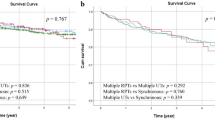
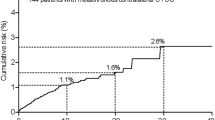
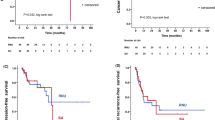
Abstract
Upper urinary tract urothelial carcinoma (UTUC) is a rare disease, which means there are little evidence-based data available to guide clinical decision-making. Although diagnosis and treatment of UTUC have improved significantly over the last 5 years, accurate risk stratification remains a challenge owing to the difficulty of clinical staging. A number of potential prognostic factors have been identified, encompassing clinical characteristics, pathological factors and molecular markers. Tumor stage and lymph node status are the most important predictors of survival in patients with UTUC. Preoperative evaluation for hydronephrosis can identify patients at risk of non-organ-confined disease. In the subgroup of patients with stage ≥pT2 disease, a longer interval between diagnosis and radical nephroureterectomy is associated with a higher risk of disease recurrence and cancer-specific mortality. Extensive tumor necrosis, sessile tumor architecture and lymphovascular invasion are independent predictors of clinical outcomes for patients with UTUC treated with radical nephroureterectomy. The incorporation of such prognosticators into clinical prediction models might help to guide decision-making with regard to timing of surveillance, type of treatment, performance of lymphadenectomy, and consideration of neoadjuvant or adjuvant systemic therapies.
Key Points
-
Tumor stage and lymph node status are the most important predictors of survival in patients with upper urinary tract urothelial carcinoma (UTUC)
-
Preoperative evaluation for hydronephrosis can identify patients at risk of non-organ-confined UTUC
-
In the subgroup of patients with stage ≥pT2 disease, a longer interval from diagnosis to radical nephroureterectomy (more than 3 months) is associated with higher risk of disease recurrence and cancer-specific mortality
-
Tumor location (renal pelvis versus ureter) has no prognostic impact when adjusted for the effects of established features of disease severity
-
Extensive tumor necrosis (defined as >10% of the tumor area) and sessile tumor architecture are independent predictors of clinical outcomes for patients with UTUC treated with radical nephroureterectomy
-
Lymphovascular invasion is associated with established features of biologically aggressive UTUC and is a strong predictor of disease recurrence and cancer-specific mortality
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Jemal, A., Siegel, R., Xu, J. & Ward, E. Cancer statistics, 2010. CA Cancer J. Clin. 60, 277–300 (2010).
Margulis, V. et al. Preoperative multivariable prognostic model for prediction of nonorgan confined urothelial carcinoma of the upper urinary tract. J. Urol. 184, 453–458 (2010).
Rouprêt, M. et al. European guidelines for the diagnosis and management of upper urinary tract urothelial cell carcinomas: 2011 update. Eur. Urol. 59, 584–594 (2011).
Shariat, S. F. et al. Multiple biomarkers improve prediction of bladder cancer recurrence and mortality in patients undergoing cystectomy. Cancer 112, 315–325 (2008).
Shariat, S. F. et al. Combination of multiple molecular markers can improve prognostication in patients with locally advanced and lymph node positive bladder cancer. J. Urol. 183, 68–75 (2010).
Lughezzani, G. et al. Gender-related differences in patients with stage I to III upper tract urothelial carcinoma: results from the Surveillance, Epidemiology, and End Results database. Urology 75, 321–327 (2010).
Fernández, M. I. et al. Evidence-based sex-related outcomes after radical nephroureterectomy for upper tract urothelial carcinoma: results of large multicenter study. Urology 73, 142–146 (2009).
Shariat, S. F. et al. Gender differences in radical nephroureterectomy for upper tract urothelial carcinoma. World J. Urol. doi: 10.1007/s00345-010-0594-7.
Svatek, R. A. et al. Evidence-based gender related outcomes after radical cystectomy: results of a large multicenter study [abstract 1744]. J. Urol. 181, 629 (2009).
Jeldres, C. et al. Gender is an important predictor of cancer-specific survival in patient with transitional cell carcinoma after radical cystectomy [abstract 1761]. J. Urol. 181, 635 (2009).
Shariat, S. F. et al. Advanced patient age is associated with inferior cancer-specific survival after radical nephroureterectomy. BJU Int. 105, 1672–1677 (2010).
Chromecki, T. F. et al. Chronological age is not an independent predictor of clinical outcomes after radical nephroureterectomy. World J. Urol. doi: 10.1007/s00345-011-0677-0.
Matsumoto, K. et al. Racial differences in the outcome of patients with urothelial carcinoma of the upper urinary tract: an international study BJU Int. doi: 10.1111/j.1464-410X.2011.10188.x.
Oosterlinck, W. et al. EAU guidelines on diagnosis and treatment of upper urinary tract transitional cell carcinoma. Eur. Urol. 46, 147–154 (2004).
Hall, M. C. et al. Prognostic factors, recurrence, and survival in transitional cell carcinoma of the upper urinary tract: a 30-year experience in 252 patients. Urology 52, 594–601 (1998).
Raman, J. D. et al. Does preoperative symptom classification impact prognosis in patients with clinically localized upper-tract urothelial carcinoma managed by radical nephroureterectomy? Urol. Oncol. doi: 10.1016/j.urolonc.2009.11.007.
Brien, J. C. et al. Preoperative hydronephrosis, ureteroscopic biopsy grade and urinary cytology can improve prediction of advanced upper tract urothelial carcinoma. J. Urol. 184, 69–73 (2010).
Stimson, C. J. et al. Preoperative hydronephrosis predicts extravesical and node positive disease in patients undergoing cystectomy for bladder cancer. J. Urol. 183, 1732–1737 (2010).
Cho, K. S., Hong, S. J., Cho, N. H. & Choi, Y. D. Grade of hydronephrosis and tumor diameter as preoperative prognostic factors in ureteral transitional cell carcinoma. Urology 70, 662–666 (2007).
Ng, C. K. et al. Does the presence of hydronephrosis on preoperative axial CT imaging predict worse outcomes for patients undergoing nephroureterectomy for upper-tract urothelial carcinoma? Urol. Oncol. 29, 27–32 (2011).
Gore, J. L. et al. Mortality increases when radical cystectomy is delayed more than 12 weeks: results from a Surveillance, Epidemiology, and End Results-Medicare analysis. Cancer 115, 988–996 (2009).
Nielsen, M. E. et al. A delay in radical cystectomy of >3 months is not associated with a worse clinical outcome. BJU Int. 100, 1015–1020 (2007).
Waldert, M. et al. A delay in radical nephroureterectomy can lead to upstaging. BJU Int. 105, 812–817 (2010).
Margulis, V. et al. Outcomes of radical nephroureterectomy: a series from the Upper Tract Urothelial Carcinoma Collaboration. Cancer 115, 1224–1233 (2009).
Scher, H. et al. NCCN urothelial cancer practice guidelines. National Comprehensive Cancer Network. Oncology (Williston Park) 12, 225–271 (1998).
Capitanio, U. et al. Comparison of oncologic outcomes for open and laparoscopic nephroureterectomy: a multi-institutional analysis of 1249 cases. Eur. Urol. 56, 1–9 (2009).
Favaretto, R. L. et al. Comparison between laparoscopic and open radical nephroureterectomy in a contemporary group of patients: are recurrence and disease-specific survival associated with surgical technique? Eur. rol. 58, 645–651 (2010).
Kamihira, O. et al. Laparoscopic radical nephroureterectomy: a multicenter analysis in Japan. Eur. Urol. 55, 1397–1407 (2009).
Rouprêt, M. et al. Oncologic control after open or laparoscopic nephroureterectomy for upper urinary tract transitional cell carcinoma: a single center experience. Urology 69, 656–661 (2007).
Favaretto, R. L. et al. The effect of tumor location on prognosis in patients treated with radical nephroureterectomy at Memorial Sloan–Kettering Cancer Center. Eur. Urol. 58, 574–580 (2010).
Isbarn, H. et al. Location of the primary tumor is not an independent predictor of cancer specific mortality in patients with upper urinary tract urothelial carcinoma. J. Urol. 182, 2177–2181 (2009).
Raman, J. D. et al. Impact of tumor location on prognosis for patients with upper tract urothelial carcinoma managed by radical nephroureterectomy. Eur. Urol. 57, 1072–1079 (2010).
Langner, C. et al. pT classification, grade, and vascular invasion as prognostic indicators in urothelial carcinoma of the upper urinary tract. Mod. Pathol. 19, 272–279 (2006).
Novara, G. et al. Independent predictors of cancer-specific survival in transitional cell carcinoma of the upper urinary tract: multi-institutional dataset from 3 European centers. Cancer 110, 1715–1722 (2007).
Novara, G. et al. Prognostic role of lymphovascular invasion in patients with urothelial carcinoma of the upper urinary tract: an international validation study. Eur. Urol. 57, 1064–1071 (2010).
Lopez-Beltran, A., Bassi, P., Pavone-Macaluso, M. & Montironi, R. Handling and pathology reporting of specimens with carcinoma of the urinary bladder, ureter, and renal pelvis. Eur. Urol. 45, 257–266 (2004).
Bolenz, C. et al. Risk stratification of patients with nodal involvement in upper tract urothelial carcinoma: value of lymph-node density. BJU Int. 103, 302–306 (2009).
Mullerad, M. et al. Bladder cancer as a prognostic factor for upper tract transitional cell carcinoma. J. Urol. 172, 2177–2181 (2004).
Lehmann, J. et al. Transitional cell carcinoma of the ureter: prognostic factors influencing progression and survival. Eur. Urol. 51, 1281–1288 (2007).
Li, W. M. et al. The prognostic predictors of primary ureteral transitional cell carcinoma after radical nephroureterectomy. J. Urol. 182, 451–458 (2009).
Li, C. C. et al. Significant predictive factors for prognosis of primary upper urinary tract cancer after radical nephroureterectomy in Taiwanese patients. Eur. Urol. 54, 1127–1134 (2008).
Remzi, M. et al. Tumour architecture is an independent predictor of outcomes after nephroureterectomy: a multi-institutional analysis of 1363 patients. BJU Int. 103, 307–311 (2009).
Chade, D. C. et al. Clinical outcome of primary versus secondary bladder carcinoma in situ. J. Urol. 184, 464–469 (2010).
Shariat, S. F. et al. Concomitant carcinoma in situ is a feature of aggressive disease in patients with organ-confined TCC at radical cystectomy. Eur. Urol. 51, 152–160 (2007).
Pieras, E. et al. Concomitant carcinoma in situ and tumour size are prognostic factors for bladder recurrence after nephroureterectomy for upper tract transitional cell carcinoma. BJU Int. 106, 1319–1323 (2010).
Wheat, J. C. et al. Concomitant carcinoma in situ is a feature of aggressive disease in patients with organ confined urothelial carcinoma following radical nephroureterectomy. Urol. Oncol. doi: 10.1016/j.urolonc.2010.01.001.
Otto, W. et al. Concomitant carcinoma in situ as an independent prognostic parameter for recurrence and survival in upper tract urothelial carcinoma: a multicenter analysis of 772 patients. World J. Urol. doi: 10.1007/s00345-011-0645-8.
Langner, C. et al. Tumor necrosis as prognostic indicator in transitional cell carcinoma of the upper urinary tract. J. Urol. 176, 910–913 (2006).
Zigeuner, R. et al. Tumour necrosis is an indicator of aggressive biology in patients with urothelial carcinoma of the upper urinary tract. Eur. Urol. 57, 575–581 (2010).
Seitz, C. et al. Association of tumor necrosis with pathological features and clinical outcome in 754 patients undergoing radical nephroureterectomy for upper tract urothelial carcinoma: an international validation study. J. Urol. 184, 1895–1900 (2010).
Hong, B. et al. Prognostic value of lymphovascular invasion in transitional cell carcinoma of upper urinary tract. Urology 65, 692–696 (2005).
Kim, D. S. et al. Lymphovascular invasion and pT stage are prognostic factors in patients treated with radical nephroureterectomy for localized upper urinary tract transitional cell carcinoma. Urology 75, 328–332 (2010).
Saito, K. et al. Lymphovascular invasion is independently associated with poor prognosis in patients with localized upper urinary tract urothelial carcinoma treated surgically. J. Urol. 178, 2291–2296 (2007).
Bolenz, C. et al. Lymphangiogenesis occurs in upper tract urothelial carcinoma and correlates with lymphatic tumour dissemination and poor prognosis. BJU Int. 103, 1040–1046 (2009).
Chung, S. D. et al. Lymphovascular invasion predicts poor outcome of urothelial carcinoma of renal pelvis after nephroureterectomy. BJU Int. 103, 1047–1051 (2009).
Kikuchi, E. et al. Lymphovascular invasion predicts clinical outcomes in patients with node-negative upper tract urothelial carcinoma. J. Clin. Oncol. 27, 612–618 (2009).
Margulis, V., Lotan, Y., Montorsi, F. & Shariat, S. F. Predicting survival after radical cystectomy for bladder cancer. BJU Int. 102, 15–22 (2008).
Simone, G. et al. Independent prognostic value of tumour diameter and tumour necrosis in upper urinary tract urothelial carcinoma. BJU Int. 103, 1052–1057 (2009).
Langner, C., Hutterer, G., Chromecki, T., Rehak, P. & Zigeuner, R. Patterns of invasion and histological growth as prognostic indicators in urothelial carcinoma of the upper urinary tract. Virchows Arch. 448, 604–611 (2006).
Fritsche, H. M. et al. Macroscopic sessile tumor architecture is a pathologic feature of biologically aggressive upper tract urothelial carcinoma. Urol. Oncol. doi: 10.1016/j.urolonc.2010.07.010.
Fromont, G. et al. Tissue microarray analysis of the prognostic value of E-cadherin, Ki67, p53, p27, survivin and MSH2 expression in upper urinary tract transitional cell carcinoma. Eur. Urol. 48, 764–770 (2005).
Winkler, A. et al. CD24 expression in urothelial carcinoma of the upper urinary tract correlates with tumour progression. Virchows Arch. 450, 59–64 (2007).
Nakanishi, K. et al. Expression of hypoxia-inducible factor-1alpha protein predicts survival in patients with transitional cell carcinoma of the upper urinary tract. Clin. Cancer Res. 11, 2583–2590 (2005).
Kamai, T. et al. Prognostic significance of p27Kip1 and Ki-67 expression in carcinoma of the renal pelvis and ureter. BJU Int. 86, 14–19 (2000).
Barbieri, C. E. et al. Tissue-based molecular markers for bladder cancer. Minerva Urol. Nefrol. 62, 241–258 (2010).
Zigeuner, R. et al. Prognostic impact of p63 and p53 expression in upper urinary tract transitional cell carcinoma. Urology 63, 1079–1083 (2004).
Nakanishi, K. et al. Expression of bcl-2 oncoprotein in transitional cell carcinoma of the upper urinary tract. Virchows Arch. 432, 445–450 (1998).
Nakanishi, K. et al. Expression of survivin does not predict survival in patients with transitional cell carcinoma of the upper urinary tract. Virchows Arch. 441, 559–563 (2002).
Nakanishi, K. et al. Expression of telomerase mRNA component (hTR) in transitional cell carcinoma of the upper urinary tract. Cancer 86, 2109–2116 (1999).
Leibl, S. et al. EGFR expression in urothelial carcinoma of the upper urinary tract is associated with disease progression and metaplastic morphology. APMIS 116, 27–32 (2008).
Youssef, R. F. et al. Prognostic effect of urinary bladder carcinoma in situ on clinical outcome of subsequent upper tract urothelial carcinoma. Urology 77, 861–866 (2010).
Eltz, S., Comperat, E., Cussenot, O. & Rouprêt, M. Molecular and histological markers in urothelial carcinomas of the upper urinary tract. BJU Int. 102, 532–535 (2008).
Akkad, T. et al. Fluorescence in situ hybridization for detecting upper urinary tract tumors—a preliminary report. Urology 70, 753–757 (2007).
Luo, B. et al. Utility of fluorescence in situ hybridization in the diagnosis of upper urinary tract urothelial carcinoma. Cancer Genet. Cytogenet. 189, 93–97 (2009).
Mian, C. et al. Fluorescence in situ hybridisation in the diagnosis of upper urinary tract tumours. Eur. Urol. 58, 288–292 (2010).
Herman, M. P., Svatek, R. S., Lotan, Y., Karakiewicz, P. I. & Shariat, S. F. Urine-based biomarkers for the early detection and surveillance of non-muscle invasive bladder cancer. Minerva Urol. Nefrol. 60, 217–235 (2008).
Nieder, A. M., Soloway, M. S. & Herr, H. W. Should we abandon the FISH test? Eur. Urol. 51, 1469–1471 (2007).
Chen, A. A. & Grasso, M. Is there a role for FISH in the management and surveillance of patients with upper tract transitional-cell carcinoma? J. Endourol. 22, 1371–1374 (2008).
Johannes, J. R., Nelson, E., Bibbo, M. & Bagley, D. H. Voided urine fluorescence in situ hybridization testing for upper tract urothelial carcinoma surveillance. J. Urol. 184, 879–882 (2010).
Rouprêt, M. et al. Microsatellite instability as indicator of MSH2 gene mutation in patients with upper urinary tract transitional cell carcinoma. J. Med. Genet. 41, e91 (2004).
Acher, P., Kiela, G., Thomas, K. & O'Brien, T. Towards a rational strategy for the surveillance of patients with Lynch syndrome (hereditary non-polyposis colon cancer) for upper tract transitional cell carcinoma. BJU Int. 106, 300–302 (2010).
Rouprêt, M., Yates, D. R., Comperat, E. & Cussenot, O. Upper urinary tract urothelial cell carcinomas and other urological malignancies involved in the hereditary nonpolyposis colorectal cancer (lynch syndrome) tumor spectrum. Eur. Urol. 54, 1226–1236 (2008).
Rouprêt, M. et al. Checkup and management of upper urinary tract tumours in 2010: An update from the committee of cancer from the French National Association of Urology [French]. Prog. Urol. 20, 260–271 (2010).
Rouprêt, M., Azzouzi, A. R. & Cussenot, O. Microsatellite instability and transitional cell carcinoma of the upper urinary tract. BJU Int. 96, 489–492 (2005).
Saito, K. et al. The impact of preoperative serum C-reactive protein on the prognosis of patients with upper urinary tract urothelial carcinoma treated surgically. BJU Int. 100, 269–273 (2007).
Elstein, A. S. Heuristics and biases: selected errors in clinical reasoning. Acad. Med. 74, 791–794 (1999).
Vlaev, I. & Chater, N. Game relativity: how context influences strategic decision making. J. Exp. Psychol. Learn. Mem. Cogn. 32, 131–149 (2006).
Hogarth, R. M. & Karelaia, N. Heuristic and linear models of judgment: matching rules and environments. Psychol. Rev. 114, 733–758 (2007).
Ross, P. L. et al. Comparisons of nomograms and urologists' predictions in prostate cancer. Semin. Urol. Oncol. 20, 82–88 (2002).
Ross, P. L., Scardino, P. T. & Kattan, M. W. A catalog of prostate cancer nomograms. J. Urol. 165, 1562–1568 (2001).
Specht, M. C., Kattan, M. W., Gonen, M., Fey, J. & Van Zee, K. J. Predicting nonsentinel node status after positive sentinel lymph biopsy for breast cancer: clinicians versus nomogram. Ann. Surg. Oncol. 12, 654–659 (2005).
Walz, J. et al. Clinicians are poor raters of life-expectancy before radical prostatectomy or definitive radiotherapy for localized prostate cancer. BJU Int. 100, 1254–1258 (2007).
Shariat, S. F., Capitanio, U., Jeldres, C. & Karakiewicz, P. I. Can nomograms be superior to other prediction tools? BJU Int. 103, 492–495 (2009).
Shariat, S. F., Karakiewicz, P. I., Suardi, N. & Kattan, M. W. Comparison of nomograms with other methods for predicting outcomes in prostate cancer: a critical analysis of the literature. Clin. Cancer Res. 14, 4400–4407 (2008).
Bensalah, K., Montorsi, F. & Shariat, S. F. Challenges of cancer biomarker profiling. Eur. Urol. 52, 1601–1609 (2007).
Acknowledgements
C. P. Vega, University of California, Irvine, CA, is the author of and is solely responsible for the content of the learning objectives, questions and answers of the Medscape, LLC-accredited continuing medical education activity associated with this article.
Author information
Authors and Affiliations
Contributions
T. F. Chromecki was responsible for researching the data and writing the article. T. F Chromecki, K. Bensalah, M. Remzi, G. Verhoest, E. K. Cha, D. S. Scherr, G. Novara, P. I. Karakiewicz and S. F. Shariat contributed equally to the discussion of content and reviewing the manuscript before submission.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Chromecki, T., Bensalah, K., Remzi, M. et al. Prognostic factors for upper urinary tract urothelial carcinoma. Nat Rev Urol 8, 440–447 (2011). https://doi.org/10.1038/nrurol.2011.96
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrurol.2011.96
This article is cited by
-
Effect of subsequent bladder cancer on survival in upper tract urothelial carcinoma patients post-radical nephroureterectomy: a systematic review and meta-analysis
BMC Urology (2023)
-
Presence of secondary bladder cancer following radical nephroureterectomy for upper tract urothelial carcinoma: characteristics, risk factors, and predictive value
BMC Urology (2022)
-
Pre-therapy serum albumin-to-globulin ratio in patients treated with neoadjuvant chemotherapy and radical nephroureterectomy for upper tract urothelial carcinoma
World Journal of Urology (2021)
-
Transperitoneal radical nephroureterectomy is associated with worse disease progression than retroperitoneal radical nephroureterectomy in patients with upper urinary tract urothelial carcinoma
Scientific Reports (2019)
-
Optimal Management of Upper Tract Urothelial Carcinoma: an Unmet Need
Current Treatment Options in Oncology (2019)