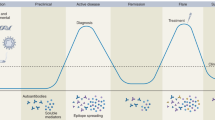
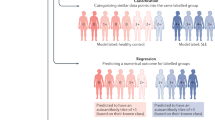
Key Points
-
Current systemic autoimmune disease classification criteria are commonly used for diagnosing diseases and tend to be ambiguous, generic or difficult to assess, which might lead to misdiagnosis or leave patients undiagnosed
-
Ample evidence demonstrates that rheumatic autoimmune diseases share molecular disease pathways
-
Discriminating between diseases by comparing molecular profiles is a feasible approach that could be useful in clinical practice, if adequately implemented
-
Disease stratification has previously been based on supervised methods and predefined clinical diagnoses
-
Integration of multi-layered data with unsupervised clustering should provide valuable information about disease mechanisms and treatment responses by simultaneously considering multiple types of information
-
Precision medicine of systemic autoimmune diseases should start with a new robust molecular classification of autoimmune disorders and the identification of biomarkers for use in routine clinical practice
Abstract
Autoimmune rheumatic diseases pose many problems that have, in general, already been solved in the field of cancer. The heterogeneity of each disease, the clinical similarities and differences between different autoimmune rheumatic diseases and the large number of patients that remain without a diagnosis underline the need to reclassify these diseases via new approaches. Knowledge about the molecular basis of systemic autoimmune diseases, along with the availability of bioinformatics tools capable of handling and integrating large volumes of various types of molecular data at once, offer the possibility of reclassifying these diseases. A new taxonomy could lead to the discovery of new biomarkers for patient stratification and prognosis. Most importantly, this taxonomy might enable important changes in clinical trial design to reach the expected outcomes or the design of molecularly targeted therapies. In this Review, we discuss the basis for a new molecular taxonomy for autoimmune rheumatic diseases. We highlight the evidence surrounding the idea that these diseases share molecular features related to their pathogenesis and development and discuss previous attempts to classify these diseases. We evaluate the tools available to analyse and combine different types of molecular data. Finally, we introduce PRECISESADS, a project aimed at reclassifying the systemic autoimmune diseases.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Change history
21 February 2018
In the original version of this article, concatenation and non-concatenation were incorrectly referred to as catenation and non-catenation in the subheadings in Table 2 and in a subheading on page 87 in the main text. These errors have now been corrected in the PDF and HTML versions of the article.
References
Cancer Genome Atlas Research Network et al. The Cancer Genome Atlas pan-cancer analysis project. Nat. Genet. 45, 1113–1120 (2013).
Hoadley, K. A. et al. Multiplatform analysis of 12 cancer types reveals molecular classification within and across tissues of origin. Cell 158, 929–944 (2014).
Curtis, C. et al. The genomic and transcriptomic architecture of 2,000 breast tumours reveals novel subgroups. Nature 486, 346–352 (2012).
Cohen, A. S. et al. Preliminary criteria for the classification of systemic lupus erythematosus. Bull. Rheum. Dis. 21, 643–648 (1971).
Tan, E. M. et al. The 1982 revised criteria for the classification of systemic lupus erythematosus. Arthritis Rheum. 25, 1271–1277 (1982).
Hochberg, M. C. Updating the American College of Rheumatology revised criteria for the classification of systemic lupus erythematosus [letter]. Arthritis Rheum. 40, 1725 (1997).
Petri, M. et al. Derivation and validation of the Systemic Lupus International Collaborating Clinics classification criteria for systemic lupus erythematosus. Arthritis Rheum. 64, 2677–2686 (2012).
Wilson, W. A. et al. International consensus statement on preliminary classification criteria for definite antiphospholipid syndrome: report of an international workshop. Arthritis Rheum. 42, 1309–1311 (1999).
Miyakis, S. et al. International consensus statement on an update of the classification criteria for definite antiphospholipid syndrome (APS). J. Thromb. Haemost. 4, 295–306 (2006).
Asherson, R. A. et al. Catastrophic antiphospholipid syndrome: international consensus statement on classification criteria and treatment guidelines. Lupus 12, 530–534 (2003).
Cervera, R. et al. Validation of the preliminary criteria for the classification of catastrophic antiphospholipid syndrome. Ann. Rheum. Dis. 64, 1205–1209 (2005).
[No authors listed] DIAGNOSTIC criteria for rheumatoid arthritis: 1958 revision by a committee of the American Rheumatism Association. Ann. Rheum. Dis. 18, 49–51 (1959).
Arnett, F. C. et al. The American Rheumatism Association 1987 revised criteria for the classification of rheumatoid arthritis. Arthritis Rheum. 31, 315–324 (1988).
Aletaha, D. et al. 2010 rheumatoid arthritis classification criteria: an American College of Rheumatology/European League Against Rheumatism collaborative initiative. Ann. Rheum. Dis. 69, 1580–1588 (2010).
McDuffie, F. C. Nature and possible significance of rheumatoid factor. South. Med. J. 58, 1126–1130 (1965).
Sharp, G. C., Irvin, W. S., Tan, E. M., Gould, R. G. & Holman, H. R. Mixed connective tissue disease — an apparently distinct rheumatic disease syndrome associated with a specific antibody to an extractable nuclear antigen (ENA). Am. J. Med. 52, 148–159 (1972).
Alarcon-Segovia, D. & Cardiel, M. H. Comparison between 3 diagnostic criteria for mixed connective tissue disease. Study of 593 patients. J. Rheumatol. 16, 328–334 (1989).
Gunnarsson, R., Molberg, O., Gilboe, I. M., Gran, J. T. & Group, P. S. The prevalence and incidence of mixed connective tissue disease: a national multicentre survey of Norwegian patients. Ann. Rheum. Dis. 70, 1047–1051 (2011).
Swanton, J. & Isenberg, D. Mixed connective tissue disease: still crazy after all these years. Rheum. Dis. Clin. North Amer. 31, 421–436 (2005).
Subcommittee for Scleroderma Criteria of the American Rheumatism Association Diagnostic and Therapeutic Criteria Committee. Preliminary criteria for the classification of systemic sclerosis (scleroderma). Arthritis Rheum. 23, 581–590 (1980).
Maricq, H. R., Harper, F. E., Khan, M. M., Tan, E. M. & LeRoy, E. C. Microvascular abnormalities as possible predictors of disease subsets in Raynaud phenomenon and early connective tissue disease. Clin. Exp. Rheumatol. 1, 195–205 (1983).
Weiner, E. S. et al. Prognostic significance of anticentromere antibodies and anti-topoisomerase I antibodies in Raynaud's disease. A prospective study. Arthritis Rheum. 34, 68–77 (1991).
LeRoy, E. C. & Medsger, T. A. Jr. Criteria for the classification of early systemic sclerosis. J. Rheumatol. 28, 1573–1576 (2001).
Koenig, M. et al. Autoantibodies and microvascular damage are independent predictive factors for the progression of Raynaud's phenomenon to systemic sclerosis: a twenty-year prospective study of 586 patients, with validation of proposed criteria for early systemic sclerosis. Arthritis Rheum. 58, 3902–3912 (2008).
Lonzetti, L. S. et al. Updating the American College of Rheumatology preliminary classification criteria for systemic sclerosis: addition of severe nailfold capillaroscopy abnormalities markedly increases the sensitivity for limited scleroderma. Arthritis Rheum. 44, 735–736 (2001).
van den Hoogen, F. et al. 2013 classification criteria for systemic sclerosis: an American College of Rheumatology/European League against Rheumatism collaborative initiative. Arthritis Rheum. 65, 2737–2747 (2013).
LeRoy, E. C. et al. Scleroderma (systemic sclerosis): classification, subsets and pathogenesis. J. Rheumatol 15, 202–205 (1988).
Steen, V. D. & Medsger, T. A. Jr. Epidemiology and natural history of systemic sclerosis. Rheum. Dis. Clin. North Am. 16, 1–10 (1990).
Hao, Y. et al. Early mortality in a multinational systemic sclerosis inception cohort. Arthritis Rheumatol 69, 1067–1077 (2017).
Simeon-Aznar, C. P. et al. Registry of the Spanish network for systemic sclerosis: survival, prognostic factors, and causes of death. Medicine 94, e1728 (2015).
Fransen, J. et al. Clinical prediction of 5-year survival in systemic sclerosis: validation of a simple prognostic model in EUSTAR centres. Ann. Rheum. Dis. 70, 1788–1792 (2011).
Ho, K. T. & Reveille, J. D. The clinical relevance of autoantibodies in scleroderma. Arthritis Res. Ther. 5, 80–93 (2003).
Beretta, L. & Santaniello, A. Models for prediction of death in systemic sclerosis: current perspectives and future directions. Expert Rev. Clin. Immunol. 7, 391–393 (2011).
Assassi, S. et al. Skin gene expression correlates of severity of interstitial lung disease in systemic sclerosis. Arthritis Rheum. 65, 2917–2927 (2013).
Lofgren, S. et al. Integrated, multicohort analysis of systemic sclerosis identifies robust transcriptional signature of disease severity. JCI Insight 1, e89073 (2016).
Vigone, B. et al. Role of class II human leucocyte antigens in the progression from early to definite systemic sclerosis. Rheumatology (Oxford) 54, 707–711 (2015).
Vitali, C. et al. Classification criteria for Sjögren's syndrome: a revised version of the European criteria proposed by the American-European Consensus Group. Ann. Rheum. Dis. 61, 554–558 (2002).
Skopouli, F. N., Drosos, A. A., Papaioannou, T. & Moutsopoulos, H. M. Preliminary diagnostic criteria for Sjögren's syndrome. Scand. J. Rheumatol. Suppl. 61, 22–25 (1986).
Rasmussen, A. et al. Comparison of the American-European Consensus Group Sjögren's syndrome classification criteria to newly proposed American College of Rheumatology criteria in a large, carefully characterised sicca cohort. Ann. Rheumat. Dis. 73, 31–38 (2014).
Shiboski, S. C. et al. American College of Rheumatology classification criteria for Sjögren's syndrome: a data-driven, expert consensus approach in the Sjögren's International Collaborative Clinical Alliance cohort. Arthritis Care Res. 64, 475–487 (2012).
Shiboski, C. H. et al. 2016 American College of Rheumatology/European League Against Rheumatism classification criteria for primary Sjögren's syndrome: a consensus and data-driven methodology involving three international patient cohorts. Arthritis Rheumatol. 69, 35–45 (2017).
Miceli-Richard, C. & Criswell, L. A. Genetic, genomic and epigenetic studies as tools for elucidating disease pathogenesis in primary Sjögren's syndrome. Expert Rev. Clin. Immunol. 10, 437–444 (2014).
Li, Y. R. et al. Genetic sharing and heritability of paediatric age of onset autoimmune diseases. Nat. Commun. 6, 8442 (2015).
Alarcon-Segovia, D. et al. Familial aggregation of systemic lupus erythematosus, rheumatoid arthritis, and other autoimmune diseases in 1,177 lupus patients from the GLADEL cohort. Arthritis Rheum. 52, 1138–1147 (2005).
Jarvinen, P. & Aho, K. Twin studies in rheumatic diseases. Semin. Arthritis Rheum. 24, 19–28 (1994).
Aho, K., Koskenvuo, M., Tuominen, J. & Kaprio, J. Occurrence of rheumatoid arthritis in a nationwide series of twins. J. Rheumatol 13, 899–902 (1986).
Feghali-Bostwick, C., Medsger, T. A. Jr & Wright, T. M. Analysis of systemic sclerosis in twins reveals low concordance for disease and high concordance for the presence of antinuclear antibodies. Arthritis Rheum. 48, 1956–1963 (2003).
MacArthur, J. et al. The new NHGRI-EBI Catalog of published genome-wide association studies (GWAS Catalog). Nucleic Acids Res. 45, D896–D901 (2017).
Grumet, F. C., Coukell, A., Bodmer, J. G., Bodmer, W. F. & McDevitt, H. O. Histocompatibility (HL-A) antigens associated with systemic lupus erythematosus. A possible genetic predisposition to disease. N. Engl. J. Med. 285, 193–196 (1971).
Stastny, P. Association of the B-cell alloantigen DRw4 with rheumatoid arthritis. N. Engl. J. Med. 298, 869–871 (1978).
Cortes, A. & Brown, M. A. Promise and pitfalls of the Immunochip. Arthritis Res. Ther. 13, 101 (2011).
McCarthy, S. et al. A reference panel of 64,976 haplotypes for genotype imputation. Nat. Genet. 48, 1279–1283 (2016).
Jia, X. et al. Imputing amino acid polymorphisms in human leukocyte antigens. PLoS ONE 8, e64683 (2013).
Zheng, X. et al. HIBAG—HLA genotype imputation with attribute bagging. Pharmacogenom. J. 14, 192–200 (2014).
Raychaudhuri, S. et al. Five amino acids in three HLA proteins explain most of the association between MHC and seropositive rheumatoid arthritis. Nat. Genet. 44, 291–296 (2012).
Okada, Y. et al. Contribution of a non-classical HLA gene, HLA-DOA, to the risk of rheumatoid arthritis. Am. J. Hum. Genet. 99, 366–374 (2016).
Fernando, M. M. et al. Identification of two independent risk factors for lupus within the MHC in United Kingdom families. PLoS Genet. 3, e192 (2007).
Morris, D. L. et al. Unraveling multiple MHC gene associations with systemic lupus erythematosus: model choice indicates a role for HLA alleles and non-HLA genes in Europeans. Am. J. Hum. Genet. 91, 778–793 (2012).
Kim, K. et al. The HLA-DRβ1 amino acid positions 11-13-26 explain the majority of SLE-MHC associations. Nat. Commun. 5, 5902 (2014).
Sun, C. et al. High-density genotyping of immune-related loci identifies new SLE risk variants in individuals with Asian ancestry. Nat. Genet. 48, 323–330 (2016).
Mayes, M. D. et al. Immunochip analysis identifies multiple susceptibility loci for systemic sclerosis. Am. J. Hum. Genet. 94, 47–61 (2014).
Arnett, F. C. et al. Major histocompatibility complex (MHC) class II alleles, haplotypes and epitopes which confer susceptibility or protection in systemic sclerosis: analyses in 1300 Caucasian, African-American and Hispanic cases and 1000 controls. Ann. Rheum. Dis. 69, 822–827 (2010).
Gladman, D. D. et al. HLA markers for susceptibility and expression in scleroderma. J. Rheumatol. 32, 1481–1487 (2005).
Lessard, C. J. et al. Variants at multiple loci implicated in both innate and adaptive immune responses are associated with Sjögren's syndrome. Nat. Genet. 45, 1284–1292 (2013).
Cobb, B. L., Lessard, C. J., Harley, J. B. & Moser, K. L. Genes and Sjogren's syndrome. Rheum. Dis. Clin. North Am. 34, 847–868 (2008).
Nakano, M. et al. Clinical significance of anticentromere antibodies in patients with systemic lupus erythematosus. J. Rheumatol. 27, 1403–1407 (2000).
Gussin, H. A., Ignat, G. P., Varga, J. & Teodorescu, M. Anti-topoisomerase I (anti-Scl-70) antibodies in patients with systemic lupus erythematosus. Arthritis Rheum. 44, 376–383 (2001).
Ohta, A., Nagai, M., Nishina, M., Tomimitsu, H. & Kohsaka, H. Age at onset and gender distribution of systemic lupus erythematosus, polymyositis/dermatomyositis, and systemic sclerosis in Japan. Mod. Rheumatol. 23, 759–764 (2013).
Farh, K. K. et al. Genetic and epigenetic fine mapping of causal autoimmune disease variants. Nature 518, 337–343 (2015).
Sugiura-Ogasawara, M. et al. The first genome-wide association study identifying new susceptibility loci for obstetric antiphospholipid syndrome. J. Hum. Genet. 62, 831–838 (2017).
Marquez, A. et al. A combined large-scale meta-analysis identifies COG6 as a novel shared risk locus for rheumatoid arthritis and systemic lupus erythematosus. Ann. Rheum. Dis. 76, 286–294 (2017).
Lopez-Isac, E. et al. Brief report: IRF4 newly identified as a common susceptibility locus for systemic sclerosis and rheumatoid arthritis in a cross-disease meta-analysis of genome-wide association studies. Arthritis Rheumatol. 68, 2338–2344 (2016).
Martin, J. E. et al. A systemic sclerosis and systemic lupus erythematosus pan-meta-GWAS reveals new shared susceptibility loci. Hum. Mol. Genet. 22, 4021–4029 (2013).
Li, Y. R. et al. Meta-analysis of shared genetic architecture across ten pediatric autoimmune diseases. Nat. Med. 21, 1018–1027 (2015).
Milano, A. et al. Molecular subsets in the gene expression signatures of scleroderma skin. PLoS ONE 3, e2696 (2008).
Assassi, S. et al. Dissecting the heterogeneity of skin gene expression patterns in systemic sclerosis. Arthritis Rheumatol. 67, 3016–3026 (2015).
van Baarsen, L. G. et al. Synovial tissue heterogeneity in rheumatoid arthritis in relation to disease activity and biomarkers in peripheral blood. Arthritis Rheum. 62, 1602–1607 (2010).
van der Pouw Kraan, T. C. et al. Discovery of distinctive gene expression profiles in rheumatoid synovium using cDNA microarray technology: evidence for the existence of multiple pathways of tissue destruction and repair. Genes Immun. 4, 187–196 (2003).
Hall, J. C. et al. Molecular subsetting of interferon pathways in Sjögren's syndrome. Arthritis Rheumatol. 67, 2437–2446 (2015).
Peterson, K. S. et al. Characterization of heterogeneity in the molecular pathogenesis of lupus nephritis from transcriptional profiles of laser-captured glomeruli. J. Clin. Invest. 113, 1722–1733 (2004).
Nzeusseu Toukap, A. et al. Identification of distinct gene expression profiles in the synovium of patients with systemic lupus erythematosus. Arthritis Rheum. 56, 1579–1588 (2007).
Dey-Rao, R., Smith, J. R., Chow, S. & Sinha, A. A. Differential gene expression analysis in CCLE lesions provides new insights regarding the genetics basis of skin versus systemic disease. Genomics 104, 144–155 (2014).
Nakou, M. et al. Gene expression in systemic lupus erythematosus: bone marrow analysis differentiates active from inactive disease and reveals apoptosis and granulopoiesis signatures. Arthritis Rheum. 58, 3541–3549 (2008).
Gardner, H. et al. Gene profiling of scleroderma skin reveals robust signatures of disease that are imperfectly reflected in the transcript profiles of explanted fibroblasts. Arthritis Rheum. 54, 1961–1973 (2006).
Brkic, Z. et al. Prevalence of interferon type I signature in CD14 monocytes of patients with Sjögren's syndrome and association with disease activity and BAFF gene expression. Ann. Rheum. Dis. 72, 728–735 (2013).
Brkic, Z. et al. The interferon type I signature is present in systemic sclerosis before overt fibrosis and might contribute to its pathogenesis through high BAFF gene expression and high collagen synthesis. Ann. Rheum. Dis. 75, 1567–1573 (2016).
Liu, X. et al. Correlation of interferon-inducible chemokine plasma levels with disease severity in systemic sclerosis. Arthritis Rheum. 65, 226–235 (2013).
Rose, T. et al. SIGLEC1 is a biomarker of disease activity and indicates extraglandular manifestation in primary Sjögren's syndrome. RMD Open 2, e000292 (2016).
Chen, W., Cao, H., Lin, J., Olsen, N. & Zheng, S. G. Biomarkers for primary Sjögren's syndrome. Genom. Proteom. Bioinformat. 13, 219–223 (2015).
Bradley, S. J., Suarez-Fueyo, A., Moss, D. R., Kyttaris, V. C. & Tsokos, G. C. T. Cell transcriptomes describe patient subtypes in systemic lupus erythematosus. PLoS ONE 10, e0141171 (2015).
Banchereau, R. et al. Personalized immunomonitoring uncovers molecular networks that stratify lupus patients. Cell 165, 1548–1550 (2016).
Bennett, L. et al. Interferon and granulopoiesis signatures in systemic lupus erythematosus blood. J. Exp. Med. 197, 711–723 (2003).
Maas, K. et al. Cutting edge: molecular portrait of human autoimmune disease. J. Immunol. 169, 5–9 (2002).
Maas, K., Chen, H., Shyr, Y., Olsen, N. J. & Aune, T. Shared gene expression profiles in individuals with autoimmune disease and unaffected first-degree relatives of individuals with autoimmune disease. Hum. Mol. Genet. 14, 1305–1314 (2005).
Liu, Z., Maas, K. & Aune, T. M. Identification of gene expression signatures in autoimmune disease without the influence of familial resemblance. Hum. Mol. Genet. 15, 501–509 (2006).
Olsen, N. et al. A gene expression signature for recent onset rheumatoid arthritis in peripheral blood mono-nuclear cells. Ann. Rheum. Dis. 63, 1387–1392 (2004).
Assassi, S. et al. Systemic sclerosis and lupus: points in an interferon-mediated continuum. Arthritis Rheum. 62, 589–598 (2010).
Higgs, B. W. et al. Patients with systemic lupus erythematosus, myositis, rheumatoid arthritis and scleroderma share activation of a common type I interferon pathway. Ann. Rheum. Dis. 70, 2029–2036 (2011).
Toro-Dominguez, D., Carmona-Saez, P. & Alarcon-Riquelme, M. E. Shared signatures between rheumatoid arthritis, systemic lupus erythematosus and Sjögren's syndrome uncovered through gene expression meta-analysis. Arthritis Res. Ther. 16, 489 (2014).
Tuller, T., Atar, S., Ruppin, E., Gurevich, M. & Achiron, A. Common and specific signatures of gene expression and protein-protein interactions in autoimmune diseases. Genes Immun. 14, 67–82 (2013).
Bonasio, R., Tu, S. & Reinberg, D. Molecular signals of epigenetic states. Science 330, 612–616 (2010).
Jones, P. A. Functions of DNA methylation: islands, start sites, gene bodies and beyond. Nat. Rev. Genet. 13, 484–492 (2012).
Urdinguio, R. G., Sanchez-Mut, J. V. & Esteller, M. Epigenetic mechanisms in neurological diseases: genes, syndromes, and therapies. Lancet Neurol. 8, 1056–1072 (2009).
Vidal, E. et al. A DNA methylation map of human cancer at single base-pair resolution. Oncogene 36, 5648–5657 (2017).
Aslani, S. et al. Epigenetic alterations underlying auto-immune diseases. Autoimmunity 49, 69–83 (2016).
Coit, P. et al. Genome-wide DNA methylation study suggests epigenetic accessibility and transcriptional poising of interferon-regulated genes in naive CD4+ T cells from lupus patients. J. Autoimmun. 43, 78–84 (2013).
Richardson, B. et al. Evidence for impaired T cell DNA methylation in systemic lupus erythematosus and rheumatoid arthritis. Arthritis Rheum. 33, 1665–1673 (1990).
Corvetta, A., Della Bitta, R., Luchetti, M. M. & Pomponio, G. 5-Methylcytosine content of DNA in blood, synovial mononuclear cells and synovial tissue from patients affected by autoimmune rheumatic diseases. J. Chromatogr. 566, 481–491 (1991).
Altorok, N. et al. Genome-wide DNA methylation patterns in naive CD4+ T cells from patients with primary Sjogren's syndrome. Arthritis Rheumatol. 66, 731–739 (2014).
Lei, W. et al. Abnormal DNA methylation in CD4+ T cells from patients with systemic lupus erythematosus, systemic sclerosis, and dermatomyositis. Scand. J. Rheumatol 38, 369–374 (2009).
Absher, D. M. et al. Genome-wide DNA methylation analysis of systemic lupus erythematosus reveals persistent hypomethylation of interferon genes and compositional changes to CD4+ T-cell populations. PLoS Genet. 9, e1003678 (2013).
Altorok, N., Tsou, P. S., Coit, P., Khanna, D. & Sawalha, A. H. Genome-wide DNA methylation analysis in dermal fibroblasts from patients with diffuse and limited systemic sclerosis reveals common and subset-specific DNA methylation aberrancies. Ann. Rheum. Dis. 74, 1612–1620 (2015).
Javierre, B. M. et al. Changes in the pattern of DNA methylation associate with twin discordance in systemic lupus erythematosus. Genome Res. 20, 170–179 (2010).
Yeung, K. S. et al. Genome-wide DNA methylation analysis of Chinese patients with systemic lupus erythematosus identified hypomethylation in genes related to the type I interferon pathway. PoS ONE 12, e0169553 (2017).
Attig, L., Gabory, A. & Junien, C. Nutritional developmental epigenomics: immediate and long-lasting effects. Proc. Nutr. Soc. 69, 221–231.
Minami, Y. et al. Intakes of vitamin B6 and dietary fiber and clinical course of systemic lupus erythematosus: a prospective study of Japanese female patients. J. Epidemiol. 21, 246–254 (2011).
Lian, X. et al. DNA demethylation of CD40L in CD4+ T cells from women with systemic sclerosis: a possible explanation for female susceptibility. Arthritis Rheum. 64, 2338–2345 (2012).
Liao, J. et al. CD40L demethylation in CD4+ T cells from women with rheumatoid arthritis. Clin. Immunol. 145, 13–18 (2012).
Lu, Q. et al. Demethylation of CD40LG on the inactive X in T cells from women with lupus. J. Immunol. 179, 6352–6358 (2007).
Zhao, M. et al. IFI44L promoter methylation as a blood biomarker for systemic lupus erythematosus. Ann. Rheum. Dis. 75, 1998–2006 (2016).
Barrett, T. et al. NCBI GEO: archive for functional genomics data sets — update. Nucleic Acids Res. 41, D991–995 (2013).
Lappalainen, I. et al. The European Genome-phenome Archive of human data consented for biomedical research. Nat. Genet. 47, 692–695 (2015).
Woodcock, J. & LaVange, L. M. Master protocols to study multiple therapies, multiple diseases, or both. N. Engl. J. Med. 377, 62–70 (2017).
Ricano-Ponce, I. et al. Refined mapping of autoimmune disease associated genetic variants with gene expression suggests an important role for non-coding RNAs. J. Autoimmun. 68, 62–74 (2016).
Odhams, C. A. et al. Mapping eQTLs with RNA-seq reveals novel susceptibility genes, non-coding RNAs and alternative-splicing events in systemic lupus erythematosus. Hum. Mol. Genet. 26, 1003–1017 (2017).
Kasela, S. et al. Pathogenic implications for autoimmune mechanisms derived by comparative eQTL analysis of CD4+ versus CD8+ T cells. PLoS Genet. 13, e1006643 (2017).
Westra, H. J. et al. Systematic identification of trans eQTLs as putative drivers of known disease associations. Nat. Genet. 45, 1238–1243 (2013).
Walsh, A. M. et al. Integrative genomic deconvolution of rheumatoid arthritis GWAS loci into gene and cell type associations. Genome Biol. 17, 79 (2016).
Ai, R. et al. Joint-specific DNA methylation and transcriptome signatures in rheumatoid arthritis identify distinct pathogenic processes. Nat. Commun. 7, 11849 (2016).
Zhu, H. et al. Whole-genome transcription and DNA methylation analysis of peripheral blood mononuclear cells identified aberrant gene regulation pathways in systemic lupus erythematosus. Arthritis Res. Ther. 18, 162 (2016).
Kim-Howard, X. et al. ITGAM coding variant (rs1143679) influences the risk of renal disease, discoid rash and immunological manifestations in patients with systemic lupus erythematosus with European ancestry. Ann. Rheum. Dis. 69, 1329–1332 (2010).
Tasaki, S. et al. Multiomic disease signatures converge to cytotoxic CD8 T cells in primary Sjögren's syndrome. Ann. Rheum. Dis. 76, 1458–1466 (2017).
Hudson, M. et al. Novel insights into systemic autoimmune rheumatic diseases using shared molecular signatures and an integrative analysis. Epigenetics 12, 433–440 (2017).
Bailey, P. et al. Genomic analyses identify molecular subtypes of pancreatic cancer. Nature 531, 47–52 (2016).
Lombardo, M. V. et al. Unsupervised data-driven stratification of mentalizing heterogeneity in autism. Sci. Rep. 6, 35333 (2016).
Wang, B. et al. Similarity network fusion for aggregating data types on a genomic scale. Nat. Methods 11, 333–337 (2014).
Mo, Q. et al. Pattern discovery and cancer gene identification in integrated cancer genomic data. Proc. Natl Acad. Sci. USA 110, 4245–4250 (2013).
Kirk, P., Griffin, J. E., Savage, R. S., Ghahramani, Z. & Wild, D. L. Bayesian correlated clustering to integrate multiple datasets. Bioinformatics 28, 3290–3297 (2012).
Lock, E. F. & Dunson, D. B. Bayesian consensus clustering. Bioinformatics 29, 2610–2616 (2013).
Cho, D. Y. & Przytycka, T. M. Dissecting cancer heterogeneity with a probabilistic genotype-phenotype model. Nucleic Acids Res. 41, 8011–8020 (2013).
Yuan, Y., Savage, R. S. & Markowetz, F. Patient-specific data fusion defines prognostic cancer subtypes. PLoS Comput. Biol. 7, e1002227 (2011).
Menichetti, G., Remondini, D., Panzarasa, P., Mondragon, R. J. & Bianconi, G. Weighted multiplex networks. PLoS ONE 9, e97857 (2014).
Meng, C., Kuster, B., Culhane, A. C. & Gholami, A. M. A multivariate approach to the integration of multi-omics datasets. BMC Bioinformatics 15, 162 (2014).
Kim, S. et al. Integrative phenotyping framework (iPF): integrative clustering of multiple omics data identifies novel lung disease subphenotypes. BMC Genomics 16, 924 (2015).
He, S. et al. ICM: a web server for integrated clustering of multi-dimensional biomedical data. Nucleic Acids Res. 44, W154–159 (2016).
Speicher, N. K. & Pfeifer, N. Integrating different data types by regularized unsupervised multiple kernel learning with application to cancer subtype discovery. Bioinformatics 31, i268–275 (2015).
Jamin, C. et al. Multi-center harmonization of flow cytometers in the context of the European “PRECISESADS” project. Autoimmun. Rev. 15, 1038–1045 (2016).
Bennett, G. A. et al. Proposed diagnostic criteria for rheumatoid arthritis. Bull. Rheum. Dis. 7, 121–124 (1956).
Ropes, M. W. et al. 1958 Revision of diagnostic criteria for rheumatoid arthritis. Bull. Rheum. Dis. 9, 175–176 (1958).
Acknowledgements
The authors' work is supported in part by a EU/EFPIA/ Innovative Medicines Initiative Joint Undertaking PRECISESADS grant (no. 115565). G.B. is supported by financing from Genzyme/Sanofi in the context of the PRECISESADS project.
Author information
Authors and Affiliations
Contributions
All authors researched the data for the article, provided substantial contributions to discussions of its content, wrote the article and reviewed and/or edited the manuscript before submission.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests. G.B. and M.E.A-R are public employees of the Andalusian Government.
Related links
Supplementary information
Supplementary information S1 (figure)
Timeline of the classification criteria for systemic lupus erythematosus (blue), primary antiphospholipid syndrome (orange, APS), rheumatoid arthritis (RA, grey), mixed connective tissue disease (yellow), systemic sclerosis (SSc, green), and Sjögren syndrome (red), showing the criteria for each disease and the criteria that changed (included or excluded), as well as the time these criteria were published. (PDF 385 kb)
Glossary
- Next-generation sequencing
-
A group of massive parallel technologies that enable the sequencing of millions of DNA or RNA fragments in a short period of time. Also known as high-throughput sequencing.
- Type I interferon signature
-
An increased expression of type I interferon regulated or inducible genes, which have a major role in the activation of both the innate and adaptive immune systems.
- Gene expression modules
-
Sets of genes with highly correlated expressed patterns (co-expressed).
- Umbrella trial
-
A trial intended to study multiple targeted therapies in the context of a single disease or multiple diseases.
- Quantitative trait loci
-
Loci whose allelic variation is associated with the variation of a quantitative feature, such as gene expression (expression quantitative trait loci (eQTL)) or methylation (methylation QTL (mQTL)).
- Integrative clustering
-
A statistical method in which heterogeneous datasets are combined (data integration) and samples are grouped by similarity (clusters).
- Supervised analysis
-
Comparative analysis where prior biological and/or classification knowledge is required.
- Unsupervised analysis
-
In contrast to supervised analysis, no prior biological information and/or classification is required; the goal is to obtain new information based only on molecular information.
- Concatenation algorithms
-
A type of data integration algorithm where the different layers of information are analysed together before any data modelling or transformation is performed.
- Feature selection
-
A type of dimensionality reduction technique, which consists of selecting a subset of the most relevant features (for example, genes).
- Overfitting
-
When a statistical model fits random error or noise, instead of the real features of the datasets; overfitting can occur if the number of variables in the model is much higher than the number of observations.
- Dimensionality reduction
-
The process of reducing the number of variables (features) of a dataset; it can be divided in feature selection and feature extraction.
- Model-based algorithms
-
A type of data integration algorithm that fits data to a statistical model in order to infer or predict some features from the general population.
- Bayesian statistics
-
A mathematical method for calculating posterior probabilities (Bayesian probability) based on prior and current information.
- Parametric methods
-
A group of statistical techniques that assume that the datasets come from populations that follow a probability distribution defined by fixed parameters.
- Longitudinal datasets
-
Sets of repeated observations of the same variables at multiple points in time.
- Transformation-based algorithms
-
A type of data integration algorithm, which applies a mathematical function to each point of the datasets, transforming all the different types of data into a common feature space.
- Feature space
-
A set of values which summarize any kind of information (for example gene expression values for transcriptomic information); different data types can share a common feature space if transformed to have the same dimensions and the same range of values
- Network theory
-
The study of graphs as the representation of the relationship between the features of multiple observations; for example, networks could be defined as graphs with the nodes representing individuals and the lines between nodes (the edges) representing connections in gene expression profiles.
Rights and permissions
About this article
Cite this article
Barturen, G., Beretta, L., Cervera, R. et al. Moving towards a molecular taxonomy of autoimmune rheumatic diseases. Nat Rev Rheumatol 14, 75–93 (2018). https://doi.org/10.1038/nrrheum.2017.220
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrrheum.2017.220
This article is cited by
-
Targeting macrophage M1 polarization suppression through PCAF inhibition alleviates autoimmune arthritis via synergistic NF-κB and H3K9Ac blockade
Journal of Nanobiotechnology (2023)
-
The promise of precision medicine in rheumatology
Nature Medicine (2022)
-
A Summary on the Genetics of Systemic Lupus Erythematosus, Rheumatoid Arthritis, Systemic Sclerosis, and Sjögren’s Syndrome
Clinical Reviews in Allergy & Immunology (2022)
-
Genome-wide association study identifies Sjögren’s risk loci with functional implications in immune and glandular cells
Nature Communications (2022)
-
Lymphocyte subset clustering analysis in treatment-naive patients with systemic lupus erythematosus
Clinical Rheumatology (2021)