Abstract
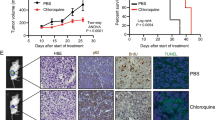
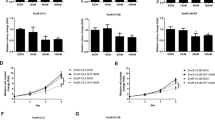
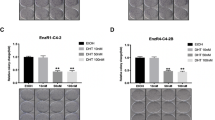
Bicalutamide is a non-steroidal antiandrogen used in the treatment of prostate cancer. Although widely accepted as an androgen receptor antagonist, the mechanism by which it induces apoptosis remains unclear. Defining exact pathways by which bicalutamide induces its apoptotic effects would help to advance its clinical applications. We aimed to (a) examine the apoptotic effects of bicalutamide at 24 h and (b) comment on the role of the caspases and calpains in mediating bicalutamide-induced apoptosis in androgen-dependent and androgen-independent cells. PWR-1E, PC-3 and DU-145 cells were treated with bicalutamide and assessed for apoptosis by flow cytometry at 24 h. DU-145 cells were used to compare differences between two different metastatic receptor-negative cells and to verify apoptotic induction at 48 h. To delineate a specific pathway of action for bicalutamide, PC-3 and PWR-1E cells were pretreated with specific inhibitors of caspase-dependent (zVAD-FMK) and caspase-independent pathways (calpain 2 inhibitor). Bicalutamide induced apoptosis in androgen-dependent PWR-1E cells via a caspase-dependent and calpain-independent mechanism. In androgen-independent PC-3 cells, bicalutamide also induced apoptosis by mechanisms that were partially inhibited by pan-caspase inhibition but were partially calpain dependent. Understanding into how bicalutamide exerts its effects in androgen-independent cells will yield further insights into the treatment of hormone-refractory disease.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 4 print issues and online access
$259.00 per year
only $64.75 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Saad F . Treatment of bone complications in advanced prostate cancer: rationale for bisphosphonate use and results of a phase III trial with zoledronic acid. Semin Oncol 2002; 29 (6 Suppl 21): 19–27.
Blackledge G . Casodex—mechanisms of action and opportunities for usage. Cancer 1993; 72 (12 Suppl): 3830–3833.
Anderson J . The role of antiandrogen monotherapy in the treatment of prostate cancer. BJU Int 2003; 91: 455–461.
Lekas E, Bergh A, Damber JE . Effects of finasteride and bicalutamide on prostatic blood flow in the rat. BJU Int 2000; 85: 962–965.
Nickerson T, Pollak M . Bicalutamide. (Casodex)-induced prostate regression involves increased expression of genes encoding insulin-like growth factor binding proteins. Urology 1999; 54: 1120–1125.
Culig Z, Hoffmann J, Erdel M, Hobisch A, Hittmair A, Bartsch G . Switch from antagonist to agonist of the androgen receptor bicalutamide is associated with prostate tumour progression in a new model system. Br J Cancer 1999; 81: 242–251.
Madarova J, Lukesova M, Hlobilkova A, Rihakova P, Murray PG, Student V . Androgen sensitivity related proteins in hormone-sensitive and hormone-insensitive prostate cancer cell lines treated by androgen antagonist bicalutamide. Neoplasma 2001; 48: 419–424.
Zhan P, Lee EC, Packman K, Tenniswood M . Induction of invasive phenotype by casodex in hormone-sensitive prostate cancer cells. J Steroid Biochem Mol Biol 2002; 83: 101–111.
Kerr JF, Wyllie AH, Currie AR . Apoptosis: a basic biological phenomenon with wide-ranging implications in tissue kinetics. Br J Cancer 1972; 26: 239–257.
Hetts SW . To die or not to die: an overview of apoptosis and its role in disease. JAMA 1998; 279: 300–307.
Borner C, Monney L . Apoptosis without caspases: an inefficient molecular guillotine? Cell Death Differ 1999; 6: 497–507.
Masiello D, Cheng S, Bubley GJ, Lu LM, Balk SP . Bicalutamide functions as an androgen receptor antagonist by assembly of a transcriptionally inactive receptor. J Biol Chem 2002; 277: 26321–26326.
Lee EC, Zhan P, Schallhom R, Packman K, Tenniswood M . Antiandrogen-induced cell death in LNCaP human prostate cancer cells. Cell Death Differ 2003; 10: 761–771.
Chen CD, Welsbie DS, Tran C, Baek SH, Chen R, Vasella T et al. Molecular determinants of resistance to antiandrogen therapy. Nat Med 2004; 10: 33–39.
Berges RR, Vukanovic J, Epstein JI, Carmichel M, Cisek L, Johnson DE et al. Implication of cell kinetic changes during the progression of human prostatic cancer. Clin Cancer Res 1995; 1: 473–480.
Denis L, Mahler C . Pharmacodynamics and pharmacokinetics of bicalutamide: defining an active dosing regimen. Urology 1996; 47 (1A suppl): 26–28.
Vicentini C, Festuccia C, Angelucci A, Gravina GL, Muzi P, Eleuterio E et al. Bicalutamide dose dependently inhibits proliferation in human prostatic carcinoma cell lines and primary cultures. Anticancer Res 2002; 22: 2917–2922.
Li X, Marani M, Mannucci R, Kinsey B, Andriani F, Nicoletti I et al. Overexpression of BCL-X(L) underlies the molecular basis for resistance to staurosporine-induced apoptosis in PC-3 cells. Cancer Res 2001; 61: 1699–1706.
Carafoli E, Molinari M . Calpain: a protease in search of a function? Biochem Biophys Res Commun 1998; 247: 193–203.
Wood DE, Newcomb EW . Caspase-dependent activation of calpain during drug-induced apoptosis. J Biol Chem 1999; 274: 8309–8315.
Mamoune A, Luo JH, Lauffenburger D, Wells A . Calpain-2 as a target for limiting prostate cancer invasion. Cancer Res 2003; 63: 4632–4640.
Zhang H, Hoff H, Sell C . Downregulation of IRS-1 protein in thapsigargin-treated human prostate epithelial cells. Exp Cell Res 2003; 289: 352–358.
Zhu DM, Uckun FM . Calpain inhibitor II induces caspase-dependent apoptosis in human acute lymphoblastic leukemia and non-Hodgkin's lymphoma cells as well as some solid tumor cells. Clin Cancer Res 2000; 6: 2456–2463.
Lu Q, Mellgren RL . Calpain inhibitors and serine protease inhibitors can produce apoptosis in HL-60 cells. Arch Biochem Biophys 1996; 334: 175–181.
Bouchal J, Baunforth KNR, Svachova MM, Murray PG, Von Angerer E, Kolar Z . Microarray analysis of bicalutamide action on telomerase activity, p53 pathway and viability of prostate carcinoma cell lines. J Pharm Pharmacol 2005; 57: 83–92.
Acknowledgements
Michael St John Floyd Jr was a British Urological Foundation Scholar during the course of this work.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
St John Floyd, M., Teahan, S., Fitzpatrick, J. et al. Differential mechanisms of bicalutamide-induced apoptosis in prostate cell lines. Prostate Cancer Prostatic Dis 12, 25–33 (2009). https://doi.org/10.1038/pcan.2008.23
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/pcan.2008.23
Keywords
This article is cited by
-
Renal Damaging Effect Elicited by Bicalutamide Therapy Uncovered Multiple Action Mechanisms As Evidenced by the Cell Model
Scientific Reports (2019)
-
Expression of FGD4 positively correlates with the aggressive phenotype of prostate cancer
BMC Cancer (2018)
-
Differential expression of AP-1 transcription factors in human prostate LNCaP and PC-3 cells: role of Fra-1 in transition to CRPC status
Molecular and Cellular Biochemistry (2017)
-
New trends for overcoming ABCG2/BCRP-mediated resistance to cancer therapies
Journal of Experimental & Clinical Cancer Research (2015)
-
Metformin enhances the antiproliferative and apoptotic effect of bicalutamide in prostate cancer
Prostate Cancer and Prostatic Diseases (2012)