Abstract
Approval of the anti-CD20 antibody rituximab for the treatment of moderate-to-severe rheumatoid arthritis in patients who fail to respond to anti-tumor-necrosis-factor agents has raised interest in B-cell-directed therapy for this disease. A number of direct and indirect modalities with distinct mechanisms of action are being investigated, including anti-CD20 and anti-CD22 therapies, and new approaches for blocking members of the tumor necrosis factor cytokine family including B cell activating factor (BAFF) and a proliferation ligand (APRIL), which are at late stages of clinical development. Clinical experience is most extensive with rituximab, and suggests that targeting 'autoimmune' memory B cells is a feasible approach for treating autoimmune disease. Although anti-CD20 therapy has only been approved for rheumatoid arthritis thus far, data suggest this approach could be valid for other autoimmune diseases, including systemic lupus erythematosus, Sjögren's syndrome, vasculitides, autoimmune cytopenias, and neurologic and dermatologic autoimmune diseases. Additional studies of direct and indirect B-cell-directed treatments are needed before we can draw conclusions as to the value of this approach in patients with various autoimmune diseases and whether more precisely defined techniques than these are required to target the complex humoral system effectively.
Key Points
-
Approval of rituximab as a B-cell-directed therapy for rheumatoid arthritis has raised interest in B-cell-directed therapies for other autoimmune diseases
-
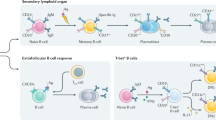
Studies of B-cell-depletion approaches have shown that peripheral immunoglobulins are probably generated by plasma cells with different lifespans
-
Short-lived plasma cells produce an important array of activity-related autoantibodies, whereas long-lived plasma cells are mainly responsible for protective immunoglobulin titers
-
Approaches that target adaptive immunity, such as B-cell depletion with anti-CD20 antibodies, involve a longer time to clinical response than do approaches that preferentially target innate immunity
-
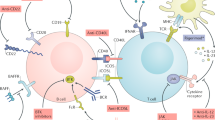
Anti-CD20 therapy selectively depletes reactive memory B cells that are otherwise refractory to conventional therapy
-
Despite reports of rituximab being effective in patients with treatment-refractory autoimmune diseases other than rheumatoid arthritis, randomized controlled clinical trials are needed to assess the value of this approach
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Dorner, T. & Burmester, G. R. The role of B cells in rheumatoid arthritis: mechanisms and therapeutic targets. Curr. Opin. Rheumatol. 15, 246–252 (2003).
Manz, R. A., Thiel, A. & Radbruch, A. Lifetime of plasma cells in the bone marrow. Nature 388, 133–134 (1997).
Amanna, I. J., Carlson, N. E. & Slifka, M. K. Duration of humoral immunity to common viral and vaccine antigens. N. Engl. J. Med. 357, 1903–1915 (2007).
Radbruch, A. et al. Competence and competition: the challenge of becoming a long-lived plasma cell. Nat. Rev. Immunol. 6, 741–750 (2006).
Edwards, J. C. W. et al. Efficacy of B-cell-targeted therapy with rituximab in patients with rheumatoid arthritis. N. Engl. J. Med. 350, 2572–2581 (2004).
Mariette, X. Therapeutic potential for B-cell modulation in Sjögren's syndrome. Rheum. Dis. Clin. North Am. 34, 1025–1033 (2008).
Tarlinton, D. B-cell memory: are subsets necessary? Nat. Rev. Immunol. 6, 785–790 (2006).
Bendelac, A., Bonneville, M. & Kearney, J. F. Autoreactivity by design: innate B and T lymphocytes. Nat. Rev. Immunol. 1, 177–186 (2001).
Pillai, S., Cariappa, A. & Moran, S. T. Marginal zone B cells. Annu. Rev. Immunol. 23, 161–196 (2005).
Tarlinton, D., Radbruch, A., Hiepe, F. & Dorner, T. Plasma cell differentiation and survival. Curr. Opin. Immunol. 20, 162–169 (2008).
Binard, A. et al. Does BAFF dysregulation play a major role in the pathogenesis of systemic lupus erythematosus? J. Autoimmun. 30, 63–67 (2008).
Lindh, E. et al. AIRE regulates T-cell-independent B-cell responses through BAFF. Proc. Natl Acad. Sci. USA 105, 18466–18471 (2008).
Cohen, S. B. et al. Rituximab for rheumatoid arthritis refractory to anti-tumor necrosis factor therapy: results of a multicenter, randomized, double-blind, placebo-controlled, phase III trial evaluating primary efficacy and safety at twenty-four weeks. Arthritis Rheum. 54, 2793–2806 (2006).
Emery, P. et al. The efficacy and safety of rituximab in patients with active rheumatoid arthritis despite methotrexate treatment—results of a phase IIb randomized, double-blind, placebo-controlled, dose-ranging trial. Arthritis Rheum. 54, 1390–1400 (2006).
Cambridge, G. et al. Serological changes following B cell depletion therapy in systemic lupus erythematosus: relationship with BLyS. Arthritis Rheum. 50, S645–S646 (2004).
Walsh, C. A. E., Fearon, U., FitzGerald, O., Veale, D. J. & Bresnihan, B. Decreased CD20 expression in rheumatoid arthritis synovium following 8 weeks of rituximab therapy. Clin. Exp. Rheumatol. 26, 656–658 (2008).
Thurlings, R. M. et al. Synovial tissue response to rituximab: mechanism of action and identification of biomarkers of response. Ann. Rheum. Dis. 67, 917–925 (2008).
Kavanaugh, A. et al. Assessment of rituximab's immunomodulatory synovial effects (ARISE trial). 1: clinical and synovial biomarker results. Ann. Rheum. Dis. 67, 402–408 (2008).
Vos, K. et al. Early effects of rituximab on the synovial cell infiltrate in patients with rheumatoid arthritis. Arthritis Rheum. 56, 772–778 (2007).
Cambridge, G. et al. Serologic changes following B lymphocyte depletion therapy for rheumatoid arthritis. Arthritis Rheum. 48, 2146–2154 (2003).
Keogh, K. A., Ytterberg, S. R., Fervenza, F. C. & Specks, U. Rituximab for remission induction in severe ANCA-associated vasculitis: report of a prospective open-label pilot trial in 10 patients [abstract]. Arthritis Rheum. 50, S270 (2004).
Ferraro, A. J., Day, C. J., Drayson, M. T. & Savage, C. O. Effective therapeutic use of rituximab in refractory Wegener's granulomatosis. Nephrol. Dial. Transplant. 20, 622–625 (2005).
Cambridge, G. et al. B cell depletion therapy in systemic lupus erythematosus: relationships among serum B lymphocyte stimulator levels, autoantibody profile and clinical response. Ann. Rheum. Dis. 67, 1011–1016 (2008).
Dorner, T. & Radbruch, A. Antibodies and B cell memory in viral immunity. Immunity 27, 384–392 (2007).
Vieira, P. & Rajewsky, K. Persistence of memory B-cells in mice deprived of T-cell help. Int. Immunol. 2, 487–494 (1990).
Odendahl, M. et al. Disturbed peripheral B lymphocyte homeostasis in systemic lupus erythematosus. J. Immunol. 165, 5970–5979 (2000).
Roll, P., Dorner, T. & Tony, H. P. Anti-CD20 therapy in patients with rheumatoid arthritis—predictors of response and B cell subset regeneration after repeated treatment. Arthritis Rheum. 58, 1566–1575 (2008).
Anolik, J. H. et al. Delayed memory B cell recovery in peripheral blood and lymphoid tissue in systemic lupus erythematosus after B cell depletion therapy. Arthritis Rheum. 56, 3044–3056 (2007).
Weller, S. et al. Human blood IgM “memory” B cells are circulating splenic marginal zone B cells harboring a prediversified immunoglobulin repertoire. Blood 104, 3647–3654 (2004).
Kruetzmann, S. et al. Human immunoglobulin M memory B cells controlling Streptococcus pneumoniae infections are generated in the spleen. J. Exp. Med. 197, 939–945 (2003).
Martinez-Gamboa, L. et al. Role of the spleen in peripheral memory B-cell homeostasis in patients with autoimmune thrombocytopenia purpura. Clin. Immunol. 130, 199–212 (2009).
Tsuiji, M. et al. A checkpoint for autoreactivity in human IgM+ memory B cell development. J. Exp. Med. 203, 393–400 (2006).
Tak, P. P. et al. Atacicept in patients with rheumatoid arthritis. Arthritis Rheum. 58, 61–72 (2008).
Weinblatt, M. et al. Safety of the selective costimulation modulator abatacept in rheumatoid arthritis patients receiving background biologic and nonbiologic disease-modifying antirheumatic drugs—a one-year randomized, placebo-controlled study. Arthritis Rheum. 54, 2807–2816 (2006).
Dorner, T. & Burmester, G. R. New approaches of B-cell-directed therapy: beyond rituximab. Curr. Opin. Rheumatol. 20, 263–268 (2008).
Eisenberg, R. Targeting B cells in systemic lupus erythematosus: not just déjà vu all over again. Arthritis Res. Ther. 8, 108 (2006).
Looney, R. J., Anolik, J. & Sanz, I. B cells as therapeutic targets for rheumatic diseases. Curr. Opin. Rheumatol. 16, 180–185 (2004).
Edwards, J. C. W. & Cambridge, G. Sustained improvement in rheumatoid arthritis following a protocol designed to deplete B lymphocytes. Rheumatology 40, 205–211 (2001).
Leandro, M. J. et al. Treatment of refractory lupus nephritis with B lymphocyte depletion [abstract]. Arthritis Rheum. 48, S378 (2003).
Coles, A. J. et al. Alemtuzumab vs. interferon β1a in early multiple sclerosis. N. Engl. J. Med. 359, 1786–1801 (2008).
Genovese, M. C. et al. Safety and clinical activity of ocrelizumab (a humanized antibody targeting CD20+ B cells) in combination with methotrexate (MTX) in moderate-severe rheumatoid arthritis (RA) patients (pts) (Ph I/II ACTION study). Arthritis Rheum. 54, S66–S67 (2006).
Silverman, G. J. & Boyle, D. L. Understanding the mechanistic basis in rheumatoid arthritis for clinical response to anti-CD20 therapy: the B-cell roadblock hypothesis. Immunol. Rev. 223, 175–185 (2008).
Sfikakis, P. P. et al. Remission of proliferative lupus nephritis following anti-B cell therapy is preceded by downregulation of the T cell costimulatory molecule CD40-ligand [abstract]. Arthritis Rheum. 50, S227 (2004).
Stasi, R. et al. Response to B-cell-depleting therapy with rituximab reverts the abnormalities of T-cell subsets in patients with idiopathic thrombocytopenic purpura. Blood 110, 2924–2930 (2007).
Taylor, R. P. & Lindorfer, M. A. Immunotherapeutic mechanisms of anti-CD20 monoclonal antibodies. Curr. Opin. Immunol. 20, 444–449 (2008).
Stasi, R., Stipa, E., Forte, V., Meo, P. & Amadori, S. To the editor: variable patterns of response to rituximab treatment in adults with chronic idiopathic thrombocytopenic purpura. Blood 99, 3872–3873 (2002).
Martin, F. & Chan, A. C. Pathogenic roles of B cells in human autoimmunity: insights from the clinic. Immunity 20, 517–527 (2004).
Hauser, S. L. et al. B-cell depletion with rituximab in relapsing-remitting multiple sclerosis. N. Engl. J. Med. 358, 676–688 (2008).
Dass, S. et al. Reduction of fatigue in Sjögren syndrome with rituximab: results of a randomised, double-blind, placebo-controlled pilot study. Ann. Rheum. Dis. 67, 1541–1544 (2008).
Leandro, M. J., Edwards, J. C., Cambridge, G., Ehrenstein, M. R. & Isenberg, D. A. An open study of B lymphocyte depletion in systemic lupus erythematosus. Arthritis Rheum. 46, 2673–2677 (2002).
Keogh, K. A., Wylam, M. E., Fervenza, F. C. & Specks, U. Rituximab—a novel mechanism-based therapy for refractory ANCA associated vasculitis. J. Am. Soc. Nephrol. 14, 38A–39A (2003).
Merrill, J. T. et al. Efficacy and safety of rituximab in patients with moderately to severely active systemic lupus erythematosus (SLE): results from the randomized, double-blind phase II/III study EXPLORER. Arthritis Rheum. 58, 4029–4030 (2008).
ClinicalTrials.gov: a service of the US National Institutes of Health. A study to evaluate the efficacy and safety of rituximab in subjects with ISN/RPS Class III or IV lupus nephritis (LUNAR), [online]
Ng, K. P. et al. B cell depletion therapy in systemic lupus erythematosus: long term follow-up and predictors of response. Ann. Rheum. Dis. 66, 1259–1262 (2007).
Mease, P. J. B cell-targeted therapy in autoimmune disease: rationale, mechanisms, and clinical application. J. Rheumatol. 35, 1245–1255 (2008).
Wiendl, H. & Hohlfeld, R. Immunopathogenesis and therapy of inflammatory myopathies. Akt. Neurol. 35, 185–191 (2008).
Salama, A. D. & Pusey, C. D. Drug insight: rituximab in renal disease and transplantation. Nat. Clin. Pract. Nephrol. 2, 221–230 (2006).
Zaja, F. et al. Rituximab for the treatment of type II mixed cryoglobulinemia. Arthritis Rheum. 46, 2252–2254 (2002).
Zand, M. S. Therapeutic antibody agents for B-cell immunomodulation in renal transplantation. Transplantation 84, S11–S19 (2007).
Wingerchuk, D. M. & Weinshenker, B. C. Neuromyelitis optica. Curr. Treat. Opt. Neurol. 10, 55–66 (2008).
Pranzatelli, M. R. et al. Rituximab (anti-CD20) adjunctive therapy for opsoclonus–myoclonus syndrome. J. Pediatr. Hematol. Oncol. 28, 585–593 (2006).
Sieb, J. P. Myasthenia gravis: emerging new therapy options. Curr. Opin. Pharmacol. 5, 303–307 (2005).
Sorce, M., Arico, M. & Bongiorno, M. R. Rituximab in refractory pemphigus vulgaris. Dermatol. Ther. 21, S6–S9 (2008).
Wallet-Faber, N. et al. Epidermolysis bullosa acquisita following bullous pemphigoid, successfully treated with the anti-CD20 monoclonal antibody rituximab. Dermatology 215, 252–255 (2007).
Garvey, B. Rituximab in the treatment of autoimmune haematological disorders. Br. J. Haematol. 141, 149–169 (2008).
Wache, A., Gil, L. & Komarnicki, M. Rituximab in haematology and oncology in 10 years of experience. Wspolczesna Onkol. Contemp. Oncol. 12, 173–178 (2008).
Verlinden, A. et al. Treatment of mixed type autoimmune haemolytic anaemia with autologous peripheral blood stem cell transplantation resulting in disappearance of the warm-type antibody and clinical remission of haemolysis. Acta Clin. Belg. 62, 380 (2007).
Schollkopf, C. et al. Rituximab in chronic cold agglutinin disease: a prospective study of 20 patients. Leuk. Lymphoma 47, 253–260 (2006).
Aggarwal, A. et al. Rituximab for autoimmune haemophilia: a proposed treatment algorithm. Haemophilia 11, 13–19 (2005).
Sperr, W. R., Lechner, K. & Pabinger, I. Rituximab for the treatment of acquired antibodies to factor VIII. Haematologica 92, 66–71 (2007).
Stasi, R., Pagano, A., Stipa, E. & Amadori, S. Rituximab chimeric anti-CD20 monoclonal antibody treatment for adults with chronic idiopathic thrombocytopenic purpura. Blood 98, 952–957 (2001).
Dorner, T. et al. Initial clinical trial of epratuzumab (humanized anti-CD22 antibody) for immunotherapy of systemic lupus erythematosus. Arthritis Res. Ther. 8, (2006).
Steinfeld, S. D. et al. Initial clinical study of immunotherapy in primary Sjögren's syndrome with humanized anti-CD22 antibody epratuzumab. Ann. Rheum. Dis. 64, 311 (2005).
Jacobi, A. M. et al. Differential effects of epratuzumab on peripheral blood B cells of patients with systemic lupus erythematosus versus normal controls. Ann. Rheum. Dis. 67, 450–457 (2008).
[No authors listed]. Belimumab: anti-BLyS human monoclonal antibody, anti-BLyS monoclonal antibody, BmAb, human monoclonal antibody to B-lymphocyte stimulator. Drugs RD 9, 197–202 (2008).
Dall'Era, M. et al. Reduced B lymphocyte and immunoglobulin levels after atacicept treatment in patients with systemic lupus erythematosus. Arthritis Rheum. 56, 4142–4150 (2007).
Dall'Era, M. et al. Trial of atacicept in patients with systemic lupus erythematosus (SLE). Arthritis Rheum. 54, 4042–4043 (2006).
Tak, P. P. et al. Atacicept in patients with rheumatoid arthritis. Arthritis Rheum. 58, 61–72 (2008).
Neubert, K. et al. The proteasome inhibitor bortezomib depletes plasma cells and protects mice with lupus-like disease from nephritis. Nat. Med. 14, 748–755 (2008).
Dorner, T. & Lipsky, P. E. B-cell targeting: a novel approach to immune intervention today and tomorrow. Exp. Opin. Biol. Ther. 7, 1287–1299 (2007).
Alexander, T. et al. Depletion of the autoreactive immunological memory followed by autologous haemopoietic stem cell transplantation in patients with refractory SLE induces long-term remissions through de novo generation of a juvenile and tolerant immune system. Bone Marrow Transplant. 41, S2–S3 (2008).
Kavanaugh, A. F. B cell targeted therapies: safety considerations. J. Rheumatol. 33, 18–23 (2006).
US Department of Health & Human Services Postmarketing Reviews—Volume 1, Number 1, Fall 2007. Rituximab (marketed as Rituxan®): progressive multifocal leukoencephalopathy (PML), [online]
Calabrese, L. H., Molloy, E. S., Huang, D. R. & Ransohoff, R. M. Progressive multifocal leukoencephalopathy in rheumatic diseases—evolving clinical and pathologic patterns of disease. Arthritis Rheum. 56, 2116–2128 (2007).
Acknowledgements
The work was supported in part by SFB 650 and Deutsche Forschungsgemeinschaft grants Do492/5–5 and Do492/7–1.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
T. Dörner declares that he has acted as a consultant for Genentech, he has received grant or research support (including clinical trials) from Immunomedics and has acted as a consultant, been involved in speakers' bureaux (honoraria) and received grant or research support (including clinical trials) from Roche.
Gerd R. Burmester declares that he has received grant or research support (including clinical trials) from Medimmune, and has acted as a consultant, been involved in speakers' bureaux (honoraria) and received grant or research support (including clinical trials) from Roche and UCB.
Andreas Radbruch declares no competing interests.
Rights and permissions
About this article
Cite this article
Dörner, T., Radbruch, A. & Burmester, G. B-cell-directed therapies for autoimmune disease. Nat Rev Rheumatol 5, 433–441 (2009). https://doi.org/10.1038/nrrheum.2009.141
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrrheum.2009.141
This article is cited by
-
Effects of rituximab dose on hepatitis B reactivation in patients with resolved infection undergoing immunologic incompatible kidney transplantation
Scientific Reports (2018)
-
Tolerance, loss of tolerance and regaining tolerance to self by immune-mediated events
Immunologic Research (2017)
-
Beyond pan-B-cell-directed therapy — new avenues and insights into the pathogenesis of SLE
Nature Reviews Rheumatology (2016)
-
Rheumatoid factor, not antibodies against citrullinated proteins, is associated with baseline disease activity in rheumatoid arthritis clinical trials
Arthritis Research & Therapy (2015)
-
γ-secretase directly sheds the survival receptor BCMA from plasma cells
Nature Communications (2015)