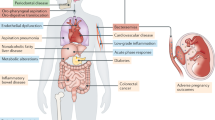
Abstract
Periodontitis is a chronic inflammatory disease that is characterized by loss of the periodontal ligament and alveolar bone, and is a major cause of tooth loss. Results from clinical and epidemiologic studies have suggested that periodontitis and tooth loss are more prevalent in individuals with rheumatoid arthritis (RA). However, the strength and temporality of the association are uncertain. Several biologically plausible causal and noncausal mechanisms might account for this association between periodontitis and RA. There is evidence to suggest that periodontitis could indeed be a causal factor in the initiation and maintenance of the autoimmune inflammatory response that occurs in RA. If proven, chronic periodontitis might represent an important modifiable risk factor for RA. In addition, patients with RA might show an increased risk of developing periodontitis and tooth loss through various mechanisms. Moreover, exposure to common genetic, environmental or behavioral factors might contribute to a noncausal association between both conditions.
Key Points
-
Periodontitis and tooth loss are highly prevalent in patients with rheumatoid arthritis (RA)
-
Both causal and noncausal mechanisms might be responsible for the association between periodontitis and RA
-
Periodontitis might be an important modifiable risk factor for RA
-
Mechanistic studies into the links between periodontitis and RA and the possible causal role of oral infections in the pathogenesis of RA are warranted
-
Well-designed epidemiologic studies are needed to establish the strength and temporality of the association between RA and periodontitis/tooth loss
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Morris, A. J. et al. The oral cleanliness and periodontal health of UK adults in 1998. Br. Dent. J. 191, 186–192 (2001).
Van der Weijden, G. A. & Timmerman, M. F. A systematic review on the clinical efficacy of subgingival debridement in the treatment of chronic periodontitis. J. Clin. Periodontol. 29 (Suppl. 3), 55–71 (2002).
Heitz-Mayfield, L. J. et al. A systematic review of the effect of surgical debridement vs non-surgical debridement for the treatment of chronic periodontitis. J. Clin. Periodontol. 29 (Suppl. 3), 92–102 (2002).
Phipps, K. R. & Stevens, V. J. Relative contribution of caries and periodontal disease in adult tooth loss for an HMO dental population. J. Public Health Dent. 55, 250–252 (1995).
Hung, H. C. et al. Tooth loss and dietary intake. J. Am. Dent. Assoc. 134, 1185–1192 (2003).
Mack, F. et al. The impact of tooth loss on general health related to quality of life among elderly Pomeranians: results from the study of health in Pomerania (SHIP-O). Int. J. Prosthodont. 18, 414–419 (2005).
Tonetti, M. S. et al. Treatment of periodontitis and endothelial function. N. Engl. J. Med. 356, 911–920 (2007).
Dietrich, T. et al. Age-dependent associations between chronic periodontitis/edentulism and risk of coronary heart disease. Circulation 117, 1668–1674 (2008).
Socransky, S. S. et al. Microbial complexes in subgingival plaque. J. Clin. Periodontol. 25, 134–144 (1998).
Page, R. C. & Kornman, K. S. The pathogenesis of human periodontitis: an introduction. Periodontol. 2000 14, 9–11 (1997).
Kantarci, A. et al. Neutrophil-mediated tissue injury in periodontal disease pathogenesis: findings from localized aggressive periodontitis. J. Periodontol. 74, 66–75 (2003).
Chapple, I. L. & Matthews, J. B. The role of reactive oxygen and antioxidant species in periodontal tissue destruction. Periodontol. 2000 43, 160–232 (2007).
Milward, M. R. et al. Differential activation of NF-κB and gene expression in oral epithelial cells by periodontal pathogens. Clin. Exp. Immunol. 148, 307–324 (2007).
Hujoel, P. P. et al. The dentogingival epithelial surface area revisited. J. Periodontal. Res. 36, 48–55 (2001).
Kinane, D. F. et al. Bacteraemia following periodontal procedures. J. Clin. Periodontol. 32, 708–713 (2005).
Matthews, J. B. et al. Hyperactivity and reactivity of peripheral blood neutrophils in chronic periodontitis. Clin. Exp. Immunol. 147, 255–264 (2007).
Figueredo, C. M. et al. Increased release of elastase from in vitro activated peripheral neutrophils in patients with adult periodontitis. J. Clin. Periodontol. 26, 206–211 (1999).
Loos, B. G. Systemic markers of inflammation in periodontitis. J. Periodontol. 76, 2106–2115 (2005).
D'Aiuto, F. et al. Acute effects of periodontal therapy on bio-markers of vascular health. J. Clin. Periodontol. 34, 124–129 (2007).
Wright, H. J. et al. Periodontitis associates with a type 1 IFN signature in peripheral blood neutrophils. J. Immunol. 181, 5775–5784 (2008).
Chapple, I. L. et al. Compromised GCF total antioxidant capacity in periodontitis: cause or effect? J. Clin. Periodontol. 34, 103–110 (2007).
Matthews, J. B. et al. Neutrophil hyper-responsiveness in periodontitis. J. Dent. Res. 86, 718–722 (2007).
Michalowicz, B. S. et al. Periodontal findings in adult twins. J. Periodontol. 62, 293–299 (1991).
Shapira, L. et al. The secretion of PGE2, IL-1 beta, IL-6, and TNF alpha by adherent mononuclear cells from early onset periodontitis patients. J. Periodontol. 65, 139–146 (1994).
Gemmell, E. et al. The role of T cells in periodontal disease: homeostasis and autoimmunity. Periodontol. 2000 43, 14–40 (2007).
Loos, B. G. et al. Identification of genetic risk factors for periodontitis and possible mechanisms of action. J. Clin. Periodontol. 32 (Suppl. 6), 159–179 (2005).
Gabriel, S. E. et al. Mortality in rheumatoid arthritis: have we made an impact in 4 decades? J. Rheumatol. 26, 2529–2533 (1999).
Laurell, L. et al. General oral status in adults with rheumatoid arthritis. Community Dent. Oral Epidemiol. 17, 230–233 (1989).
Tolo, K. & Jorkjend, L. Serum antibodies and loss of periodontal bone in patients with rheumatoid arthritis. J. Clin. Periodontol. 17, 288–291 (1990).
Albandar, J. M. Some predictors of radiographic alveolar bone height reduction over 6 years. J. Periodontal. Res. 25, 186–192 (1990).
Kasser, U. R. et al. Risk for periodontal disease in patients with longstanding rheumatoid arthritis. Arthritis Rheum. 40, 2248–2251 (1997).
Mercado, F. et al. Is there a relationship between rheumatoid arthritis and periodontal disease? J. Clin. Periodontol. 27, 267–272 (2000).
Mercado, F. B. et al. Relationship between rheumatoid arthritis and periodontitis. J. Periodontol. 72, 779–787 (2001).
Georgiou, T. O. et al. Prevalence of systemic diseases in Brisbane general and periodontal practice patients. Aust. Dent. J. 49, 177–184 (2004).
Helenius, M. & Leirisalo-Repo, M. Oral complications in patients with rheumatoid arthritis [Finnish]. Duodecim. 121, 2333–2337 (2005).
Al-Shammari, K. F. et al. Risk indicators for tooth loss due to periodontal disease. J. Periodontol. 76, 1910–1918 (2005).
Abou-Raya, A. et al. Periodontal disease and rheumatoid arthritis: is there a link? Scand. J. Rheumatol. 34, 408–410 (2005).
Marotte, H. et al. The association between periodontal disease and joint destruction in rheumatoid arthritis extends the link between the HLA-DR shared epitope and severity of bone destruction. Ann. Rheum. Dis. 65, 905–909 (2006).
Havemose-Poulsen, A. et al. Periodontal and hematological characteristics associated with aggressive periodontitis, juvenile idiopathic arthritis, and rheumatoid arthritis. J. Periodontol. 77, 280–288 (2006).
Abou-Raya, S. et al. Rheumatoid arthritis, periodontal disease and coronary artery disease. Clin. Rheumatol. 27, 421–427 (2008).
Pischon, N. et al. Association among rheumatoid arthritis, oral hygiene, and periodontitis. J. Periodontol. 79, 979–986 (2008).
de Pablo, P. et al. Association of periodontal disease and tooth loss with rheumatoid arthritis in the US population. J. Rheumatol. 35, 70–76 (2008).
Sjostrom, L. et al. Periodontal conditions in adults with rheumatoid arthritis. Community Dent. Oral Epidemiol. 17, 234–236 (1989).
Ishi Ede, P. et al. Periodontal condition in patients with rheumatoid arthritis. Braz. Oral Res. 22, 72–77 (2008).
Tonetti, M. S. & Claffey, N. Advances in the progression of periodontitis and proposal of definitions of a periodontitis case and disease progression for use in risk factor research. Group C consensus report of the 5th European Workshop in Periodontology. J. Clin. Periodontol. 32 (Suppl. 6), 210–213 (2005).
Page, R. C. & Eke, P. Case definitions for use in population based surveillance of periodontitis. J. Periodontol. 78, 1387–1399 (2007).
Mikuls, T. R. et al. Antibody responses to Porphyromonas gingivalis (P. gingivalis) in subjects with rheumatoid arthritis and periodontitis. Int. Immunopharmacol. 9, 38–42 (2008).
Blicher, B. et al. Validation of self-reported periodontal disease: a systematic review. J. Dent. Res. 84, 881–890 (2005).
Bingham, C. et al. Periodontal disease symptoms are independently associated with rheumatoid arthritis disease activity [abstract #1937]. Arthritis Rheum. 58, S876 (2008).
Mikuls, T. R. et al. Coffee, tea, and caffeine consumption and risk of rheumatoid arthritis: results from the Iowa Women's Health Study. Arthritis Rheum. 46, 83–91 (2002).
Karlson, E. W. et al. Coffee consumption and risk of rheumatoid arthritis. Arthritis Rheum. 48, 3055–3060 (2003).
Rasch, E. K. et al. Prevalence of rheumatoid arthritis in persons 60 years of age and older in the United States: effect of different methods of case classification. Arthritis Rheum. 48, 917–926 (2003).
Mutlu, S. et al. Gingival and periodontal health in systemic lupus erythematosus. Community Dent. Oral Epidemiol. 21, 158–161 (1993).
Novo, E. et al. Periodontitis and anti-neutrophil cytoplasmic antibodies in systemic lupus erythematosus and rheumatoid arthritis: a comparative study. J. Periodontol. 70, 185–188 (1999).
Miranda, L. A. et al. Periodontal conditions in patients with juvenile idiopathic arthritis. J. Clin. Periodontol. 30, 969–974 (2003).
Miranda, L. A. et al. Changes in periodontal and rheumatological conditions after 2 years in patients with juvenile idiopathic arthritis. J. Periodontol. 77, 1695–1700 (2006).
Reichert, S. et al. Is there a relationship between juvenile idiopathic arthritis and periodontitis? J. Clin. Periodontol. 33, 317–323 (2006).
Schiodt, M. et al. Periodontal disease in primary Sjögren's syndrome. Oral Dis. 7, 106–108 (2001).
Kuru, B. et al. Clinical and microbiological studies of periodontal disease in Sjögren syndrome patients. J. Clin. Periodontol. 29, 92–102 (2002).
Jorkjend, L. et al. Periodontitis, caries and salivary factors in Sjögren's syndrome patients compared to sex- and age-matched controls. J. Oral Rehabil. 30, 369–378 (2003).
Soto-Rojas, A. E. et al. Oral manifestations in patients with Sjögren's syndrome. J. Rheumatol. 25, 906–910 (1998).
Christensen, L. B. et al. Dental caries and dental health behavior of patients with primary Sjögren syndrome. Acta Odontol. Scand. 59, 116–120 (2001).
Moen, K. et al. Immunoglobulin G and A antibody responses to Bacteroides forsythus and Prevotella intermedia in sera and synovial fluids of arthritis patients. Clin. Diagn. Lab. Immunol. 10, 1043–1050 (2003).
Moen, K. et al. Synovial inflammation in active rheumatoid arthritis and psoriatic arthritis facilitates trapping of a variety of oral bacterial DNAs. Clin. Exp. Rheumatol. 24, 656–663 (2006).
Ogrendik, M. et al. Serum antibodies to oral anaerobic bacteria in patients with rheumatoid arthritis. MedGenMed. 7, 2 (2005).
Travis, J. et al. Porphyromonas gingivalis proteinases as virulence factors in the development of periodontitis. J. Periodontal. Res. 32, 120–125 (1997).
McGraw, W. T. et al. Purification, characterization, and sequence analysis of a potential virulence factor from Porphyromonas gingivalis, peptidylarginine deiminase. Infect. Immun. 67, 3248–3256 (1999).
Rosenstein, E. D. et al. Hypothesis: the humoral immune response to oral bacteria provides a stimulus for the development of rheumatoid arthritis. Inflammation 28, 311–318 (2004).
Kinloch, A. et al. Synovial fluid is a site of citrullination of autoantigens in inflammatory arthritis. Arthritis Rheum. 58, 2287–2295 (2008).
Lundberg, K. et al. Antibodies to citrullinated alpha-enolase peptide 1 are specific for rheumatoid arthritis and cross-react with bacterial enolase. Arthritis Rheum. 58, 3009–3019 (2008).
Ribeiro, J. et al. Periodontal infection as a possible severity factor for rheumatoid arthritis. J. Clin. Periodontol. 32, 412–416 (2005).
Al-Katma, M. K. et al. Control of periodontal infection reduces the severity of active rheumatoid arthritis. J. Clin. Rheumatol. 13, 134–137 (2007).
Hirsch, H. Z. et al. Local production of IgA- and IgM-rheumatoid factors in adult periodontal disease. J. Clin. Immunol. 9, 273–278 (1989).
Thé, J. & Ebersole, J. L. Rheumatoid factor from periodontitis patients cross-reacts with epitopes on oral bacteria. Oral Dis. 2, 253–262 (1996).
Seven, A. et al. Lipid, protein, DNA oxidation and antioxidant status in rheumatoid arthritis. Clin. Biochem. 41, 538–543 (2008).
Rossi, A. G. et al. Cyclin-dependent kinase inhibitors enhance the resolution of inflammation by promoting inflammatory cell apoptosis. Nat. Med. 12, 1056–1064 (2006).
Ramamurthy, N. S. et al. Experimental arthritis in rats induces biomarkers of periodontitis which are ameliorated by gene therapy with tissue inhibitor of matrix metalloproteinases. J. Periodontol. 76, 229–233 (2005).
Deodhar, A. A. & Woolf, A. D. Bone mass measurement and bone metabolism in rheumatoid arthritis: a review. Br. J. Rheumatol. 35, 309–322 (1996).
Walsh, N. C. et al. Rheumatic diseases: the effects of inflammation on bone. Immunol. Rev. 208, 228–251 (2005).
Wactawski-Wende, J. Periodontal diseases and osteoporosis: association and mechanisms. Ann. Periodontol. 6, 197–208 (2001).
Krall, E. A. Osteoporosis and the risk of tooth loss. Clin. Calcium 16, 287–290 (2006).
Howell, T. H. Blocking periodontal disease progression with anti-inflammatory agents. J. Periodontol. 64, 828–833 (1993).
Howell, T. H. & Williams, R. C. Nonsteroidal anti-inflammatory drugs as inhibitors of periodontal disease progression. Crit. Rev. Oral Biol. Med. 4, 177–196 (1993).
Assuma, R. et al. IL-1 and TNF antagonists inhibit the inflammatory response and bone loss in experimental periodontitis. J. Immunol. 160, 403–409 (1998).
Di Paola, R. et al. Effects of etanercept, a tumour necrosis factor-alpha antagonist, in an experimental model of periodontitis in rats. Br. J. Pharmacol. 150, 286–297 (2007).
Pers, J. O. et al. Anti-TNF-alpha immunotherapy is associated with increased gingival inflammation without clinical attachment loss in subjects with rheumatoid arthritis. J. Periodontol. 79, 1645–1651 (2008).
Arneberg, P. et al. Remaining teeth, oral dryness and dental health habits in middle-aged Norwegian rheumatoid arthritis patients. Community Dent. Oral Epidemiol. 20, 292–296 (1992).
Malmstrom, M. & Calonius, P. E. Teeth loss and the inflammation of teeth-supporting tissues in rheumatoid disease. Scand. J. Rheumatol. 4, 49–55 (1975).
Pokrajac-Zirojevic, V. et al. Arthritis and use of dental services: a population based study. Aust. Dent. J. 47, 208–213 (2002).
Pincus, T. Patient questionnaires and formal education as more significant prognostic markers than radiographs or laboratory tests for rheumatoid arthritis mortality—limitations of a biomedical model to predict long-term outcomes. Bull. NYU Hosp. Jt Dis. 65 (Suppl. 1), S29–S36 (2007).
Borrell, L. N. & Crawford, N. D. Social disparities in periodontitis among United States adults 1999–2004. Community Dent. Oral Epidemiol. 36, 383–391 (2008).
Klareskog, L. et al. Smoking as a trigger for inflammatory rheumatic diseases. Curr. Opin. Rheumatol. 19, 49–54 (2007).
Bergstrom, J. Periodontitis and smoking: an evidence-based appraisal. J. Evid. Based Dent. Pract. 6, 33–41 (2006).
Dietrich, T. et al. Tobacco use and incidence of tooth loss among US male health professionals. J. Dent. Res. 86, 373–377 (2007).
Smolen, J. S. & Steiner, G. Therapeutic strategies for rheumatoid arthritis. Nat. Rev. Drug Discov. 2, 473–488 (2003).
McInnes, I. B. & Schett, G. Cytokines in the pathogenesis of rheumatoid arthritis. Nat. Rev. Immunol. 7, 429–442 (2007).
van der Helm-van Mil, A. H. & Huizinga, T. W. Advances in the genetics of rheumatoid arthritis point to subclassification into distinct disease subsets. Arthritis Res. Ther. 10, 205 (2008).
Ohm, K. et al. Measurement of eight prostaglandins in human gingival and periodontal disease using high pressure liquid chromatography and radioimmunoassay. J. Periodontal Res. 19, 501–511 (1984).
Graves, D. Cytokines that promote periodontal tissue destruction. J. Periodontol. 79, 1585–1591 (2008).
Sorsa, T. et al. Matrix metalloproteinases: contribution to pathogenesis, diagnosis and treatment of periodontal inflammation. Ann. Med. 38, 306–321 (2006).
Havemose-Poulsen, A. et al. Cytokine profiles in peripheral blood and whole blood cell cultures associated with aggressive periodontitis, juvenile idiopathic arthritis, and rheumatoid arthritis. J. Periodontol. 76, 2276–2285 (2005).
Nilsson, M. & Kopp, S. Gingivitis and periodontitis are related to repeated high levels of circulating tumor necrosis factor-alpha in patients with rheumatoid arthritis. J. Periodontol. 79, 1689–1696 (2008).
Sorensen, L. K. et al. Blood cell gene expression profiling in subjects with aggressive periodontitis and chronic arthritis. J. Periodontol. 79, 477–485 (2008).
Lee, Y. H. et al. Association between interleukin 1 polymorphisms and rheumatoid arthritis susceptibility: a metaanalysis. J. Rheumatol. (2008).
Lee, Y. H. et al. Associations between FCGR3A polymorphisms and rheumatoid arthritis susceptibility: a metaanalysis. J. Rheumatol. 35, 2129–2135 (2008).
Havemose-Poulsen, A. et al. Polymorphisms within the IL-1 gene cluster: effects on cytokine profiles in peripheral blood and whole blood cell cultures of patients with aggressive periodontitis, juvenile idiopathic arthritis, and rheumatoid arthritis. J. Periodontol. 78, 475–492 (2007).
Kobayashi, T. et al. The interleukin-1 and Fcγ receptor gene polymorphisms in Japanese patients with rheumatoid arthritis and periodontitis. J. Periodontol. 78, 2311–2318 (2007).
Fiebig, A. et al. Polymorphisms in the interleukin-1 (IL1) gene cluster are not associated with aggressive periodontitis in a large Caucasian population. Genomics 92, 309–315 (2008).
Andonopoulos, A. P. et al. Secondary Sjögren's syndrome in rheumatoid arthritis. J. Rheumatol. 14, 1098–1103 (1987).
Acknowledgements
This work was supported by the DEPEND consortium of the Birmingham Black Country (BBC) Comprehensive Local Research Network (CLRN).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
de Pablo, P., Chapple, I., Buckley, C. et al. Periodontitis in systemic rheumatic diseases. Nat Rev Rheumatol 5, 218–224 (2009). https://doi.org/10.1038/nrrheum.2009.28
Issue Date:
DOI: https://doi.org/10.1038/nrrheum.2009.28
This article is cited by
-
Gingival bleeding as a predictor of handgrip strength—an observational study and a pilot randomized clinical trial
Clinical Oral Investigations (2024)
-
Effect of periodontitis induced by Fusobacterium nucleatum on the microbiota of the gut and surrounding organs
Odontology (2024)
-
The Oral-Vascular-Pulmonary Infection Route: a Pathogenic Mechanism Linking Oral Health Status to Acute and Post-Acute COVID-19
Current Oral Health Reports (2023)
-
Associations between inflammation-related LL-37 with subgingival microbial dysbiosis in rheumatoid arthritis patients
Clinical Oral Investigations (2022)
-
Interplay of Microbiota and Citrullination in the Immunopathogenesis of Rheumatoid Arthritis
Probiotics and Antimicrobial Proteins (2022)