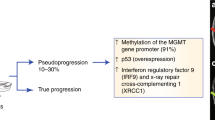
Key Points
-
Biomarkers from gliomas can be detected in the blood and cerebrospinal fluid
-
Known mutations, such as the epidermal growth factor receptor variant III (EGFRvIII), can be detected by analysing soluble, circulating proteins or circulating tumour DNA
-
MicroRNAs—which are relevant as tumour regulators—can be found in extracellular vesicles (EVs) shed by the tumour cells
-
Circulating tumour cells are too scarce to enter routine clinical use as biomarkers to monitor response to therapy
-
Tumour heterogeneity, which is particularly pronounced in glioblastoma, calls for a strategy comprising multiple biomarker molecules; EVs contain several biomarker types, making them an appealing candidate for a comprehensive biomarker
-
Emerging immuno-oncological therapies have created a need for immunological biomarkers
Abstract
Currently, gliomas are diagnosed by neuroimaging, and refined diagnosis requires resection or biopsy to obtain tumour tissue for histopathological classification and grading. Blood-derived biomarkers, therefore, would be useful as minimally invasive markers that could support diagnosis and enable monitoring of tumour growth and response to treatment. Such circulating biomarkers could distinguish true progression from therapy-associated changes such as radiation necrosis, and help evaluate the persistence or disappearance of a therapeutic target, such as an oncoprotein or a targetable gene mutation, after targeted therapy. Unlike for other tumours, circulating biomarkers for gliomas are still being defined and are not yet in use in clinical practice. Circulating tumour DNA (ctDNA) isolated from plasma has been shown to reflect the mutational status of glioblastoma, and extracellular vesicles (EVs) containing ctDNA, microRNA and proteins function as rapidly adapting reservoirs for glioma biomarkers such as typical DNA mutations, regulatory microRNAs and oncoproteins. Ideally, circulating tumour cells could enable profiling of the whole-tumour genome, but they are difficult to detect and can reflect only a single cell type of the heterogeneous tumour composition, whereas EVs reflect the complex heterogeneity of the whole tumour, as well as its adaptations to therapy. Although all categories of potential blood-derived biomarkers need to be developed further, findings from other tumour types suggest that EVs are the most promising biomarkers.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Freidlin, B. & Korn, E. L. Biomarker enrichment strategies: matching trial design to biomarker credentials. Nat. Rev. Clin. Oncol. 11, 81–90 (2014).
Alix-Panabieres, C. & Pantel, K. Circulating tumor cells: liquid biopsy of cancer. Clin. Chem. 59, 110–118 (2013).
Auer, M., Heitzer, E., Ulz, P., Geigl, J. B. & Speicher, M. R. Single circulating tumor cell sequencing for monitoring. Oncotarget 4, 812–813 (2013).
Heitzer, E., Ulz, P. & Geigl, J. B. Circulating tumor DNA as a liquid biopsy for cancer. Clin. Chem. 61, 112–123 (2015).
Mishra, P. J. Non-coding RNAs as clinical biomarkers for cancer diagnosis and prognosis. Expert Rev. Mol. Diagn. 14, 917–919 (2014).
Tsujiura, M. et al. Liquid biopsy of gastric cancer patients: circulating tumor cells and cell-free nucleic acids. World J. Gastroenterol. 20, 3265–3286 (2014).
Brown, K. J. et al. The human secretome atlas initiative: implications in health and disease conditions. Biochim. Biophys. Acta 1834, 2454–2461 (2013).
Schaaij-Visser, T. B., de Wit, M., Lam, S. W. & Jimenez, C. R. The cancer secretome, current status and opportunities in the lung, breast and colorectal cancer context. Biochim. Biophys. Acta 1834, 2242–2258 (2013).
Crowley, E., Di Nicolantonio, F., Loupakis, F. & Bardelli, A. Liquid biopsy: monitoring cancer-genetics in the blood. Nat. Rev. Clin. Oncol. 10, 472–484 (2013).
Diaz, L. A. Jr & Bardelli, A. Liquid biopsies: genotyping circulating tumor DNA. J. Clin. Oncol. 32, 579–586 (2014).
Weller, M. et al. EANO guideline for the diagnosis and treatment of anaplastic gliomas and glioblastoma. Lancet Oncol. 15, e395–e403 (2014).
Wen, P. Y. et al. Updated response assessment criteria for high-grade gliomas: response assessment in neuro-oncology working group. J. Clin. Oncol. 28, 1963–1972 (2010).
Kros, J. M. et al. Circulating glioma biomarkers. Neuro Oncol. 17, 343–360 (2014).
Louis, D. N. et al. The 2007 WHO classification of tumours of the central nervous system. Acta Neuropathol. 114, 97–109 (2007).
Eckel-Passow, J. E. et al. Glioma groups based on 1p/19q, IDH, and TERT promoter mutations in tumors. N. Engl. J. Med. 372, 2499–2508 (2015).
Reuss, D. E. et al. Adult IDH wild type astrocytomas biologically and clinically resolve into other tumor entities. Acta Neuropathol. 130, 407–417 (2015).
Weller, M. et al. Molecular classification of diffuse cerebral WHO grade II/III gliomas using genome- and transcriptome-wide profiling improves stratification of prognostically distinct patient groups. Acta Neuropathol. 129, 679–693 (2015).
Louis, D. N. et al. International Society Of Neuropathology—Haarlem consensus guidelines for nervous system tumor classification and grading. Brain Pathol. 24, 429–435 (2014).
Riemenschneider, M. J., Jeuken, J. W., Wesseling, P. & Reifenberger, G. Molecular diagnostics of gliomas: state of the art. Acta Neuropathol. 120, 567–584 (2010).
Parsons, D. W. et al. An integrated genomic analysis of human glioblastoma multiforme. Science 321, 1807–1812 (2008).
Lai, R. K. et al. Genome-wide methylation analyses in glioblastoma multiforme. PLoS ONE 9, e89376 (2014).
Thomas, L., Di Stefano, A. L. & Ducray, F. Predictive biomarkers in adult gliomas: the present and the future. Curr. Opin. Oncol. 25, 689–694 (2013).
Stupp, R. & Hegi, M. E. Neuro-oncology: oligodendroglioma and molecular markers. Lancet Neurol. 6, 10–12 (2007).
Aldape, K., Zadeh, G., Mansouri, S., Reifenberger, G. & von Deimling, A. Glioblastoma: pathology, molecular mechanisms and markers. Acta Neuropathol. 129, 829–848 (2015).
Comprehensive genomic characterization defines human glioblastoma genes and core pathways. Nature 455, 1061–1068 (2008).
Phillips, H. S. et al. Molecular subclasses of high-grade glioma predict prognosis, delineate a pattern of disease progression, and resemble stages in neurogenesis. Cancer Cell 9, 157–173 (2006).
Verhaak, R. G. et al. Integrated genomic analysis identifies clinically relevant subtypes of glioblastoma characterized by abnormalities in PDGFRA, IDH1, EGFR, and NF1. Cancer Cell 17, 98–110 (2010).
Sottoriva, A. et al. Intratumor heterogeneity in human glioblastoma reflects cancer evolutionary dynamics. Proc. Natl Acad. Sci. USA 110, 4009–4014 (2013).
Westphal, M. & Lamszus, K. The neurobiology of gliomas: from cell biology to the development of therapeutic approaches. Nat. Rev. Neurosci. 12, 495–508 (2011).
Brennan, C. W. et al. The somatic genomic landscape of glioblastoma. Cell 155, 462–477 (2013).
Gollapalli, K. et al. Investigation of serum proteome alterations in human glioblastoma multiforme. Proteomics 12, 2378–2390 (2012).
Hermansen, S. K. & Kristensen, B. W. MicroRNA biomarkers in glioblastoma. J. Neurooncol. 114, 13–23 (2013).
Lang, M. F. et al. Genome-wide profiling identified a set of miRNAs that are differentially expressed in glioblastoma stem cells and normal neural stem cells. PLoS ONE 7, e36248 (2012).
Lemee, J. M. et al. Proteomic analysis of glioblastomas: what is the best brain control sample? J. Proteomics 85, 165–173 (2013).
Huang, S. S. et al. Linking proteomic and transcriptional data through the interactome and epigenome reveals a map of oncogene-induced signaling. PLoS Comput. Biol. 9, e1002887 (2013).
Biomarkers Definitions Working Group. Biomarkers and surrogate endpoints: preferred definitions and conceptual framework. Clin. Pharmacol. Ther. 69, 89–95 (2001).
Gutman, S. & Kessler, L. G. The US Food and Drug Administration perspective on cancer biomarker development. Nat. Rev. Cancer 6, 565–571 (2006).
Esteller, M. et al. Inactivation of the DNA-repair gene MGMT and the clinical response of gliomas to alkylating agents. N. Engl. J. Med. 343, 1350–1354 (2000).
Hegi, M. E. et al. MGMT gene silencing and benefit from temozolomide in glioblastoma. N. Engl. J. Med. 352, 997–1003 (2005).
Stupp, R. et al. Radiotherapy plus concomitant and adjuvant temozolomide for glioblastoma. N. Engl. J. Med. 352, 987–996 (2005).
Park, C. K. et al. The changes in MGMT promoter methylation status in initial and recurrent glioblastomas. Transl. Oncol. 5, 393–397 (2012).
Cairncross, G. et al. Phase III trial of chemoradiotherapy for anaplastic oligodendroglioma: long-term results of RTOG 9402. J. Clin. Oncol. 31, 337–343 (2013).
Eigenbrod, S. et al. Molecular stereotactic biopsy technique improves diagnostic accuracy and enables personalized treatment strategies in glioma patients. Acta Neurochir. (Wien) 156, 1427–1440 (2014).
Ahluwalia, M. S. & Chang, S. M. Medical therapy of gliomas. J. Neurooncol. 119, 503–512 (2014).
Wen, P. Y., Lee, E. Q., Reardon, D. A., Ligon, K. L. & Alfred Yung, W. K. Current clinical development of PI3K pathway inhibitors in glioblastoma. Neuro Oncol. 14, 819–829 (2012).
Wick, W. et al. Pathway inhibition: emerging molecular targets for treating glioblastoma. Neuro Oncol. 13, 566–579 (2011).
Chinot, O. L. et al. Bevacizumab plus radiotherapy-temozolomide for newly diagnosed glioblastoma. N. Engl. J. Med. 370, 709–722 (2014).
Gilbert, M. R. et al. A randomized trial of bevacizumab for newly diagnosed glioblastoma. N. Engl. J. Med. 370, 699–708 (2014).
Eisele, G. et al. Cilengitide treatment of newly diagnosed glioblastoma patients does not alter patterns of progression. J. Neurooncol. 117, 141–145 (2014).
Chang-Hao Tsao, S. et al. Monitoring response to therapy in melanoma by quantifying circulating tumour DNA with droplet digital PCR for BRAF and NRAS mutations. Sci. Rep. 5, 11198 (2015).
Hochberg, F. H. et al. Glioma diagnostics and biomarkers: an ongoing challenge in the field of medicine and science. Expert Rev. Mol. Diagn. 14, 439–452 (2014).
Minciacchi, V. R., Freeman, M. R. & Di Vizio, D. Extracellular vesicles in cancer: exosomes, microvesicles and the emerging role of large oncosomes. Semin. Cell Dev. Biol. 40, 41–51 (2015).
Holdhoff, M., Yovino, S. G., Boadu, O. & Grossman, S. A. Blood-based biomarkers for malignant gliomas. J. Neurooncol. 113, 345–352 (2013).
Elstner, A. et al. Identification of diagnostic serum protein profiles of glioblastoma patients. J. Neurooncol. 102, 71–80 (2011).
Heitzer, E., Auer, M., Ulz, P., Geigl, J. B. & Speicher, M. R. Circulating tumor cells and DNA as liquid biopsies. Genome Med. 5, 73 (2013).
Dong, L. et al. miRNA microarray reveals specific expression in the peripheral blood of glioblastoma patients. Int. J. Oncol. 45, 746–756 (2014).
Chistiakov, D. A. & Chekhonin, V. P. Extracellular vesicles shed by glioma cells: pathogenic role and clinical value. Tumour Biol. 35, 8425–8438 (2014).
Redzic, J. S., Ung, T. H. & Graner, M. W. Glioblastoma extracellular vesicles: reservoirs of potential biomarkers. Pharmgenomics Pers. Med. 7, 65–77 (2014).
Santiago-Dieppa, D. R. et al. Extracellular vesicles as a platform for 'liquid biopsy' in glioblastoma patients. Expert Rev. Mol. Diagn. 14, 819–825 (2014).
Akers, J. C. et al. MiR-21 in the extracellular vesicles (EVs) of cerebrospinal fluid (CSF): a platform for glioblastoma biomarker development. PLoS ONE 8, e78115 (2013).
Noerholm, M. et al. RNA expression patterns in serum microvesicles from patients with glioblastoma multiforme and controls. BMC Cancer 12, 22 (2012).
Manterola, L. et al. A small noncoding RNA signature found in exosomes of GBM patient serum as a diagnostic tool. Neuro Oncol. 16, 520–527 (2014).
Mikaelian, I., Scicchitano, M., Mendes, O., Thomas, R. A. & Leroy, B. E. Frontiers in preclinical safety biomarkers: microRNAs and messenger RNAs. Toxicol. Pathol. 41, 18–31 (2013).
Toss, A., Mu, Z., Fernandez, S. & Cristofanilli, M. CTC enumeration and characterization: moving toward personalized medicine. Ann. Transl. Med. 2, 108 (2014).
Muller, C. et al. Hematogenous dissemination of glioblastoma multiforme. Sci. Transl. Med. 6, 247ra101 (2014).
Mimeault, M. & Batra, S. K. Molecular biomarkers of cancer stem/progenitor cells associated with progression, metastases, and treatment resistance of aggressive cancers. Cancer Epidemiol. Biomarkers Prev. 23, 234–254 (2014).
Popescu, I. D. et al. Potential serum biomarkers for glioblastoma diagnostic assessed by proteomic approaches. Proteome Sci. 12, 47 (2014).
Lange, R. P. et al. Evaluation of eight plasma proteins as candidate blood-based biomarkers for malignant gliomas. Cancer Invest. 32, 423–429 (2014).
Capper, D. et al. 2-Hydroxyglutarate concentration in serum from patients with gliomas does not correlate with IDH1/2 mutation status or tumor size. Int. J. Cancer 131, 766–768 (2012).
Skog, J. et al. Glioblastoma microvesicles transport RNA and proteins that promote tumour growth and provide diagnostic biomarkers. Nat. Cell Biol. 10, 1470–1476 (2008).
Shao, H. et al. Protein typing of circulating microvesicles allows real-time monitoring of glioblastoma therapy. Nat. Med. 18, 1835–1840 (2012).
Schuster, J. et al. A phase II, multicenter trial of rindopepimut (CDX-110) in newly diagnosed glioblastoma: the ACT III study. Neuro Oncol. 17, 854–861 (2015).
Francis, G. & Stein, S. Circulating cell-free tumour DNA in the management of cancer. Int. J. Mol. Sci. 16, 14122–14142 (2015).
Lebofsky, R. et al. Circulating tumor DNA as a non-invasive substitute to metastasis biopsy for tumor genotyping and personalized medicine in a prospective trial across all tumor types. Mol. Oncol. 9, 783–790 (2014).
Lavon, I., Refael, M., Zelikovitch, B., Shalom, E. & Siegal, T. Serum DNA can define tumor-specific genetic and epigenetic markers in gliomas of various grades. Neuro Oncol. 12, 173–180 (2010).
Bettegowda, C. et al. Detection of circulating tumor DNA in early- and late-stage human malignancies. Sci. Transl. Med. 6, 224ra24 (2014).
Diaz, L. A., Jr. et al. The molecular evolution of acquired resistance to targeted EGFR blockade in colorectal cancers. Nature 486, 537–540 (2012).
Wang, W. Y. et al. Plasma EBV DNA clearance rate as a novel prognostic marker for metastatic/recurrent nasopharyngeal carcinoma. Clin. Cancer Res. 16, 1016–1024 (2010).
Diehl, F. et al. Circulating mutant DNA to assess tumor dynamics. Nat. Med. 14, 985–990 (2008).
Boisselier, B. et al. Detection of IDH1 mutation in the plasma of patients with glioma. Neurology 79, 1693–1698 (2012).
Floyd, D. & Purow, B. Micro-masters of glioblastoma biology and therapy: increasingly recognized roles for microRNAs. Neuro Oncol. 16, 622–627 (2014).
McManus, M. T. MicroRNAs and cancer. Semin. Cancer Biol. 13, 253–258 (2003).
Godlewski, J., Krichevsky, A. M., Johnson, M. D., Chiocca, E. A. & Bronisz, A. Belonging to a network-microRNAs, extracellular vesicles, and the glioblastoma microenvironment. Neuro Oncol. 17, 652–662 (2015).
Chen, X., Liang, H., Zhang, J., Zen, K. & Zhang, C. Y. Secreted microRNAs: a new form of intercellular communication. Trends Cell Biol. 22, 125–132 (2012).
Rosenfeld, N. et al. MicroRNAs accurately identify cancer tissue origin. Nat. Biotechnol. 26, 462–469 (2008).
Tumilson, C. A., Lea, R. W., Alder, J. E. & Shaw, L. Circulating microRNA biomarkers for glioma and predicting response to therapy. Mol. Neurobiol. 50, 545–558 (2014).
Hua, D. et al. A catalogue of glioblastoma and brain MicroRNAs identified by deep sequencing. OMICS 16, 690–699 (2012).
Lin, H. M. et al. Circulating microRNAs are associated with docetaxel chemotherapy outcome in castration-resistant prostate cancer. Br. J. Cancer 110, 2462–2471 (2014).
Sozzi, G. et al. Clinical utility of a plasma-based miRNA signature classifier within computed tomography lung cancer screening: a correlative MILD trial study. J. Clin. Oncol. 32, 768–773 (2014).
Srinivasan, S., Patric, I. R. & Somasundaram, K. A ten-microRNA expression signature predicts survival in glioblastoma. PLoS ONE 6, e17438 (2011).
Hayes, J. et al. Prediction of clinical outcome in glioblastoma using a biologically relevant nine-microRNA signature. Mol. Oncol. 9, 704–714 (2014).
Li, R. et al. Identification of intrinsic subtype-specific prognostic microRNAs in primary glioblastoma. J. Exp. Clin. Cancer Res. 33, 9 (2014).
Sandmann, T. et al. Patients with proneural glioblastoma may derive overall survival benefit from the addition of bevacizumab to first-line radiotherapy and temozolomide: retrospective analysis of the AVAglio trial. J. Clin. Oncol. http://dx.doi.org/10.1200/JCO.2015.61.5005.
Chen, H., Li, X., Li, W. & Zheng, H. miR-130a can predict response to temozolomide in patients with glioblastoma multiforme, independently of O6-methylguanine-DNA methyltransferase. J. Transl. Med. 13, 69 (2015).
Kushwaha, D. et al. A genome-wide miRNA screen revealed miR-603 as a MGMT-regulating miRNA in glioblastomas. Oncotarget 5, 4026–4039 (2014).
Qu, S., Guan, J. & Liu, Y. Identification of microRNAs as novel biomarkers for glioma detection: a meta-analysis based on 11 articles. J. Neurol. Sci. 348, 181–187 (2015).
Teplyuk, N. M. et al. MicroRNAs in cerebrospinal fluid identify glioblastoma and metastatic brain cancers and reflect disease activity. Neuro Oncol. 14, 689–700 (2012).
Zhang, W. et al. miR-181d: a predictive glioblastoma biomarker that downregulates MGMT expression. Neuro Oncol. 14, 712–719 (2012).
Verbeek, B., Southgate, T. D., Gilham, D. E. & Margison, G. P. O6-Methylguanine-DNA methyltransferase inactivation and chemotherapy. Br. Med. Bull. 85, 17–33 (2008).
Belting, M. & Wittrup, A. Nanotubes, exosomes, and nucleic acid-binding peptides provide novel mechanisms of intercellular communication in eukaryotic cells: implications in health and disease. J. Cell Biol. 183, 1187–1191 (2008).
Fitzner, D. et al. Selective transfer of exosomes from oligodendrocytes to microglia by macropinocytosis. J. Cell Sci. 124, 447–458 (2011).
Kucharzewska, P. & Belting, M. Emerging roles of extracellular vesicles in the adaptive response of tumour cells to microenvironmental stress. J. Extracell. Vesicles 2, 20304 (2013).
Belting, M. & Christianson, H. C. Role of exosomes and microvesicles in hypoxia-associated tumour development and cardiovascular disease. J. Intern. Med. 278, 251–263 (2015).
Mathivanan, S. et al. Vesiclepedia [online], (2015).
Valadi, H. et al. Exosome-mediated transfer of mRNAs and microRNAs is a novel mechanism of genetic exchange between cells. Nat. Cell Biol. 9, 654–659 (2007).
Morishita, M. et al. Quantitative analysis of tissue distribution of the B16BL6-derived exosomes using a streptavidin-lactadherin fusion protein and iodine-125-labeled biotin derivative after intravenous injection in mice. J. Pharm. Sci. 104, 705–713 (2015).
Muller, L. et al. Exosomes isolated from plasma of glioma patients enrolled in a vaccination trial reflect antitumor immune activity and might predict survival. Oncoimmunology 4, e1008347 (2015).
Shao, H. et al. Chip-based analysis of exosomal mRNA mediating drug resistance in glioblastoma. Nat. Commun. 6, 6999 (2015).
Harshyne, L. A., Nasca, B. J., Kenyon, L. C., Andrews, D. W. & Hooper, D. C. Serum exosomes and cytokines promote a T-helper cell type 2 environment in the peripheral blood of glioblastoma patients. Neuro Oncol. http://dx.doi.org/10.1093/neuonc/nov107.
Best, M. G. et al. Liquid biopsies in patients with diffuse glioma. Acta Neuropathol. 129, 849–865 (2015).
Antonyak, M. A. & Cerione, R. A. Microvesicles as mediators of intercellular communication in cancer. Methods Mol. Biol. 1165, 147–173 (2014).
Mahmoudi, K., Ezrin, A. & Hadjipanayis, C. Small extracellular vesicles as tumor biomarkers for glioblastoma. Mol. Aspects Med. http://dx.doi.org/10.1016/j.mam.2015.06.008.
Muller, L., Hong, C. S., Stolz, D. B., Watkins, S. C. & Whiteside, T. L. Isolation of biologically-active exosomes from human plasma. J. Immunol. Methods 411, 55–65 (2014).
Yoshioka, Y. et al. Ultra-sensitive liquid biopsy of circulating extracellular vesicles using ExoScreen. Nat. Commun. 5, 3591 (2014).
Santana, S. M., Antonyak, M. A., Cerione, R. A. & Kirby, B. J. Microfluidic isolation of cancer-cell-derived microvesicles from hetergeneous extracellular shed vesicle populations. Biomed. Microdevices 16, 869–877 (2014).
Atai, N. A. et al. Heparin blocks transfer of extracellular vesicles between donor and recipient cells. J. Neurooncol. 115, 343–351 (2013).
Bronisz, A. et al. Extracellular vesicles modulate the glioblastoma microenvironment via a tumor suppression signaling network directed by miR-1. Cancer Res. 74, 738–750 (2014).
Cossetti, C. et al. Extracellular vesicles from neural stem cells transfer IFN-γ via Ifngr1 to activate Stat1 signaling in target cells. Mol. Cell 56, 193–204 (2014).
Cirkel, G. A., Gadellaa-van Hooijdonk, C. G., Koudijs, M. J., Willems, S. M. & Voest, E. E. Tumor heterogeneity and personalized cancer medicine: are we being outnumbered? Future Oncol. 10, 417–428 (2014).
Macarthur, K. M. et al. Detection of brain tumor cells in the peripheral blood by a telomerase promoter-based assay. Cancer Res. 74, 2152–2159 (2014).
Sullivan, J. P. et al. Brain tumor cells in circulation are enriched for mesenchymal gene expression. Cancer Discov. 4, 1299–1309 (2014).
Seymour, T., Nowak, A. & Kakulas, F. Targeting aggressive cancer stem cells in glioblastoma. Front. Oncol. 5, 159 (2015).
Singh, S. K. et al. Identification of a cancer stem cell in human brain tumors. Cancer Res. 63, 5821–5828 (2003).
Papadaki, M. A. et al. Co-expression of putative stemness and epithelial-to-mesenchymal transition markers on single circulating tumour cells from patients with early and metastatic breast cancer. BMC Cancer 14, 651 (2014).
Tinhofer, I., Saki, M., Niehr, F., Keilholz, U. & Budach, V. Cancer stem cell characteristics of circulating tumor cells. Int. J. Radiat. Biol. 90, 622–627 (2014).
Reardon, D. A., Ballman, K. V., Buckner, J. C., Chang, S. M. & Ellingson, B. M. Impact of imaging measurements on response assessment in glioblastoma clinical trials. Neuro Oncol. 16 (Suppl. 7), vii24–vii35 (2014).
Qiao, X. J. et al. Arterial spin-labeling perfusion MRI stratifies progression-free survival and correlates with epidermal growth factor receptor status in glioblastoma. AJNR Am. J. Neuroradiol. 36, 672–677 (2015).
Li, Y. et al. Survival analysis in patients with newly diagnosed glioblastoma using pre- and postradiotherapy MR spectroscopic imaging. Neuro Oncol. 15, 607–617 (2013).
Chaumeil, M. M. et al. Hyperpolarized 13C MR spectroscopic imaging can be used to monitor Everolimus treatment in vivo in an orthotopic rodent model of glioblastoma. Neuroimage 59, 193–201 (2012).
Chaumeil, M. M. et al. Hyperpolarized [1-13C] glutamate: a metabolic imaging biomarker of IDH1 mutational status in glioma. Cancer Res. 74, 4247–4257 (2014).
Nasrallah, I. & Dubroff, J. An overview of PET neuroimaging. Semin. Nucl. Med. 43, 449–461 (2013).
Linden, H. M. & Dehdashti, F. Novel methods and tracers for breast cancer imaging. Semin. Nucl. Med. 43, 324–329 (2013).
Jansen, N. L. et al. Prognostic significance of dynamic 18F-FET PET in newly diagnosed astrocytic high-grade glioma. J. Nucl. Med. 56, 9–15 (2015).
Mankoff, D. A., Pryma, D. A. & Clark, A. S. Molecular imaging biomarkers for oncology clinical trials. J. Nucl. Med. 55, 525–528 (2014).
Reardon, D. A. et al. Immunotherapy advances for glioblastoma. Neuro Oncol. 16, 1441–1458 (2014).
Parney, I. F. Basic concepts in glioma immunology. Adv. Exp. Med. Biol. 746, 42–52 (2012).
Wurdinger, T., Deumelandt, K., van der Vliet, H. J., Wesseling, P. & de Gruijl, T. D. Mechanisms of intimate and long-distance cross-talk between glioma and myeloid cells: how to break a vicious cycle. Biochim. Biophys. Acta 1846, 560–575 (2014).
Graner, M. W. et al. Proteomic and immunologic analyses of brain tumor exosomes. FASEB J. 23, 1541–1557 (2009).
Bodmer, S. et al. Immunosuppression and transforming growth factor-beta in glioblastoma. Preferential production of transforming growth factor-beta 2. J. Immunol. 143, 3222–3229 (1989).
Preusser, M., Lim, M., Hafler, D. A., Reardon, D. A. & Sampson, J. H. Prospects of immune checkpoint modulators in the treatment of glioblastoma. Nat. Rev. Neurol. http://dx.doi.org/10.1038/nrneurol.2015.139.
Castro, M. G., Baker, G. J. & Lowenstein, P. R. Blocking immunosuppressive checkpoints for glioma therapy: the more the merrier! Clin. Cancer Res. 20, 5147–5149 (2014).
Vlahovic, G., Fecci, P. E., Reardon, D. & Sampson, J. H. Programmed death ligand 1 (PD-L1) as an immunotherapy target in patients with glioblastoma. Neuro Oncol. 17, 1043–1045 (2015).
Berghoff, A. S. et al. Programmed death ligand 1 expression and tumor-infiltrating lymphocytes in glioblastoma. Neuro Oncol. 17, 1064–1075 (2015).
Sims, J. S., Ung, T. H., Neira, J. A., Canoll, P. & Bruce, J. N. Biomarkers for glioma immunotherapy: the next generation. J. Neurooncol. 123, 359–372 (2015).
Albulescu, R. et al. Cytokine patterns in brain tumour progression. Mediators Inflamm. 2013, 979748 (2013).
Zhou, M. et al. Circulating levels of the innate and humoral immune regulators CD14 and CD23 are associated with adult glioma. Cancer Res. 70, 7534–7542 (2010).
Arimappamagan, A. et al. A fourteen gene GBM prognostic signature identifies association of immune response pathway and mesenchymal subtype with high risk group. PLoS ONE 8, e62042 (2013).
Yin, Y. et al. Tumor-secreted miR-214 induces regulatory T cells: a major link between immune evasion and tumor growth. Cell Res. 24, 1164–1180 (2014).
Katsuda, T., Kosaka, N. & Ochiya, T. The roles of extracellular vesicles in cancer biology: toward the development of novel cancer biomarkers. Proteomics 14, 412–425 (2014).
Cossetti, C. et al. Extracellular membrane vesicles and immune regulation in the brain. Front. Physiol. 3, 117 (2012).
Ellert-Miklaszewska, A. et al. Molecular definition of the pro-tumorigenic phenotype of glioma-activated microglia. Glia 61, 1178–1190 (2013).
Jiang, Y. & Uhrbom, L. On the origin of glioma. Ups. J. Med. Sci. 117, 113–121 (2012).
Deck, J. H., Eng, L. F. & Woodcock, S. M. The role of glial fibrillary acidic protein in the diagnosis of central nervous system tumors. Acta Neuropathol. 42, 183–190 (1978).
Guo, C., Pirozzi, C. J., Lopez, G. Y. & Yan, H. Isocitrate dehydrogenase mutations in gliomas: mechanisms, biomarkers and therapeutic target. Curr. Opin. Neurol. 24, 648–652 (2011).
Acknowledgements
Neuro-oncology research in the Department of Neurosurgery, University Hospital Eppendorf has been supported by the Deutsche Forschungsgemeinschaft (LA1300), the Deutsche Krebshilfe, the Roggenbuck Stiftung, Hamburg and the Rickertsen Stiftung, Hamburg.
Author information
Authors and Affiliations
Contributions
Both authors contribute equally to all aspects of preparation of the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Westphal, M., Lamszus, K. Circulating biomarkers for gliomas. Nat Rev Neurol 11, 556–566 (2015). https://doi.org/10.1038/nrneurol.2015.171
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrneurol.2015.171
This article is cited by
-
A systematic review of extracellular vesicles as non-invasive biomarkers in glioma diagnosis, prognosis, and treatment response monitoring
Molecular Biology Reports (2021)
-
Exosomal noncoding RNAs in Glioma: biological functions and potential clinical applications
Molecular Cancer (2020)
-
Profiling cytotoxic microRNAs in pediatric and adult glioblastoma cells by high-content screening, identification, and validation of miR-1300
Oncogene (2020)
-
P2X7 receptors and glioma cells
Purinergic Signalling (2020)
-
Circulating MicroRNA as Novel Potential Biomarkers for the Diagnosis of Highly Malignant Gliomas
Neuroscience and Behavioral Physiology (2020)