Abstract
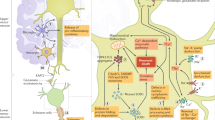
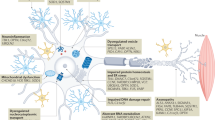
Amyotrophic lateral sclerosis (ALS) is a neurodegenerative disease that results in progressive loss of bulbar and limb function. Patients typically die from respiratory failure within 3 years of symptom onset. The incidence of ALS in Europe is 2–3 cases per 100,000 individuals in the general population, and the overall lifetime risk of developing the disease is 1:400. ALS is familial in 5% of cases, and shows a Mendelian pattern of inheritance. ALS is recognized to overlap with frontotemporal dementia. Diagnosis is made on clinical grounds, using internationally recognized consensus criteria, after exclusion of conditions that can mimic ALS. The Revised ALS Functional Rating Scale is currently the most widely used assessment tool; scores are used to predict survival, and have been employed extensively in clinical trials. Riluzole remains the only effective drug, and extends the average survival of patients by 3–6 months. Optimal treatment is based on symptom management and preservation of quality of life, provided in a multidisciplinary setting. The discovery of further effective disease-modifying therapies remains a critical need for patients with this devastating condition.
Key Points
-
Amyotrophic lateral sclerosis (ALS) is a syndrome of progressive deterioration involving the corticospinal tract, brainstem, and anterior horn cells of the spinal cord
-
The risk of developing ALS peaks between the ages of 50 years and 75 years; disease rates are elevated in populations of white European ancestry, and reduced in mixed populations
-
No definitive test for ALS exists; the diagnosis is established by excluding other causes of progressive upper motor neuron and lower motor neuron dysfunction
-
Up to 15% of patients with ALS have frontotemporal dementia, and a further 25% have evidence of cognitive impairment, mainly executive dysfunction
-
Clinical care is based on symptom management; however, riluzole, the only available disease-modifying drug, improves patients' survival early in the course of the disease
-
Further improvements in survival will depend on advances in understanding the origins and spread of this syndrome
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Charcot, J. M. De la sclérose latérale amyotrophique [French]. Progrès Médical 2, 325 (1874).
Kiernan, M. C. et al. Amyotrophic lateral sclerosis. Lancet 377, 942–955 (2011).
Deng, H. X. et al. FUS-immunoreactive inclusions are a common feature in sporadic and non-SOD1 familial amyotrophic lateral sclerosis. Ann. Neurol. 67, 739–748 (2010).
Beleza-Meireles, A. & Al-Chalabi, A. Genetic studies of amyotrophic lateral sclerosis: controversies and perspectives. Amyotroph. Lateral Scler. 10, 1–14 (2009).
Beghi, E. et al. for the Eurals Consortium. The epidemiology and treatment of ALS: focus on the heterogeneity of the disease and critical appraisal of therapeutic trials. Amyotroph. Lateral Scler. 12, 1–10 (2011).
Byrne, S. et al. Rate of familial amyotrophic lateral sclerosis: a systematic review and meta-analysis. J. Neurol. Neurosurg. Psychiatry 82, 623–627 (2011).
Logroscino, G. et al. Incidence of amyotrophic lateral sclerosis in Europe. J. Neurol. Neurosurg. Psychiatry 81, 385–390 (2010).
Kuzuhara, S. Muro Disease: Amyotrophic lateral sclerosis/parkinsonism-dementia complex in Kii Peninsula of Japan. Brain Nerve 63, 119–129 (2011).
Chiò, A. et al. Epidemiology of ALS in Italy: a 10-year prospective population-based study. Neurology 72, 725–731 (2009).
O'Toole, O. et al. Epidemiology and clinical features of amyotrophic lateral sclerosis in Ireland between 1995 and 2004. J. Neurol. Neurosurg. Psychiatry 79, 30–32 (2008).
Johnston, C. A. et al. Amyotrophic lateral sclerosis in an urban setting: a population based study of inner city London. J. Neurol. 253, 1642–1643 (2006).
Alonso, A., Logroscino, G., Jick, S. S. & Hernán, M. A. Incidence and lifetime risk of motor neuron disease in the United Kingdom: a population-based study. Eur. J. Neurol. 16, 745–751 (2009).
Cronin, S., Hardiman, O. & Traynor, B. J. Ethnic variation in the incidence of ALS: a systematic review. Neurology 68, 1002–1007 (2007).
Zaldivar, T. et al. Reduced frequency of ALS in an ethnically mixed population: a population-based mortality study. Neurology 72, 1640–1650 (2009).
Steele, J. C. Parkinsonism-dementia complex of Guam. Mov. Disord. 20 (Suppl. 12), S99–S107 (2005).
Kuzuhara, S. & Kokubo, Y. Atypical parkinsonism of Japan: amyotrophic lateral sclerosis-parkinsonism-dementia complex of the Kii peninsula of Japan (Muro disease): an update. Mov. Disord. 20 (Suppl. 12), S108–S113 (2005).
Steele, J. C. & McGeer, P. L. The ALS/PDC syndrome of Guam and the cycad hypothesis. Neurology 70, 1984–1990 (2008).
Sutedja, N. A. et al. What we truly know about occupation as a risk factor for ALS: a critical and systematic review. Amyotroph. Lateral Scler. 10, 295–301 (2009).
Chiò, A., Benzi, G., Dossena, M., Mutani, R. & Mora, G. Severely increased risk of amyotrophic lateral sclerosis among Italian professional football players. Brain 128, 472–476 (2005).
Chio, A. et al. ALS in Italian professional soccer players: the risk is still present and could be soccer-specific. Amyotroph. Lateral Scler. 10, 205–209 (2009).
Kasarskis, E. J. et al. Clinical aspects of ALS in Gulf War veterans. Amyotroph. Lateral Scler. 10, 35–41 (2009).
Horner, R. D. et al. Amyotrophic lateral sclerosis among 1991 Gulf War veterans: evidence for a time-limited outbreak. Neuroepidemiology 31, 28–32 (2008).
Armon, C. Smoking may be considered an established risk factor for sporadic ALS. Neurology 73, 1693–1698 (2009).
Alonso, A., Logroscino, G. & Hernán, M. A. Smoking and the risk of amyotrophic lateral sclerosis: a systematic review and meta-analysis. J. Neurol. Neurosurg. Psychiatry 81, 1249–1252 (2010).
Sutedja, N. A. et al. Exposure to chemicals and metals and risk of amyotrophic lateral sclerosis: a systematic review. Amyotroph. Lateral Scler. 10, 302–309 (2009).
Ferguson, T. A. & Elman, L. B. Clinical presentation and diagnosis of amyotrophic lateral sclerosis. NeuroRehabilitation 22, 409–416 (2007).
Kiernan, M. C. et al. Amyotrophic lateral sclerosis. Lancet 377, 942–955 (2011).
Ravits, J. M. & La Spada, A. R. ALS motor phenotype heterogeneity, focality, and spread: deconstructing motor neuron degeneration. Neurology 73, 805–811 (2009).
Eisen, A., Kim, S. & Pant, B. Amyotrophic lateral sclerosis (ALS): a phylogenetic disease of the corticomotoneuron? Muscle Nerve 15, 219–224 (1992).
Donaghy, C. et al. Slow saccades in bulbar-onset motor neurone disease. J. Neurol. 257, 1134–1140 (2010).
Donaghy, C. et al. Ocular fixation instabilities in motor neurone disease. A marker of frontal lobe dysfunction? J. Neurol. 256, 420–426 (2009).
Chiò, A. ISIS Survey: an international study on the diagnostic process and its implications in amyotrophic lateral sclerosis. J. Neurol. 246 (Suppl. 3), III1–III5 (1999).
Phukan, J., Pender, N. P. & Hardiman, O. Cognitive impairment in amyotrophic lateral sclerosis. Lancet Neurol. 6, 994–1003 (2007).
Strong, M. J. et al. Consensus criteria for the diagnosis of frontotemporal cognitive and behavioural syndromes in amyotrophic lateral sclerosis. Amyotroph. Lateral Scler. 10, 131–136 (2009).
Strong, M. J. The syndromes of frontotemporal dysfunction in amyotrophic lateral sclerosis. Amyotroph. Lateral Scler. 9, 323–338 (2008).
Raaphorst, J., de Visser, M., Linssen, W. H., de Haan, R. J. & Schmand, B. The cognitive profile of amyotrophic lateral sclerosis: A meta-analysis. Amyotroph. Lateral Scler. 11, 27–37 (2010).
Elamin, M. et al. Executive dysfunction is a negative prognostic indictor in patients with ALS without dementia. Neurology 76, 1263–1269 (2011).
Phukan, J. et al. The syndrome of cognitive impairment in amyotrophic lateral sclerosis: a population-based study. J. Neurol. Neurosurg. Psychiatry http://dx.doi.org/10.1136/jnnp-2011-300188.
Abrahams, S. et al. Verbal fluency and executive dysfunction in amyotrophic lateral sclerosis (ALS). Neuropsychologia 38, 734–747 (2000).
Woolley, S. C. et al. Detecting frontotemporal dysfunction in ALS: utility of the ALS Cognitive Behavioral Screen (ALS-CBS). Amyotroph. Lateral Scler. 11, 303–311 (2010).
Gilbert, R. M., Fahn, S., Mitsumoto, H. & Rowland, L. P. Parkinsonism and motor neuron diseases: twenty-seven patients with diverse overlap syndromes. Mov. Disord. 25, 1868–1875 (2010).
Farníková, K., Kanovský, P., Nestrasil, I. & Otruba, P. Coexistence of parkinsonism, dementia and upper motor neuron syndrome in four Czech patients. J. Neurol. Sci. 296, 47–54 (2010).
Fallis, B. A. & Hardiman, O. Aggregation of neurodegenerative disease in ALS kindreds. Amyotroph. Lateral Scler. 10, 95–98 (2009).
Przedborski, S., Vila, M. & Jackson-Lewis, V. Neurodegeneration: what is it and where are we? J. Clin. Invest. 111, 3–10 (2003).
Gordon, P. H. et al. The natural history of primary lateral sclerosis. Neurology 66, 647–653 (2006).
Brugman, F. et al. Primary lateral sclerosis as a phenotypic manifestation of familial ALS. Neurology 64, 1778–1779 (2005).
Brugman, F. et al. Differentiation of hereditary spastic paraparesis from primary lateral sclerosis in sporadic adult-onset upper motor neuron syndromes. Arch. Neurol. 66, 509–514 (2009).
Visser, J. et al. Disease course and prognostic factors of progressive muscular atrophy. Arch. Neurol. 64, 522–528 (2007).
Ince, P. G. et al. Corticospinal tract degeneration in the progressive muscular atrophy variant of ALS. Neurology 60, 1252–1258 (2003).
Van den Berg-Vos, R. M. et al. A long-term prospective study of the natural course of sporadic adult-onset lower motor neuron syndromes. Arch. Neurol. 66, 751–757 (2009).
Traynor, B. J. et al. Amyotrophic lateral sclerosis mimic syndromes: a population-based study. Arch. Neurol. 57, 108–113 (2000).
Davenport, R. J., Swingler, R. J., Chancellor, A. M. & Warlow, C. P. Avoiding false positive diagnoses of motor neuron disease: lessons from the Scottish Motor Neuron Disease Register. J. Neurol. Neurosurg. Psychiatry 60, 147–151 (1996).
Cats, E. A. et al. Correlates of outcome and response to IVIg in 88 patients with multifocal motor neuropathy. Neurology 75, 818–825 (2010).
Finsterer, J. Perspectives of Kennedy's disease. J. Neurol. Sci. 298, 1–10 (2010).
Brooks, B. R. El Escorial World Federation of Neurology criteria for the diagnosis of amyotrophic lateral sclerosis. Subcommittee on Motor Neuron Diseases/Amyotrophic Lateral Sclerosis of the World Federation of Neurology Research Group on Neuromuscular Diseases and the El Escorial “Clinical limits of amyotrophic lateral sclerosis” workshop contributors. J. Neurol. Sci. 124 (Suppl.), 96–107 (1994).
Miller, R. G., Munsat, T. L., Swash, M. & Brooks, B. R. Consensus guidelines for the design and implementation of clinical trials in ALS. World Federation of Neurology committee on Research. J. Neurol. Sci. 169, 2–12 (1999).
de Carvalho, M. et al. Electrodiagnostic criteria for diagnosis of ALS. Clin. Neurophysiol. 119, 497–503 (2008).
Schrooten, M., Smetcoren, C., Robberecht, W. & Van Damme, P. Benefit of the Awaji diagnostic algorithm for amyotrophic lateral sclerosis: A prospective study. Ann. Neurol. 70, 79–83 (2011).
Forbes, R. B., Colville, S. & Swingler, R. J. Are the El Escorial and Revised El Escorial criteria for ALS reproducible? A study of inter-observer agreement. Amyotroph. Lateral Scler. Other Motor Neuron Disord. 2, 135–138 (2001).
Beghi, E. et al. Reliability of the El Escorial diagnostic criteria for amyotrophic lateral sclerosis. Neuroepidemiology 21, 265–270 (2002).
Traynor, B. J. et al. Clinical features of amyotrophic lateral sclerosis according to the El Escorial and Airlie House diagnostic criteria: a population-based study. Arch. Neurol. 57, 1171–1176 (2000).
Daube, J. R. Electrodiagnostic studies in amyotrophic lateral sclerosis and other motor neuron disorders. Muscle Nerve 23, 1488–1502 (2000).
Eisen, A. & Swash, M. Clinical neurophysiology of ALS. Clin. Neurophysiol. 112, 2190–2201 (2001).
Triggs, W. J. et al. Transcranial magnetic stimulation identifies upper motor neuron involvement in motor neuron disease. Neurology 53, 605–611 (1999).
Mills, K. R. The natural history of central motor abnormalities in amyotrophic lateral sclerosis. Brain 126, 2558–2566 (2003).
Vucic, S., Howells, J., Trevillion, L. & Kiernan, M. C. Assessment of cortical excitability using threshold tracking techniques. Muscle Nerve 33, 477–486 (2006).
Vucic, S. & Kiernan, M. C. Novel threshold tracking techniques suggest that cortical hyperexcitability is an early feature of motor neuron disease. Brain 129, 2436–2446 (2006).
Byrne, S. et al. Proposed criteria for familial amyotrophic lateral sclerosis. Amyotroph. Lateral Scler. 12, 157–159 (2011).
Gros-Louis, F., Soucy, G., Larivière, R. & Julien, J. P. Intracerebroventricular infusion of monoclonal antibody or its derived Fab fragment against misfolded forms of SOD1 mutant delays mortality in a mouse model of ALS. J. Neurochem. 113, 1188–1199 (2010).
Isis Pharmaceuticals [online], (2011).
Phase II/III randomized, placebo-controlled trial of arimoclomol in SOD1 positive familial amyotropic lateral sclerosis. ClinicalTrials.gov [online], (2011).
SOD1 inhibition by pyrimethamine in familial amyotrophic lateral sclerosis (ALS). ClinicalTrials.gov [online], (2011).
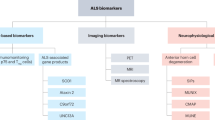
Turner, M. R., Kiernan, M. C., Leigh, P. N. & Talbot, K. Biomarkers in amyotrophic lateral sclerosis. Lancet Neurology 8, 94–109 (2009).
Hecht, M. J. et al. Hyperintense and hypointense MRI signals of the precentral gyrus and corticospinal tract in ALS: a follow-up examination including FLAIR images. J. Neurol. Sci. 199, 59–65 (2002).
Lulé, D., Ludolph, A. C. & Kassubek, J. MRI-based functional neuroimaging in ALS: an update. Amyotroph. Lateral Scler. 10, 258–268 (2009).
Unrath, A., Ludolph, A. C. & Kassubek, J. Brain metabolites in definite amyotrophic lateral sclerosis. A longitudinal proton magnetic resonance spectroscopy study. J. Neurol. 254, 1099–1106 (2007).
Senda, J. et al. Progressive and widespread brain damage in ALS: MRI voxel-based morphometry and diffusion tensor imaging study. Amyotroph. Lateral Scler. 12, 59–69 (2011).
Abrahams, S. et al. Frontotemporal white matter changes in amyotrophic lateral sclerosis. J. Neurol. 252, 321–331 (2005).
Woolley, S. C., Zhang, Y., Schuff, N., Weiner, M. W. & Katz, J. S. Neuroanatomical correlates of apathy in ALS using 4 Tesla diffusion tensor MRI. Amyotroph. Lateral Scler. 12, 52–58 (2011).
Lulé, D. et al. Neuroimaging of multimodal sensory stimulation in amyotrophic lateral sclerosis J. Neurol. Neurosurg. Psychiatry 81, 899–906 (2010).
Filippini, N. et al. Corpus Callosum involvement is a consistent feature of amyotrophic lateral sclerosis. Neurology 75, 1645–1652 (2010).
Agosta, F. et al. A longitudinal diffusion tensor MRI study of the cervical cord and brain in amyotrophic lateral sclerosis patients. J. Neurol. Neurosurg. Psychiatry 80, 53–55 (2009).
Chiò, A. et al. Prognostic factors in ALS: A critical review. Amyotroph. Lateral Scler. 10, 310–323 (2009).
Wijesekera, L. C. et al. Natural history and clinical features of the flail arm and flail leg ALS variants. Neurology 72, 1087–1094 (2009).
Burrell, J. R., Vucic, S. & Kiernan, M. C. Isolated bulbar phenotype of amyotrophic lateral sclerosis. Amyotroph. Lateral Scler. 12, 283–289 (2011).
Chiò, A. et al. Lower serum lipid levels are related to respiratory impairment in patients with ALS. Neurology 73, 1681–1685 (2009).
Dorst, J. et al. Patients with elevated triglyceride and cholesterol serum levels have a prolonged survival in amyotrophic lateral sclerosis. J. Neurol. 258, 613–617 (2011).
Sutedja, N. A. et al. Beneficial vascular risk profile is associated with amyotrophic lateral sclerosis. J. Neurol. Neurosurg. Psychiatry 82, 638–642 (2011).
Traynor, B. J. et al. Functional outcome measures as clinical trial endpoints in ALS. Neurology 63, 1933–1935 (2004).
Traynor, B. J., Alexander, M., Corr, B., Frost, E. & Hardiman, O. Effect of a multidisciplinary amyotrophic lateral sclerosis (ALS) clinic on ALS survival: a population based study, 1996–2000. J. Neurol. Neurosurg. Psychiatry 74, 1258–1261 (2003).
Van den Berg, J. P. et al. Multidisciplinary ALS care improves quality of life in patients with ALS. Neurology 65, 1264–1267 (2005).
Chiò, A. et al. Positive effects of tertiary centres for amyotrophic lateral sclerosis on outcome and use of hospital facilities. J. Neurol. Neurosurg. Psychiatry 77, 948–950 (2006).
Andersen, P. M. et al. Good practice in the management of amyotrophic lateral sclerosis: clinical guidelines. An evidence-based review with good practice points. EALSC Working Group. Amyotroph. Lateral Scler. 8, 195–213 (2007).
Miller, R. G. et al. Practice parameter update: The care of the patient with amyotrophic lateral sclerosis: multidisciplinary care, symptom management, and cognitive/behavioral impairment (an evidence-based review): report of the Quality Standards Subcommittee of the American Academy of Neurology. Neurology 73, 1227–1233 (2009).
Miller, R. G. et al. Practice parameter update: The care of the patient with amyotrophic lateral sclerosis: drug, nutritional, and respiratory therapies (an evidence-based review): report of the Quality Standards Subcommittee of the American Academy of Neurology. Neurology 73, 1218–1226 (2009).
Cudkowicz, M. E. et al. Toward more efficient clinical trials for amyotrophic lateral sclerosis. Amyotroph. Lateral Scler. 11, 259–265 (2010).
Lanka, V. & Cudkowicz, M. Therapy development for ALS: lessons learned and path forward. Amyotroph. Lateral Scler. 9, 131–140 (2008).
Miller, R. G., Mitchell, J. D., Lyon, M. & Moore, D. H. Riluzole for amyotrophic lateral sclerosis (ALS)/motor neuron disease (MND). Cochrane Database of Systematic Reviews, Issue 1. Art. No.: CD001447. doi:10.1002/14651858.CD001447.pub2 (2007).
Hardiman, O. Management of respiratory symptoms in ALS. J. Neurol. 258, 359–365 (2011).
Morgan, R. K. et al. Use of Sniff nasal-inspiratory force to predict survival in amyotrophic lateral sclerosis. Am. J. Respir. Crit. Care Med. 171, 269–274 (2005).
Bourke, S. C. et al. Effects of non-invasive ventilation on survival and quality of life in patients with amyotrophic lateral sclerosis: a randomised controlled trial. Lancet Neurol. 5, 140–147 (2006).
Mustfa, N. et al. The effect of noninvasive ventilation on ALS patients and their caregivers. Neurology 66, 1211–1217 (2006).
Pinto, S. & De Carvalho, M. Is a four-hour use of non-invasive ventilation enough to define compliance? Amyotroph. Lateral Scler. 11, 250–252 (2010).
Sancho, J., Servera, E., Díaz, J. & Marín, J. Efficacy of mechanical insufflation-exsufflation in medically stable patients with amyotrophic lateral sclerosis. Chest 125, 1400–1405 (2004).
Gilio, F. et al. Botulinum toxin type A for the treatment of sialorrhoea in amyotrophic lateral sclerosis: a clinical and neurophysiological study. Amyotroph. Lateral Scler. 11, 359–363 (2010).
Winterholler, M. G., Erbguth, F. J., Wolf, S. & Kat, S. Botulinum toxin for the treatment of sialorrhoea in ALS: serious side effects of a transductal approach. J. Neurol. Neurosurg. Psychiatry 70, 417–418 (2001).
Cooper-Knock, J., Ahmedzai, S. H. & Shaw, P. The use of subcutaneous glycopyrrolate in the management of sialorrhoea and facilitating the use of non-invasive ventilation in amyotrophic lateral sclerosis. Amyotroph. Lateral Scler. http://dx.doi.org/10.3109/17482968.2011.584195.
Neppelberg, E., Haugen, D. F., Thorsen, L. & Tysnes, O. B. Radiotherapy reduces sialorrhea in amyotrophic lateral sclerosis. Eur. J. Neurol. 14, 1373–1377 (2007).
Limousin, N. et al. Malnutrition at the time of diagnosis is associated with a shorter disease duration in ALS. J. Neurol. Sci. 297, 36–39 (2010).
Dupuis, L., Pradat, P. F., Ludolph, A. C. & Loeffler, J. P. Energy metabolism in amyotrophic lateral sclerosis. Lancet Neurol. 10, 75–82 (2011).
Kidney, D., Alexander, M., Corr, B., O'toole, O. & Hardiman, O. Oropharyngeal dysphagia in amyotrophic lateral sclerosis: neurological and dysphagia specific rating scales. Amyotroph. Lateral Scler. Other Motor Neuron Disord. 5, 150–153 (2004).
Thornton, F. J. et al. Amyotrophic lateral sclerosis: enteral nutrition provision—endoscopic or radiologic gastrostomy? Radiology 224, 713–717 (2002).
Chiò, A. et al. Percutaneous radiological gastrostomy: a safe and effective method of nutritional tube placement in advanced ALS. J. Neurol. Neurosurg. Psychiatry 75, 645–647 (2004).
Sancho, J. et al. Noninvasive respiratory muscle aids during PEG placement in ALS patients with severe ventilatory impairment. J. Neurol. Sci. 297, 55–59 (2010).
Bede, P. et al. Palliative care in amyotrophic lateral sclerosis: a review of current international guidelines and initiatives. J. Neurol. Neurosurg. Psychiatry 82, 413–418 (2011).
Neudert, C., Wasner, M. & Borasio, G. D. Patients' assessment of quality of life instruments: a randomised study of SIP, SF-36 and SEIQoL-DW in patients with amyotrophic lateral sclerosis. J. Neurol. Sci. 191, 103–109 (2001).
Jenkinson, C., Levvy, G., Fitzpatrick, R. & Garratt, A. The amyotrophic lateral sclerosis assessment questionnaire (ALSAQ-40): tests of data quality, score reliability and response rate in a survey of patients. J. Neurol. Sci. 180, 94–100 (2000).
Jenkinson, C., Fitzpatrick, R., Brennan, C. & Swash, M. Evidence for the validity and reliability of the ALS assessment questionnaire: the ALSAQ-40. Amyotroph. Lateral Scler. Other Motor Neuron Disord. 1, 33–40 (1999).
Jenkinson, C. & Fitzpatrick, R. Reduced item set for the amyotrophic lateral sclerosis assessment questionnaire: development and validation of the ALSAQ-5. J. Neurol. Neurosurg. Psychiatry 70, 70–73 (2001).
Simmons, Z. et al. The ALSSQOL: balancing physical and nonphysical factors in assessing quality of life in ALS. Neurology 67, 1659–1664 (2006).
Hardiman, O., Hickey, A. & O'Donerty, L. J. Physical decline and quality of life in amyotrophic lateral sclerosis. Amyotroph. Lateral Scler. Other Motor Neuron Disord. 5, 230–234 (2004).
De Groot, I. J., Post, M. W., Van Heuveln, T., Van den Berg, L. H. & Lindeman, E. Cross-sectional and longitudinal correlations between disease progression and different health-related quality of life domains in persons with amyotrophic lateral sclerosis. Amyotroph. Lateral Scler. 8, 356–361 (2007).
Felgoise, S. H. et al. The SEIQoL-DW for assessing quality of life in ALS: strengths and limitations. Amyotroph. Lateral Scler. 10, 456–462 (2009).
Robbins, R. A., Simmons, Z., Bremer, B. A., Walsh, S. M. & Fischer, S. Quality of life in ALS is maintained as physical function declines. Neurology 56, 442–444 (2001).
Cupp, J. et al. Psychological health in patients with ALS is maintained as physical function declines. Amyotroph. Lateral Scler. 12, 290–296 (2011).
Felgoise, S. H. et al. Psychological morbidity in ALS: the importance of psychological assessment beyond depression alone. Amyotroph. Lateral Scler. 11, 351–358 (2010).
Walsh, S. M., Bremer, B. A., Felgoise, S. H. & Simmons, Z. Religiousness is related to quality of life in patients with ALS. Neurology 60, 1527–1529 (2003).
Calvo, A. et al. Religiousness is positively associated with quality of life of ALS caregivers. Amyotroph. Lateral Scler. 12, 168–171 (2011).
Goldstein, L. H., Atkins, L., Landau, S., Brown, R. & Leigh, P. N. Predictors of psychological distress in carers of people with amyotrophic lateral sclerosis: a longitudinal study. Psychol. Med. 36, 865–875 (2006).
Gauthier, A. et al. A longitudinal study on quality of life and depression in ALS patient-caregiver couples. Neurology 68, 923–926 (2007).
Pagnini, F. et al. Existential well-being and spirituality of individuals with amyotrophic lateral sclerosis is related to psychological well-being of their caregivers. Amyotroph. Lateral Scler. 12, 105–108 (2011).
Pagnini, F. et al. Burden, depression, and anxiety in caregivers of people with amyotrophic lateral sclerosis. Psychol. Health Med. 15, 685–693 (2010).
Oliver, D. J. & Turner, M. R. Some difficult decisions in ALS/MND. Amyotroph. Lateral Scler. 11, 339–343 (2010).
Acknowledgements
O. Hardiman was funded by the European Union Health Research 7th Framework Program, 2007–2013, under grant agreement number 259,867, and by the Health Research Board Ireland.
Author information
Authors and Affiliations
Contributions
All authors contributed equally to the research of data, discussion of content, writing, and review and/or editing of the manuscript before submission.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Box
Supplementary Box (DOC 42 kb)
Rights and permissions
About this article
Cite this article
Hardiman, O., van den Berg, L. & Kiernan, M. Clinical diagnosis and management of amyotrophic lateral sclerosis. Nat Rev Neurol 7, 639–649 (2011). https://doi.org/10.1038/nrneurol.2011.153
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrneurol.2011.153
This article is cited by
-
Disruption of MAM integrity in mutant FUS oligodendroglial progenitors from hiPSCs
Acta Neuropathologica (2024)
-
Oxytosis/Ferroptosis in Neurodegeneration: the Underlying Role of Master Regulator Glutathione Peroxidase 4 (GPX4)
Molecular Neurobiology (2024)
-
Heterogeneity of cortical pTDP-43 inclusion morphologies in amyotrophic lateral sclerosis
Acta Neuropathologica Communications (2023)
-
Bone Marrow-Derived Mononuclear Cells in the Treatment of Neurological Diseases: Knowns and Unknowns
Cellular and Molecular Neurobiology (2023)
-
Glial Glutamine Homeostasis in Health and Disease
Neurochemical Research (2023)