A. & Grinstein, S. Phagosomes fuse with late endosomes and/or lysosomes by extension of membrane protrusions along microtubules: role of Rab7 and RILP. Mol. Cell Biol. 23, 6494–6506 (2003).
Huynh, K. K. et al. LAMP proteins are required for fusion of lysosomes with phagosomes. EMBO J. 26, 313–324 (2007).
Braun, V. et al. TI-VAMP/VAMP7 is required for optimal phagocytosis of opsonised particles in macrophages. EMBO J. 23, 4166–4176 (2004).
Fairn, G. D. & Grinstein, S. How nascent phagosomes mature to become phagolysosomes. Trends Immunol. 33, 397–405 (2012).
Collins, R. F., Schreiber, A. D., Grinstein, S. & Trimble, W. S. Syntaxins 13 and 7 function at distinct steps during phagocytosis. J. Immunol. 169, 3250–3256 (2002).
Xu, H. & Ren, D. Lysosomal physiology. Annu. Rev. Physiol. 77, 57–80 (2015).
Martinez, J. et al. Microtubule-associated protein 1 light chain 3 alpha (LC3)-associated phagocytosis is required for the efficient clearance of dead cells. Proc. Natl Acad. Sci. USA 108, 17396–17401 (2011).
Martinez, J. et al. Molecular characterization of LC3-associated phagocytosis reveals distinct roles for rubicon, NOX2 and autophagy proteins. Nat. Cell Biol. 17, 893–906 (2015).
Wong, S. W., Sil, P. & Martinez, J. Rubicon: LC3-associated phagocytosis and beyond. FEBS J. 285, 1379–1388 (2018).
Heckmann, B. L., Boada-Romero, E., Cunha, L. D., Magne, J. & Green, D. R. LC3-associated phagocytosis and inflammation. J. Mol. Biol. 429, 3561–3576 (2017).
Heckmann, B. L. & Green, D. R. LC3-associated phagocytosis at a glance. J. Cell Sci. 132, 222984 (2019).
Martinez, J. et al. Noncanonical autophagy inhibits the autoinflammatory, lupus-like response to dying cells. Nature 533, 115–119 (2016). A demonstration that genetic ablation of LAP in myeloid cells promotes spontaneous lupus-like disease.
Heckmann, B. L. et al. LC3-associated endocytosis facilitates beta-amyloid clearance and mitigates neurodegeneration in murine Alzheimer’s disease. Cell, 536–551.e14 (2019).
Nakamura, S. & Yoshimori, T. New insights into autophagosome-lysosome fusion. J. Cell Sci. 130, 1209–1216 (2017).
Pankiv, S. et al. FYCO1 is a Rab7 effector that binds to LC3 and PI3P to mediate microtubule plus end-directed vesicle transport. J. Cell Biol. 188, 253–269 (2010).
McEwan, D. G. et al. PLEKHM1 regulates autophagosome-lysosome fusion through HOPS complex and LC3/GABARAP proteins. Mol. Cell 57, 39–54 (2015).
Tabata, K. et al. Rubicon and PLEKHM1 negatively regulate the endocytic/autophagic pathway via a novel Rab7-binding domain. Mol. Biol. Cell 21, 4162–4172 (2010).
Heckmann, B. L. et al. Liver X receptor alpha mediates hepatic triglyceride accumulation through upregulation of G0/G1 switch gene 2 expression. JCI Insight 2, e88735 (2017).
Kidani, Y. & Bensinger, S. J. Liver X receptor and peroxisome proliferator-activated receptor as integrators of lipid homeostasis and immunity. Immunol. Rev. 249, 72–83 (2012).
Schulman, I. G. Liver X receptors link lipid metabolism and inflammation. FEBS Lett. 591, 2978–2991 (2017).
Zhang, X., Heckmann, B. L., Campbell, L. E. & Liu, J. G0S2: a small giant controller of lipolysis and adipose-liver fatty acid flux. Biochim. Biophys. Acta Mol. Cell Biol. Lipids 1862, 1146–1154 (2017).
N, A. G. et al. Apoptotic cells promote their own clearance and immune tolerance through activation of the nuclear receptor LXR. Immunity 31, 245–258 (2009).
Rebe, C. et al. Induction of transglutaminase 2 by a liver X receptor/retinoic acid receptor alpha pathway increases the clearance of apoptotic cells by human macrophages. Circ. Res. 105, 393–401 (2009).
Fond, A. M., Lee, C. S., Schulman, I. G., Kiss, R. S. & Ravichandran, K. S. Apoptotic cells trigger a membrane-initiated pathway to increase ABCA1. J. Clin. Invest. 125, 2748–2758 (2015).
Kiss, R. S., Elliott, M. R., Ma, Z., Marcel, Y. L. & Ravichandran, K. S. Apoptotic cells induce a phosphatidylserine-dependent homeostatic response from phagocytes. Curr. Biol. 16, 2252–2258 (2006).
Yoon, Y. S. et al. PPARgamma activation following apoptotic cell instillation promotes resolution of lung inflammation and fibrosis via regulation of efferocytosis and proresolving cytokines. Mucosal Immunol. 8, 1031–1046 (2015).
Cunha, L. D. et al. LC3-Associated phagocytosis in myeloid cells promotes tumor immune tolerance. Cell 175, 429–441.e16 (2018). Genetic ablation of LAP in myeloid cells of the tumour microenvironment promotes anticancer immunity in several model systems by the engagement of a type I interferon response.
Yokoyama, K. et al. Rab27a negatively regulates phagocytosis by prolongation of the actin-coating stage around phagosomes. J. Biol. Chem. 286, 5375–5382 (2011).
Kuiper, J. W. et al. Creatine kinase-mediated ATP supply fuels actin-based events in phagocytosis. PLoS Biol. 6, e51 (2008).
Lucin, K. M. et al. Microglial beclin 1 regulates retromer trafficking and phagocytosis and is impaired in Alzheimer’s disease. Neuron 79, 873–886 (2013).
Baumann, I. et al. Impaired uptake of apoptotic cells into tingible body macrophages in germinal centers of patients with systemic lupus erythematosus. Arthritis Rheum. 46, 191–201 (2002).
Liu, Z. & Davidson, A. Taming lupus-a new understanding of pathogenesis is leading to clinical advances. Nat. Med. 18, 871–882 (2012).
Berden, J. H. Lupus nephritis. Kidney Int. 52, 538–558 (1997).
van Bruggen, M. C. et al. Antigen specificity of anti-nuclear antibodies complexed to nucleosomes determines glomerular basement membrane binding in vivo. Eur. J. Immunol. 27, 1564–1569 (1997).
Witting, A., Muller, P., Herrmann, A., Kettenmann, H. & Nolte, C. Phagocytic clearance of apoptotic neurons by Microglia/Brain macrophages in vitro: involvement of lectin-, integrin-, and phosphatidylserine-mediated recognition. J. Neurochem. 75, 1060–1070 (2000).
Galloway, D. A., Phillips, A. E. M., Owen, D. R. J. & Moore, C. S. Phagocytosis in the brain: homeostasis and disease. Front. Immunol. 10, 790 (2019).
Mattson, M. P. Apoptosis in neurodegenerative disorders. Nat. Rev. Mol. Cell Biol. 1, 120–129 (2000).
Heckmann, B. L., Tummers, B. & Green, D. R. Crashing the computer: apoptosis vs. necroptosis in neuroinflammation. Cell Death Differ. 26, 41–52 (2019).
Fricker, M. et al. MFG-E8 mediates primary phagocytosis of viable neurons during neuroinflammation. J. Neurosci. 32, 2657–2666 (2012).
Fuller, A. D. & Van Eldik, L. J. MFG-E8 regulates microglial phagocytosis of apoptotic neurons. J. Neuroimmune Pharmacol. 3, 246–256 (2008).
Ferguson, T. A. & Green, D. R. Autophagy and phagocytosis converge for better vision. Autophagy 10, 165–167 (2014).
Kim, J. Y. et al. Noncanonical autophagy promotes the visual cycle. Cell 154, 365–376 (2013).
Nandrot, E. F. & Dufour, E. M. Mertk in daily retinal phagocytosis: a history in the making. Adv. Exp. Med. Biol. 664, 133–140 (2010).
Yang, H. et al. Activation of liver X receptor alleviates ocular inflammation in experimental autoimmune uveitis. Invest. Ophthalmol. Vis. Sci. 55, 2795–2804 (2014).
Fox, S., Leitch, A. E., Duffin, R., Haslett, C. & Rossi, A. G. Neutrophil apoptosis: relevance to the innate immune response and inflammatory disease. J. Innate Immun. 2, 216–227 (2010).
Juncadella, I. J. et al. Apoptotic cell clearance by bronchial epithelial cells critically influences airway inflammation. Nature 493, 547–551 (2013).
Hodge, S., Hodge, G., Scicchitano, R., Reynolds, P. N. & Holmes, M. Alveolar macrophages from subjects with chronic obstructive pulmonary disease are deficient in their ability to phagocytose apoptotic airway epithelial cells. Immunol. Cell Biol. 81, 289–296 (2003).
Vandivier, R. W. et al. Impaired clearance of apoptotic cells from cystic fibrosis airways. Chest 121, 89S (2002).
Ley, K., Miller, Y. I. & Hedrick, C. C. Monocyte and macrophage dynamics during atherogenesis. Arterioscler. Thromb. Vasc. Biol. 31, 1506–1516 (2011).
Tabas, I. Consequences and therapeutic implications of macrophage apoptosis in atherosclerosis: the importance of lesion stage and phagocytic efficiency. Arterioscler. Thromb. Vasc. Biol. 25, 2255–2264 (2005).
Poon, I. K., Lucas, C. D., Rossi, A. G. & Ravichandran, K. S. Apoptotic cell clearance: basic biology and therapeutic potential. Nat. Rev. Immunol. 14, 166–180 (2014).
Schrijvers, D. M., De Meyer, G. R., Kockx, M. M., Herman, A. G. & Martinet, W. Phagocytosis of apoptotic cells by macrophages is impaired in atherosclerosis. Arterioscler. Thromb. Vasc. Biol. 25, 1256–1261 (2005).
Vucic, E. et al. Regression of inflammation in atherosclerosis by the LXR agonist R211945: a noninvasive assessment and comparison with atorvastatin. JACC Cardiovasc. Imaging 5, 819–828 (2012).
Li, B. Z., Zhang, H. Y., Pan, H. F. & Ye, D. Q. Identification of MFG-E8 as a novel therapeutic target for diseases. Expert Opin. Ther. Targets 17, 1275–1285 (2013).
Cuchel, M. & Rader, D. J. Macrophage reverse cholesterol transport: key to the regression of atherosclerosis? Circulation 113, 2548–2555 (2006).
Oram, J. F. & Heinecke, J. W. ATP-binding cassette transporter A1: a cell cholesterol exporter that protects against cardiovascular disease. Physiol. Rev. 85, 1343–1372 (2005).
Tang, C. & Oram, J. F. The cell cholesterol exporter ABCA1 as a protector from cardiovascular disease and diabetes. Biochim. Biophys. Acta 1791, 563–572 (2009).
Zhu, X. et al. Macrophage ABCA1 reduces MyD88-dependent Toll-like receptor trafficking to lipid rafts by reduction of lipid raft cholesterol. J. Lipid Res. 51, 3196–3206 (2010).
Loirand, G., Guerin, P. & Pacaud, P. Rho kinases in cardiovascular physiology and pathophysiology. Circ. Res. 98, 322–334 (2006).
Liao, X. et al. Macrophage autophagy plays a protective role in advanced atherosclerosis. Cell Metab. 15, 545–553 (2012).
Razani, B. et al. Autophagy links inflammasomes to atherosclerotic progression. Cell Metab. 15, 534–544 (2012).
Davies, S. P., Reynolds, G. M. & Stamataki, Z. Clearance of apoptotic cells by tissue epithelia: a putative role for hepatocytes in liver efferocytosis. Front. Immunol. 9, 44 (2018).
Llacuna, L. et al. Growth arrest-specific protein 6 is hepatoprotective against murine ischemia/reperfusion injury. Hepatology 52, 1371–1379 (2010).
Vives-Pi, M., Rodriguez-Fernandez, S. & Pujol-Autonell, I. How apoptotic beta-cells direct immune response to tolerance or to autoimmune diabetes: a review. Apoptosis 20, 263–272 (2015).
O’Brien, B. A. et al. A deficiency in the in vivo clearance of apoptotic cells is a feature of the NOD mouse. J. Autoimmun. 26, 104–115 (2006).
Khalifeh-Soltani, A. et al. Mfge8 promotes obesity by mediating the uptake of dietary fats and serum fatty acids. Nat. Med. 20, 175–183 (2014).
Khanna, S. et al. Macrophage dysfunction impairs resolution of inflammation in the wounds of diabetic mice. PLoS One 5, e9539 (2010).
Maruyama, K. et al. Decreased macrophage number and activation lead to reduced lymphatic vessel formation and contribute to impaired diabetic wound healing. Am. J. Pathol. 170, 1178–1191 (2007).
Bossi, F. et al. C1q as a unique player in angiogenesis with therapeutic implication in wound healing. Proc. Natl Acad. Sci. USA 111, 4209–4214 (2014).
Bosurgi, L. et al. Macrophage function in tissue repair and remodeling requires IL-4 or IL-13 with apoptotic cells. Science 356, 1072–1076 (2017). While the cytokines IL-4 and IL-13 are involved in the repair of tissue damage during helminth infections, this article shows that efferocytosis of apoptotic cells by macrophages is also required for the repair response.
Weavers, H., Evans, I. R., Martin, P. & Wood, W. Corpse engulfment generates a molecular memory that primes the macrophage inflammatory response. Cell 165, 1658–1671 (2016). In a Drosophila model of tissue damage, efferocytosis of apoptotic cells is required to prime the participation of macrophages in tissue repair.
Beccafico, S. et al. Human muscle satellite cells show age-related differential expression of S100B protein and RAGE. Age 33, 523–541 (2011).
Zhang, S. et al. Efferocytosis fuels requirements of fatty acid oxidation and the electron transport chain to polarize macrophages for tissue repair. Cell Metab. 29, 443–456.e5 (2019). During wound injury, clearance of dying cells fills the phagocytic cell with a metabolite load nearly equal to that at rest. Here, the authors investigate how metabolic phagocytic signalling regulates the signature anti-inflammatory macrophage response.
Kawane, K. et al. Chronic polyarthritis caused by mammalian DNA that escapes from degradation in macrophages. Nature 443, 998–1002 (2006).
Park, M. C., Kwon, Y. J., Chung, S. J., Park, Y. B. & Lee, S. K. Liver X receptor agonist prevents the evolution of collagen-induced arthritis in mice. Rheumatology 49, 882–890 (2010).
Elliott, M. R. & Ravichandran, K. S. ELMO1 signaling in apoptotic germ cell clearance and spermatogenesis. Ann. NY Acad. Sci. 1209, 30–36 (2010).
Burnier, L. et al. Gas6 deficiency in recipient mice of allogeneic transplantation alleviates hepatic graft-versus-host disease. Blood 115, 3390–3397 (2010).
Yan, X. et al. Annexin-V promotes anti-tumor immunity and inhibits neuroblastoma growth in vivo. Cancer Immunol. Immunother. 61, 1917–1927 (2012).
Stach, C. M. et al. Treatment with annexin V increases immunogenicity of apoptotic human T-cells in Balb/c mice. Cell Death Differ. 7, 911–915 (2000).
Schlegel, J. et al. MERTK receptor tyrosine kinase is a therapeutic target in melanoma. J. Clin. Invest. 123, 2257–2267 (2013).
Suresh, K. et al. CD36 mediates H2O2-induced calcium influx in lung microvascular endothelial cells. Am. J. Physiol. Lung Cell Mol. Physiol. 312, L143–L153 (2017). Genetic loss or pharmacologic inhibition of CD36 attenuates the damage induced by hydrogen peroxide in the lung and protects from ischaemia–reperfusion injury.
Segawa, K. et al. Caspase-mediated cleavage of phospholipid flippase for apoptotic phosphatidylserine exposure. Science 344, 1164–1168 (2014).
Li, H., Zhu, H., Xu, C. J. & Yuan, J. Cleavage of BID by caspase 8 mediates the mitochondrial damage in the Fas pathway of apoptosis. Cell 94, 491–501 (1998).
Weinlich, R., Oberst, A., Beere, H. M. & Green, D. R. Necroptosis in development, inflammation and disease. Nat. Rev. Mol. Cell Biol. 18, 127–136 (2017).
Tummers, B. & Green, D. R. Caspase-8: regulating life and death. Immunol. Rev. 277, 76–89 (2017).
Ingram, J. P. et al. ZBP1/DAI drives RIPK3-mediated cell death induced by IFNs in the absence of RIPK1. J. Immunol. 203, 1348–1355 (2019).
Kaiser, W. J. et al. Toll-like receptor 3-mediated necrosis via TRIF, RIP3, and MLKL. J. Biol. Chem. 288, 31268–31279 (2013).
Man, S. M., Karki, R. & Kanneganti, T. D. Molecular mechanisms and functions of pyroptosis, inflammatory caspases and inflammasomes in infectious diseases. Immunol. Rev. 277, 61–75 (2017).
Kayagaki, N. et al. Caspase-11 cleaves gasdermin D for non-canonical inflammasome signalling. Nature 526, 666–671 (2015). Caspase 11 cleavage of gasdermin D is responsible for pyroptotic activation, IL-1 processing and septic shock following infection with Gram-negative bacteria.
Shi, J. et al. Cleavage of GSDMD by inflammatory caspases determines pyroptotic cell death. Nature 526, 660–665 (2015). Identification of gasdermin D as a regulator of pyroptosis downstream of inflammatory caspase activation.
Ding, J. et al. Pore-forming activity and structural autoinhibition of the gasdermin family. Nature 535, 111–116 (2016).
Doll, S. et al. ACSL4 dictates ferroptosis sensitivity by shaping cellular lipid composition. Nat. Chem. Biol. 13, 91–98 (2017). ACSL4 enriches cellular membranes with long polyunsaturated fatty acids required for ferroptosis.
Kagan, V. E. et al. Oxidized arachidonic and adrenic PEs navigate cells to ferroptosis. Nat. Chem. Biol. 13, 81–90 (2017). Discovery of a highly organized oxygenation centre leading to oxidation of specific phospholipid species that direct cells towards ferroptosis.
Rider, P., Voronov, E., Dinarello, C. A., Apte, R. N. & Cohen, I. Alarmins: feel the stress. J. Immunol. 198, 1395–1402 (2017).
Roh, J. S. & Sohn, D. H. Damage-associated molecular patterns in inflammatory diseases. Immune Netw. 18, e27 (2018).
Gong, T., Liu, L., Jiang, W. & Zhou, R. DAMP-sensing receptors in sterile inflammation and inflammatory diseases. Nat. Rev. Immunol. 20, 95–112 (2020).
Yu, X., Feng, B., He, P. & Shan, L. From chaos to harmony: responses and signaling upon microbial pattern recognition. Annu. Rev. Phytopathol. 55, 109–137 (2017).
Brubaker, S. W., Bonham, K. S., Zanoni, I. & Kagan, J. C. Innate immune pattern recognition: a cell biological perspective. Annu. Rev. Immunol. 33, 257–290 (2015).
Rubartelli, A. & Lotze, M. T. Inside, outside, upside down: damage-associated molecular-pattern molecules (DAMPs) and redox. Trends Immunol. 28, 429–436 (2007).
Broz, P. & Monack, D. M. Newly described pattern recognition receptors team up against intracellular pathogens. Nat. Rev. Immunol. 13, 551–565 (2013).
Tang, D., Kang, R., Coyne, C. B., Zeh, H. J. & Lotze, M. T. PAMPs and DAMPs: signal 0 s that spur autophagy and immunity. Immunol. Rev. 249, 158–175 (2012).
Mogensen, T. H. Pathogen recognition and inflammatory signaling in innate immune defenses. Clin. Microbiol. Rev. 22, 240–273 (2009).
Takeuchi, O. & Akira, S. Pattern recognition receptors and inflammation. Cell 140, 805–820 (2010).
Le, L. Q. et al. Mice lacking the orphan G protein-coupled receptor G2A develop a late-onset autoimmune syndrome. Immunity 14, 561–571 (2001).
Mike, E. V. et al. Neuropsychiatric systemic lupus erythematosus is dependent on sphingosine-1-phosphate signaling. Front. Immunol. 9, 2189 (2018).
Miyanishi, M., Segawa, K. & Nagata, S. Synergistic effect of Tim4 and MFG-E8 null mutations on the development of autoimmunity. Int. Immunol. 24, 551–559 (2012).
Potter, P. K., Cortes-Hernandez, J., Quartier, P., Botto, M. & Walport, M. J. Lupus-prone mice have an abnormal response to thioglycolate and an impaired clearance of apoptotic cells. J. Immunol. 170, 3223–3232 (2003).
van Zoelen, M. A. & van der Poll, T. Targeting RAGE in sepsis. Crit. Care 12, 103 (2008).
Tian, L. et al. p85alpha recruitment by the CD300f phosphatidylserine receptor mediates apoptotic cell clearance required for autoimmunity suppression. Nat. Commun. 5, 3146 (2014).
Schittenhelm, L., Hilkens, C. M. & Morrison, V. L. beta2 integrins as regulators of dendritic cell, monocyte, and macrophage function. Front. Immunol. 8, 1866 (2017).
Hanayama, R., Miyasaka, K., Nakaya, M. & Nagata, S. MFG-E8-dependent clearance of apoptotic cells, and autoimmunity caused by its failure. Curr. Dir. Autoimmun. 9, 162–172 (2006).
Kruse, K. et al. Inefficient clearance of dying cells in patients with SLE: anti-dsDNA autoantibodies, MFG-E8, HMGB-1 and other players. Apoptosis 15, 1098–1113 (2010).
Cohen, P. L. & Shao, W. H. Gas6/TAM receptors in systemic lupus erythematosus. Dis. Markers 2019, 7838195 (2019).
ten Kate, M. K. & van der Meer, J. Protein S deficiency: a clinical perspective. Haemophilia 14, 1222–1228 (2008).
Ramirez-Ortiz, Z. G. et al. The scavenger receptor SCARF1 mediates the clearance of apoptotic cells and prevents autoimmunity. Nat. Immunol. 14, 917–926 (2013).
Walport, M. J., Davies, K. A. & Botto, M. C1q and systemic lupus erythematosus. Immunobiology 199, 265–285 (1998).
Zeng, T. et al. The detection of autoantibodies to ATP-binding cassette transporter A1 and its role in the pathogenesis of atherosclerosis in patients with systemic lupus erythematosus. Clin. Biochem. 45, 1342–1346 (2012).
Lutz, S. E. et al. Contribution of pannexin1 to experimental autoimmune encephalomyelitis. PLoS One 8, e66657 (2013).
Duman, J. G., Tu, Y. K. & Tolias, K. F. Emerging roles of BAI adhesion-GPCRs in synapse development and plasticity. Neural Plast. 2016, 8301737 (2016).
Wium, M., Paccez, J. D. & Zerbini, L. F. The dual role of TAM receptors in autoimmune diseases and cancer: an overview. Cells 7, 166 (2018).
Binder, M. D. et al. Gas6 deficiency increases oligodendrocyte loss and microglial activation in response to cuprizone-induced demyelination. J. Neurosci. 28, 5195–5206 (2008).
Chu, Y. et al. Enhanced synaptic connectivity and epilepsy in C1q knockout mice. Proc. Natl Acad. Sci. USA 107, 7975–7980 (2010).
Chen, X. et al. Apoptotic engulfment pathway and schizophrenia. PLoS One 4, e6875 (2009).
Li, Z. & Weinman, S. A. Regulation of hepatic inflammation via macrophage cell death. Semin. Liver Dis. 38, 340–350 (2018).
Bellan, M. et al. Gas6/TAM system: a key modulator of the interplay between inflammation and fibrosis. Int. J. Mol. Sci. 20, 5070 (2019).
Elliott, M. R. et al. Unexpected requirement for ELMO1 in clearance of apoptotic germ cells in vivo. Nature 467, 333–337 (2010).
Cattaneo, M. The platelet P2 receptors in inflammation. Hamostaseologie 35, 262–266 (2015).
Henson, P. M. & Bratton, D. L. Allergy: airway epithelial Rac1 suppresses allergic inflammation. Curr. Biol. 23, R104–R106 (2013).
Good, M. E. et al. Pannexin 1 channels as an unexpected new target of the anti-hypertensive drug spironolactone. Circ. Res. 122, 606–615 (2018).
Liu, H. & Jiang, D. Fractalkine/CX3CR1 and atherosclerosis. Clin. Chim. Acta 412, 1180–1186 (2011).
Liu, M., Tso, P. & Woods, S. C. Receptor CD36 links a risk-associated allele to obesity and metabolic disorders. J. Biol. Chem. 293, 13349–13350 (2018). Pharmacologic inhibition of integral membrane protein CD36 significantly reduces body weight gain and improves glucose tolerance in animals receiving a high-fat diet.
Boucher, P. & Herz, J. Signaling through LRP1: protection from atherosclerosis and beyond. Biochem. Pharmacol. 81, 1–5 (2011).
Bhatia, V. K. et al. Complement C1q reduces early atherosclerosis in low-density lipoprotein receptor-deficient mice. Am. J. Pathol. 170, 416–426 (2007).
Freemerman, A. J. et al. Myeloid Slc2a1-deficient murine model revealed macrophage activation and metabolic phenotype are fueled by GLUT1. J. Immunol. 202, 1265–1286 (2019).
Thorp, E. B. Mechanisms of failed apoptotic cell clearance by phagocyte subsets in cardiovascular disease. Apoptosis 15, 1124–1136 (2010).
Qi, Y. Y., Zhou, X. J. & Zhang, H. Autophagy and immunological aberrations in systemic lupus erythematosus. Eur. J. Immunol. 49, 523–533 (2019).
Duval, C., Chinetti, G., Trottein, F., Fruchart, J. C. & Staels, B. The role of PPARs in atherosclerosis. Trends Mol. Med. 8, 422–430 (2002).
Rogers, M. A. et al. Dynamin-related protein 1 inhibition attenuates cardiovascular calcification in the presence of oxidative stress. Circ. Res. 121, 220–233 (2017).
Schledzewski, K. et al. Deficiency of liver sinusoidal scavenger receptors stabilin-1 and -2 in mice causes glomerulofibrotic nephropathy via impaired hepatic clearance of noxious blood factors. J. Clin. Invest. 121, 703–714 (2011).
Frasch, S. C. et al. G2A signaling dampens colitic inflammation via production of IFN-gamma. J. Immunol. 197, 1425–1434 (2016).
Huveneers, S., Truong, H. & Danen, H. J. Integrins: signaling, disease, and therapy. Int. J. Radiat. Biol. 83, 743–751 (2007).
Marei, H. & Malliri, A. Rac1 in human diseases: the therapeutic potential of targeting Rac1 signaling regulatory mechanisms. Small GTPases 8, 139–163 (2017).
Riuzzi, F. et al. RAGE in the pathophysiology of skeletal muscle. J. Cachexia Sarcopenia Muscle 9, 1213–1234 (2018).
Wu, G. et al. Molecular insights of Gas6/TAM in cancer development and therapy. Cell Death Dis. 8, e2700 (2017).
Acknowledgements
This work was supported by grants from the US National Institutes of Health (AI40646 and CA231620 to D.R.G.; AI138492 and CA231423 to B.L.H.), American Lebanese Syrian Associated Charities and the John H. Sununu Endowed Fellowship to B.L.H. and an EMBO Long-Term Fellowship (ALTF 1526 -2016) to E.B.R.
Author information
Authors and Affiliations
Contributions
The authors contributed equally to all aspects of the article.
Corresponding authors
Ethics declarations
Competing interests
D.R.G. is on the scientific advisory board of Inzen. The other authors declare no competing interests.
Additional information
Peer review information
Nature Reviews Molecular Cell Biology thanks Edward Thorp, Martin Herrman and the other, anonymous, reviewer for their contribution to the peer review of this work.
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Glossary
- Efferocytosis
-
Engulfment and clearance of dead and dying cells, usually (although not exclusively) by myeloid cells such as macrophages.
- Entosis
-
A process by which a living cell invades or is engulfed by another. This process differs from the clearance of apoptotic cells by phagocytosis as the internalized cell is still alive.
- Inflammasome
-
Any molecular complex capable of activating caspase 1.
- Immune tolerance
-
Lack or inhibition of an adaptive immune response to specific antigens.
- Flippase
-
A membrane protein that transports phosphatidylserine from the outer leaflet to the inner leaflet of the plasma membrane.
- Scramblase
-
A membrane protein that, on activation, equilibrates (‘scrambles’) the distribution of phospholipids between the inner and outer leaflets of the plasma membrane.
- MLKL
-
Mixed-lineage kinase domain-like protein (MLKL) is the executioner in necroptosis, disrupting the plasma membrane following its phosphorylation.
- Gasdermins
-
Proteins that, on activation, create pores in the plasma membrane; gasdermin D and gasdermin E function in pyroptosis.
- Guanine nucleotide exchange factor
-
(GEF). An enzyme that activates monomeric GTPases, including RAS, RAC and RHO. GEFs stimulate the release of GDP to allow binding of GTP.
- Dynamin
-
A member of the protein GTPase family, mainly involved in the scission of newly formed vesicles from a membrane and their fusion with another membrane.
- Microtubule-associated protein 1A/1B light chain 3 (LC3)-associated phagocytosis
-
(LAP). A non-apoptotic function of several proteins of the autophagy pathway, resulting in lipidation of LC3 family proteins on the phagosome membrane, enhancing fusion of the phagosome with lysosomes.
- SNARE complex
-
Soluble N-ethylmaleimide-sensitive fusion protein attachment protein (SNAP) receptor complex involved in fusion of the vesicular membrane to other membranes.
- Phagolysosome
-
The intracellular vesicle that results from the fusion of the phagosome with lysosomes.
- Cathepsins
-
A class of proteases mostly present in lysosomes.
- LC3 family
-
The ATG8 protein family, including microtubule-associated protein 1A/1B light chain 3 (LC3) proteins, γ-aminobutyric acid receptor-associated protein (GABARAP), GEC1 and GATE-16.
- Chromophore
-
A substance or molecule that absorbs light, critical for animal and human vision (for example, retinal).
- Langerhans cells
-
Specialized dendritic cells residing in the epidermis of the skin.
- Graft-versus-host disease
-
Disease resulting from the introduction and action of allogeneic T lymphocytes, which can occur following transplant of bone marrow from which T cells have not been removed.
Rights and permissions
About this article
Cite this article
Boada-Romero, E., Martinez, J., Heckmann, B.L. et al. The clearance of dead cells by efferocytosis. Nat Rev Mol Cell Biol 21, 398–414 (2020). https://doi.org/10.1038/s41580-020-0232-1
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41580-020-0232-1
This article is cited by
-
Apoptosis dysfunction: unravelling the interplay between ZBP1 activation and viral invasion in innate immune responses
Cell Communication and Signaling (2024)
-
Targeting immunogenic cell stress and death for cancer therapy
Nature Reviews Drug Discovery (2024)
-
Rapid unleashing of macrophage efferocytic capacity via transcriptional pause release
Nature (2024)
-
BHLHE40/41 regulate microglia and peripheral macrophage responses associated with Alzheimer’s disease and other disorders of lipid-rich tissues
Nature Communications (2024)
-
Cell death by phagocytosis
Nature Reviews Immunology (2024)