Abstract
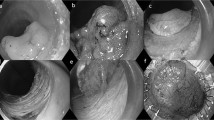
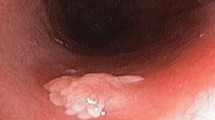
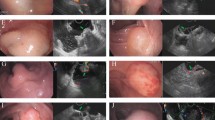
Survival rates for upper gastrointestinal cancers are poor and oesophageal cancer incidence is increasing. Upper gastrointestinal cancer is also often missed during examinations; a predicament that has not yet been sufficiently addressed. Improvements in the detection of premalignant lesions, early oesophageal and gastric cancers will enable organ-preserving endoscopic therapy, potentially reducing the number of advanced upper gastrointestinal cancers and resulting in improved prognosis. Japan is a world leader in high-quality diagnostic upper gastrointestinal endoscopy and the clinical routine in this country differs substantially from Western practice. In this Perspectives article, we review lessons learnt from Japanese gastroscopy technique, training and screening for risk stratification. We suggest a key performance indicator for upper gastrointestinal endoscopy with a minimum total procedure time of 8 min, and examine how quality assurance concepts in bowel cancer screening in the UK could be applied to upper gastrointestinal endoscopy and improve clinical practice.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Ferlay, J. S. et al. GLOBOCAN 2012 v1.0. Cancer Incidence and Mortality Worldwide: IARC CancerBase No. 11. Lyon, France: International Agency for Research on Cancer [online], (2013).
Cancer Research UK. Oesophageal cancer statistics [online], (2014).
Amin, A. et al. Gastric adenocarcinoma missed at endoscopy. J. R. Coll. Surg. Edinb. 47, 681–684 (2002).
Bloomfeld, R. S., Bridgers, D. I. 3rd & Pineau, B. C. Sensitivity of upper endoscopy in diagnosing esophageal cancer. Dysphagia 20, 278–282 (2005).
Khalil, Q., Gopalswamy, N. & Agrawal, S. Missed esophageal and gastric cancers after esophagogastroduodenoscopy in a midwestern military veteran population. South Med. J. 107, 225–228 (2014).
Raftopoulos, S. C., Segarajasingam, D. S., Burke, V., Ee, H. C. & Yusoff, I. F. A cohort study of missed and new cancers after esophagogastroduodenoscopy. Am. J. Gastroenterol. 105, 1292–1297 (2010).
Voutilainen, M. E. & Juhola, M. T. Evaluation of the diagnostic accuracy of gastroscopy to detect gastric tumours: clinicopathological features and prognosis of patients with gastric cancer missed on endoscopy. Eur. J. Gastroenterol. Hepatol. 17, 1345–1349 (2005).
Vradelis, S., Maynard, N., Warren, B. F., Keshav, S. & Travis, S. P. Quality control in upper gastrointestinal endoscopy: detection rates of gastric cancer in Oxford 2005–2008. Postgrad. Med. J. 87, 335–339 (2011).
Yalamarthi, S., Witherspoon, P., McCole, D. & Auld, C. D. Missed diagnoses in patients with upper gastrointestinal cancers. Endoscopy 36, 874–879 (2004).
Menon, S., Dhar, A. & Hoare, J. How commonly is gastric cancer missed at endoscopy: a UK primary care based-study [abstract OC-098]. Gut 61 (Suppl. 2), A43 (2012).
Menon, S. & Trudgill, N. How commonly is upper gastrointestinal cancer missed at endoscopy? A meta-analysis. Endosc. Int. Open 02, E46–E50 (2014).
Morris, E. J., Rutter, M. D., Finan, P. J., Thomas, J. D. & Valori, R. Post-colonoscopy colorectal cancer (PCCRC) rates vary considerably depending on the method used to calculate them: a retrospective observational population-based study of PCCRC in the English National Health Service. Gut http://dx.doi.org/10.1136/gutjnl-2014-308362.
Choi, K. S. et al. Performance of gastric cancer screening by endoscopy testing through the National Cancer Screening Program of Korea. Cancer Sci. 102, 1559–1564 (2011).
Hosokawa, O. et al. Difference in accuracy between gastroscopy and colonoscopy for detection of cancer. Hepatogastroenterology 54, 442–444 (2007).
Ren, W. et al. Missed diagnosis of early gastric cancer or high-grade intraepithelial neoplasia. World J. Gastroenterol. 19, 2092–2096 (2013).
Cho, Y. S. et al. Risk factors of developing interval early gastric cancer after negative endoscopy. Dig. Dis. Sci. 60, 936–943 (2015).
Gotoda, T., Iwasaki, M., Kusano, C., Seewald, S. & Oda, I. Endoscopic resection of early gastric cancer treated by guideline and expanded National Cancer Centre criteria. Br. J. Surg. 97, 868–871 (2010).
Pech, O. et al. Long-term results and risk factor analysis for recurrence after curative endoscopic therapy in 349 patients with high-grade intraepithelial neoplasia and mucosal adenocarcinoma in Barrett's oesophagus. Gut 57, 1200–1206 (2008).
Hamashima, C. et al. The Japanese guidelines for gastric cancer screening. Jpn J. Clin. Oncol. 38, 259–267 (2008).
Leung, W. K. et al. Screening for gastric cancer in Asia: current evidence and practice. Lancet Oncol. 9, 279–287 (2008).
Ichinose, M. et al. Current status of gastric cancer screening [Japanese]. Jap. J. Clin. Exp. Med. 87, 41–45 (2010).
Statistics and Information Department. Ministry of Health, Labour, and Welfare. National Reports on Cancer Screening Programs 2004; Tokyo: Health and Welfare Statistics Association 2006 [Japanese] [online], (2006).
The Japanese Society of Gastrointestinal Cancer Screening. The 2012 report [Japanese] [online], (2012).
Suzuki, H., Gotoda, T., Sasako, M. & Saito, D. Detection of early gastric cancer: misunderstanding the role of mass screening. Gastric Cancer 9, 315–319 (2006).
Japan Gastroenterological Endoscopy Society [online].
ASGE Standards of Practice Committee. Appropriate use of GI endoscopy. Gastrointest. Endosc. 75, 1127–1131 (2012).
ASGE Standards of Practice Committee. The role of endoscopy in the assessment and treatment of esophageal cancer. Gastrointest. Endosc. 77, 328–334 (2013).
ASGE Standards of Practice Committee. The role of endoscopy in Barrett's esophagus and other premalignant conditions of the esophagus. Gastrointest. Endosc. 76, 1087–1094 (2012).
NICE. Dyspepsia and gastro-oesophageal reflux disease [online], (2014).
JAG. Formative DOPS assessment form —Diagnostic upper GI endoscopy [online], (2010).
JAG. DOPS grade descriptors—Diagnostic upper GI endoscopy [online], (2010).
JAG. DOPS grade descriptors—Diagnostic upper GI endoscopy [online], (2010).
ASGE Committee on Training. Esophagogastroduodenoscopy (EGD) Core Curriculum [online], (2004).
Dinis-Ribeiro, M. et al. Management of precancerous conditions and lesions in the stomach (MAPS): guideline from the European Society of Gastrointestinal Endoscopy (ESGE), European Helicobacter Study Group (EHSG), European Society of Pathology (ESP), and the Sociedade Portuguesa de Endoscopia Digestiva (SPED). Endoscopy 44, 74–94 (2012).
Kaye, P. V. et al. The clinical utility and diagnostic yield of routine gastric biopsies in the investigation of iron deficiency anemia: a case-control study. Am. J. Gastroenterol. 103, 2883–2889 (2008).
Rey, J. F., Lambert, R. & Committee, E. Q. A. ESGE recommendations for quality control in gastrointestinal endoscopy: guidelines for image documentation in upper and lower GI endoscopy. Endoscopy 33, 901–903 (2001).
Yao, K. The endoscopic diagnosis of early gastric cancer. Ann. Gastroenterol. 26, 11–22 (2013).
Levine, D. S., Blount, P. L., Rudolph, R. E. & Reid, B. J. Safety of a systematic endoscopic biopsy protocol in patients with Barrett's esophagus. Am. J. Gastroenterol. 95, 1152–1157 (2000).
Anagnostopoulos, G. K. et al. Barrett's esophagus specialist clinic: what difference can it make? Dis. Esophagus 19, 84–87 (2006).
Fujii, T. et al. Effectiveness of premedication with pronase for improving visibility during gastroendoscopy: a randomized controlled trial. Gastrointest. Endosc. 47, 382–387 (1998).
Bhandari, P. et al. Use of Gascon and Pronase either as a pre-endoscopic drink or as targeted endoscopic flushes to improve visibility during gastroscopy: a prospective, randomized, controlled, blinded trial. Scand. J. Gastroenterol. 45, 357–361 (2010).
Ezoe, Y. et al. Magnifying narrowband imaging is more accurate than conventional white-light imaging in diagnosis of gastric mucosal cancer. Gastroenterology 141, 2017–2025 (2011).
Muto, M. et al. Early detection of superficial squamous cell carcinoma in the head and neck region and esophagus by narrow band imaging: a multicenter randomized controlled trial. J. Clin. Oncol. 28, 1566–1572 (2010).
Kodashima, S. et al. Evaluation of a new image-enhanced endoscopic technology using band-limited light for detection of esophageal squamous cell carcinoma. Dig. Endosc. 26, 164–171 (2014).
Yoshida, N. et al. Ability of a novel blue laser imaging system for the diagnosis of colorectal polyps. Dig. Endosc. 26, 250–258 (2014).
Japanese Gastric Cancer Association. Japanese gastric cancer treatment guidelines 2010 (ver. 3). Gastric Cancer 14, 113–123 (2011).
Fitzgerald, R. C. et al. British Society of Gastroenterology guidelines on the diagnosis and management of Barrett's oesophagus. Gut 63, 7–42 (2014).
Benaglia, T., Sharples, L. D., Fitzgerald, R. C. & Lyratzopoulos, G. Health benefits and cost effectiveness of endoscopic and nonendoscopic cytosponge screening for Barrett's esophagus. Gastroenterology 144, 62–73 (2013).
Kadri, S. R. et al. Acceptability and accuracy of a non-endoscopic screening test for Barrett's oesophagus in primary care: cohort study. BMJ 341, c4372 (2010).
Sami, S. S. et al. A randomized comparative effectiveness trial of novel endoscopic techniques and approaches for Barrett's esophagus screening in the community. Am. J. Gastroenterol. 110, 148–58 (2014).
Shariff, M. K. et al. Randomized crossover study comparing efficacy of transnasal endoscopy with that of standard endoscopy to detect Barrett's esophagus. Gastrointest. Endosc. 75, 954–961 (2012).
Pech, O. et al. Prospective evaluation of the macroscopic types and location of early Barrett's neoplasia in 380 lesions. Endoscopy 39, 588–593 (2007).
Enestvedt, B. K. et al. Location, location, location: does early cancer in Barrett's esophagus have a preference? Gastrointest. Endosc. 78, 462–467 (2013).
Kariyawasam, V. C. et al. Circumferential location predicts the risk of high-grade dysplasia and early adenocarcinoma in short-segment Barrett's esophagus. Gastrointest. Endosc. 75, 938–944 (2012).
Shiozaki, H. et al. Endoscopic screening of early esophageal cancer with the Lugol dye method in patients with head and neck cancers. Cancer 66, 2068–2071 (1990).
Muto, M. et al. Association of multiple Lugol-voiding lesions with synchronous and metachronous esophageal squamous cell carcinoma in patients with head and neck cancer. Gastrointest. Endosc. 56, 517–521 (2002).
Ina, H., Shibuya, H., Ohashi, I. & Kitagawa, M. The frequency of a concomitant early esophageal cancer in male patients with oral and oropharyngeal cancer. Screening results using Lugol dye endoscopy. Cancer 73, 2038–2041 (1994).
Uedo, N., Yao, K. & Ishihara, R. Screening and treating intermediate lesions to prevent gastric cancer. Gastroenterol. Clin. North Am. 42, 317–335 (2013).
Uemura, N. et al. Helicobacter pylori infection and the development of gastric cancer. N. Engl. J. Med. 345, 784–789 (2001).
Correa, P., Piazuelo, M. B. & Wilson, K. T. Pathology of gastric intestinal metaplasia: clinical implications. Am. J. Gastroenterol. 105, 493–498 (2010).
Uedo, N. Do we need multiple biopsies for assessing gastric cancer risk? Dig. Dis. Sci. 56, 926–928 (2011).
Whiting, J. L., Sigurdsson, A., Rowlands, D. C., Hallissey, M. T. & Fielding, J. W. The long term results of endoscopic surveillance of premalignant gastric lesions. Gut 50, 378–381 (2002).
Miki, K. et al. Usefulness of gastric cancer screening using the serum pepsinogen test method. Am. J. Gastroenterol. 98, 735–739 (2003).
Watabe, H. et al. Predicting the development of gastric cancer from combining Helicobacter pylori antibodies and serum pepsinogen status: a prospective endoscopic cohort study. Gut 54, 764–768 (2005).
Terasawa, T. et al. Prediction of gastric cancer development by serum pepsinogen test and Helicobacter pylori seropositivity in Eastern Asians: a systematic review and meta-analysis. PLoS ONE 9, e109783 (2014).
Corley, D. A. et al. Adenoma detection rate and risk of colorectal cancer and death. N. Engl. J. Med. 370, 1298–1306 (2014).
Logan, R. F. et al. Outcomes of the Bowel Cancer Screening Programme (BCSP) in England after the first 1 million tests. Gut 61, 1439–1446 (2012).
Bowles, C. J. et al. A prospective study of colonoscopy practice in the UK today: are we adequately prepared for national colorectal cancer screening tomorrow? Gut 53, 277–283 (2004).
Gavin, D. R. et al. The national colonoscopy audit: a nationwide assessment of the quality and safety of colonoscopy in the UK. Gut 62, 242–249 (2013).
McLean, M. H. & El-Omar, E. M. Genetics of gastric cancer. Nat. Rev. Gastroenterol. Hepatol. 11, 664–674 (2014).
ASGE/ACG Task Force on Quality in Endoscopy. Quality indicators for GI endoscopic procedures. Gastrointest. Endosc. 81, 1–80 (2015).
Gupta, N. et al. Longer inspection time is associated with increased detection of high-grade dysplasia and esophageal adenocarcinoma in Barrett's esophagus. Gastrointest. Endosc. 76, 531–538 (2012).
Matsui, D. Evaluation of optimal amount of barium meal for gastroradiography [Japanese]. Tohoku J. Radiol. Technol. 17, 114–115 (2008).
Teh, J. L. et al. Longer examination time improves detection of gastric cancer during diagnostic upper gastrointestinal endoscopy. Clin. Gastroenterol. Hepatol. 13, 480–487 (2015).
Lee, T. J. et al. Longer mean colonoscopy withdrawal time is associated with increased adenoma detection: evidence from the Bowel Cancer Screening Programme in England. Endoscopy 45, 20–26 (2013).
Butterly, L. et al. Serrated and adenomatous polyp detection increases with longer withdrawal time: results from the New Hampshire Colonoscopy Registry. Am. J. Gastroenterol. 109, 417–426 (2014).
Yao, K. Development of e-learning system for endoscopic diagnosis of gastric cancer: an international multicenter trial: Global e-Endo Study Team (GEST) [online], (2012).
Author information
Authors and Affiliations
Contributions
The authors contributed equally to all aspects of this manuscript.
Corresponding author
Ethics declarations
Competing interests
J.E.E has been an advisory board member and received research support from Cosmo Pharmaceuticals and honoraria for speaking for Abbvie and Olympus. The other authors declare no competing interests.
Rights and permissions
About this article
Cite this article
Veitch, A., Uedo, N., Yao, K. et al. Optimizing early upper gastrointestinal cancer detection at endoscopy. Nat Rev Gastroenterol Hepatol 12, 660–667 (2015). https://doi.org/10.1038/nrgastro.2015.128
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrgastro.2015.128
This article is cited by
-
The value of machine learning approaches in the diagnosis of early gastric cancer: a systematic review and meta-analysis
World Journal of Surgical Oncology (2024)
-
Satisfaction in population-based cancer screening in a Chinese rural high-risk population: the Yangzhong early diagnosis and treatment of upper gastrointestinal cancer
BMC Health Services Research (2022)
-
Long-term high-risk drinking does not change effective doses of propofol for successful insertion of gastroscope in Chinese male patients
BMC Anesthesiology (2022)
-
A prospective randomized controlled trial comparing simethicone, N-acetylcysteine, sodium bicarbonate and peppermint for visualization in upper gastrointestinal endoscopy
Surgical Endoscopy (2021)
-
Exosomal miR-1246 in serum as a potential biomarker for early diagnosis of gastric cancer
International Journal of Clinical Oncology (2020)