Key Points
-
Idiopathic pulmonary fibrosis (IPF) is the most common form of idiopathic interstitial pneumonia (IIP), a group of diseases of unknown cause that scar predominantly the gas-exchanging regions of the lungs — the alveoli. IPF is also the most lethal IIP, with a mortality rate which exceeds that of many cancers.
-
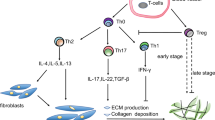
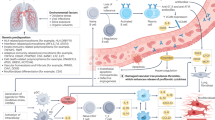
The current paradigm of pathogenesis is an aberrant healing response to epithelial cell injury, which probably occurs in response to a combination of environmental insults and/or genetic predispositions.
-
Prior to 2000, no studies of therapy had been undertaken that had provided clear evidence of efficacy.
-
More recent, better designed studies have generally been disappointing, although there is some evidence for benefit either with a combination of prednisone, azathioprine and N-acetylcysteine or with pirfenidone.
-
Novel initiatives that hold promise include the National Heart, Lung and Blood Institute's IPFnet, which aims to find effective therapy for early and late IPF; the use of expression arrays to identify pivotal targets; and the blockade of signalling pathways.
-
Future strategies need to focus on the cause and the consequences of the epithelial cell injury, without which the fibrogenic response would not occur. Further studies of the consequences of aberrant protein production by alveolar type II cells owing to genetic variants and other causes may prove fruitful.
-
Trial design, especially the issue of which end points are most meaningful, remains a challenge in IPF. However, the experiences of the past decade have resulted in considerable progress, particularly regarding the use of change in the lung volume measure, forced vital capacity, as a surrogate for the risk of mortality.
Abstract
Idiopathic pulmonary fibrosis (IPF) is the most common and most lethal diffuse fibrosing lung disease, with a mortality rate that exceeds that of many cancers. Recently, there have been many clinical trials of novel therapies for IPF. The results have mostly been disappointing, although two treatment approaches have shown some efficacy. This Review describes the difficulties of treating IPF and the approaches that have been tried or are in development, and concludes with suggestions of future therapeutic targets and strategies.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Crystal, R. G. & West, J. B. (eds) The Lung: Scientific Foundations (Raven, New York, 1991).
Westall, G. P., Stirling, R. G., Cullinan, P. C. & du Bois, R. M. Interstitial Lung Disease (eds King, T. J. & Schwarz, M. I.) 332–386 (BC Decker, Hamilton, Canada, 2003).
Vourlekis, J. S. et al. The effect of pulmonary fibrosis on survival in patients with hypersensitivity pneumonitis. Am. J. Med. 116, 662–668 (2004).
du Bois, R. M. Evolving concepts in the early and accurate diagnosis of idiopathic pulmonary fibrosis. Clin. Chest Med. 27, S17–S25 (2006).
Schwarz, M. I. & King, T. E. Jr (eds). Approach to the Evaluation and Diagnosis of Interstitial Lung Disease (BC Decker, Hamilton, Canada, 2003).
du Bois, R. M. Oxford Textbook of Medicine (eds Warrell, D. A., Cox, T. M. & Firth, J. D.) 1439–1446 (Oxford University Press, Oxford, 2003).
du Bois, R. M. & King, T. E. Jr. Challenges in pulmonary fibrosis x 5: the NSIP/UIP debate. Thorax 62, 1008–1012 (2007).
American Thoracic Society. American Thoracic Society/European Respiratory Society international multidisciplinary consensus classification of the idiopathic interstitial pneumonias. Am. J. Respir. Crit. Care Med. 165, 277–304 (2002). This classification began a more logical approach to diagnosing and researching diseases that affect the gas-exchanging regions of the lungs.
American Thoracic Society. Idiopathic pulmonary fibrosis: diagnosis and treatment. Am. J. Respir. Crit. Care Med. 161, 646–664 (2000). This statement clarified what constitutes IPF and how it is distinguishable from other diffuse fibrosing lung diseases and provided the platform for all the trials of novel therapy that ensued.
Olson, A. L. et al. Mortality from pulmonary fibrosis increased in the United States from 1992 to 2003. Am. J. Respir. Crit. Care Med. 176, 277–284 (2007). This paper provides the most accurate mortality data on IPF and emphasizes that the mortality from the disease is increasing.
Selman, M., King, T. E. & Pardo, A. Idiopathic pulmonary fibrosis: prevailing and evolving hypotheses about its pathogenesis and implications for therapy. Ann. Intern. Med. 134, 136–151 (2001). This paper stimulated more critical thinking about disease pathogenesis and highlighted an alternative paradigm to the traditional 'inflammation produces fibrosis' concept.
Iwai, K., Mori, T., Yamada, N., Yamaguchi, M. & Hosoda, Y. Idiopathic pulmonary fibrosis. Epidemiologic approaches to occupational exposure. Am. J. Respir. Crit. Care Med. 150, 670–675 (1994).
Hubbard, R., Lewis, S., Richards, K., Johnston, I. & Britton, J. Occupational exposure to metal or wood dust and aetiology of cryptogenic fibrosing alveolitis. Lancet 347, 284–289 (1996).
Steele, M. P. et al. Clinical and pathologic features of familial interstitial pneumonia. Am. J. Respir. Crit. Care Med. 172, 1146–1152 (2005).
Kuwano, K. et al. Detection of adenovirus E1A DNA in pulmonary fibrosis using nested polymerase chain reaction [see comments]. Eur. Resp. J. 10, 1445–1449 (1997).
Ueda, T. et al. Idiopathic pulmonary fibrosis and high prevalence of serum antibodies to hepatitis C virus. Am. Rev. Respir. Dis. 146, 266–268 (1992).
Tobin, R. W. et al. Increased prevalence of gastroesophageal reflux in patients with idiopathic pulmonary fibrosis. Am. J. Respir. Crit. Care Med. 158, 1804–1808 (1998).
Raghu, G., Yang, S. T., Spada, C., Hayes, J. & Pellegrini, C. A. Sole treatment of acid gastroesophageal reflux in idiopathic pulmonary fibrosis: a case series. Chest 129, 794–800 (2006).
Chambers, R. C. Procoagulant signalling mechanisms in lung inflammation and fibrosis: novel opportunities for pharmacological intervention? Br. J. Pharmacol. 153, S367–S378 (2008).
Selman, M. & Pardo, A. Role of epithelial cells in idiopathic pulmonary fibrosis: from innocent targets to serial killers. Proc. Am. Thorac. Soc. 3, 364–372 (2006).
Thomas, A. Q. et al. Heterozygosity for a surfactant protein C gene mutation associated with usual interstitial pneumonitis and cellular nonspecific interstitial pneumonitis in one kindred. Am. J. Respir. Crit. Care Med. 165, 1322–1328 (2002).
Wang, Y. et al. Genetic defects in surfactant protein A2 are associated with pulmonary fibrosis and lung cancer. Am. J. Hum. Genet. 84, 52–59 (2009).
Kaser, A. & Blumberg, R. S. Endoplasmic reticulum stress in the intestinal epithelium and inflammatory bowel disease. Semin. Immunol. 21, 156–163 (2009).
Xu, C., Bailly-Maitre, B. & Reed, J. C. Endoplasmic reticulum stress: cell life and death decisions. J. Clin. Invest. 115, 2656–2664 (2005).
Armanios, M. Y. et al. Telomerase mutations in families with idiopathic pulmonary fibrosis. N. Engl. J. Med. 356, 1317–1326 (2007).
Cronkhite, J. T. et al. Telomere shortening in familial and sporadic pulmonary fibrosis. Am. J. Respir. Crit. Care Med. 178, 729–737 (2008).
Alder, J. K. et al. Short telomeres are a risk factor for idiopathic pulmonary fibrosis. Proc. Natl Acad. Sci. USA 105, 13051–13056 (2008).
du Bois, R. M. Genetic factors in pulmonary fibrotic disorders. Semin. Respir. Crit. Care Med. 27, 581–588 (2006).
Laurent, G. J., Chambers, R. C., Hill, M. R. & McAnulty, R. J. Regulation of matrix turnover: fibroblasts, forces, factors and fibrosis. Biochem. Soc. Trans. 35, 647–651 (2007).
Davies, H. R. & Richeldi, L. Idiopathic pulmonary fibrosis: current and future treatment options. Am. J. Respir. Med. 1, 211–224 (2002).
Richeldi, L., Davies, H. R., Ferrara, G. & Franco, F. Corticosteroids for idiopathic pulmonary fibrosis. Cochrane Database Syst. Rev. CD002880 (2003).
Davies, H. R., Richeldi, L. & Walters, E. H. Immunomodulatory agents for idiopathic pulmonary fibrosis. Cochrane Database Syst. Rev. CD003134 (2003).
Raghu, G. et al. Azathioprine combined with prednisolone in the treatment of idiopathic pulmonary fibrosis: a prospective, double-blind, randomized, placebo-controlled clinical trial. Am. Rev. Respir. Dis. 144, 291–296 (1991).
Douglas, W. W. et al. Colchicine versus prednisone in the treatment of idiopathic pulmonary fibrosis. A randomized prospective study. Am. J. Respir. Crit. Care Med. 158, 220–225 (1998).
Ziesche, R., Hofbauer, E., Wittmann, K., Petkov, V. & Block, L. H. A preliminary study of long-term treatment with interferon gamma-1b and low-dose prednisolone in patients with idiopathic pulmonary fibrosis [see comments]. N. Engl. J. Med. 341, 1264–1269 (1999).
Nathan, S. D. et al. Interferon gamma-1b as therapy for idiopathic pulmonary fibrosis: an intrapatient analysis. Respiration 71, 77–82 (2004).
Antoniou, K. M. et al. Long-term clinical effects of interferon gamma-1b and colchicine in idiopathic pulmonary fibrosis. Eur. Respir. J. 28, 496–504 (2006).
Raghu, G. et al. A placebo-controlled trial of interferon gamma-1b in patients with idiopathic pulmonary fibrosis. N. Engl. J. Med. 350, 125–133 (2004).
King, T. E. Jr et al. Effect of interferon gamma-1b on survival in patients with idiopathic pulmonary fibrosis (INSPIRE): a multicentre, randomised, placebo-controlled trial. Lancet 374, 222–228 (2009). The largest study in IPF reported to date which, although negative, showed that large studies can be done in rare lung diseases.
King, T. E. Jr et al. BUILD-1: a randomized placebo-controlled trial of bosentan in idiopathic pulmonary fibrosis. Am. J. Respir. Crit. Care Med. 177, 75–81 (2008).
Raghu, G., Johnson, W. C., Lockhart, D. & Mageto, Y. Treatment of idiopathic pulmonary fibrosis with a new antifibrotic agent, pirfenidone: results of a prospective, open-label Phase II study. Am. J. Respir. Crit. Care Med. 159, 1061–1069 (1999).
Azuma, A. et al. Double-blind, placebo-controlled trial of pirfenidone in patients with idiopathic pulmonary fibrosis. Am. J. Respir. Crit. Care Med. 171, 1040–1047 (2005).
Demedts, M. et al. High-dose acetylcysteine in idiopathic pulmonary fibrosis. N. Engl. J. Med. 353, 2229–2242 (2005). Refs 42 and 43 are the only two published studies that show a benefit of treatment in IPF.
Hoyles, R. K. et al. A multicenter, prospective, randomized, double-blind, placebo-controlled trial of corticosteroids and intravenous cyclophosphamide followed by oral azathioprine for the treatment of pulmonary fibrosis in scleroderma. Arthritis Rheum. 54, 3962–3970 (2006).
Tashkin, D. P. et al. Effects of 1-year treatment with cyclophosphamide on outcomes at 2 years in scleroderma lung disease. Am. J. Respir. Crit. Care Med. 176, 1026–1034 (2007).
Tashkin, D. P. et al. Cyclophosphamide versus placebo in scleroderma lung disease. N. Engl. J. Med. 354, 2655–2666 (2006).
Raghu, G. et al. Treatment of idiopathic pulmonary fibrosis with etanercept: an exploratory, placebo-controlled trial. Am. J. Respir. Crit. Care Med. 178, 948–955 (2008).
Kubo, H. et al. Anticoagulant therapy for idiopathic pulmonary fibrosis. Chest 128, 1475–1482 (2005).
Moeller, A., Ask, K., Warburton, D., Gauldie, J. & Kolb, M. The bleomycin animal model: a useful tool to investigate treatment options for idiopathic pulmonary fibrosis? Int. J. Biochem. Cell Biol. 40, 362–382 (2008).
Selman, M. et al. Gene expression profiles distinguish idiopathic pulmonary fibrosis from hypersensitivity pneumonitis. Am. J. Respir. Crit. Care Med. 173, 188–198 (2006).
Yang, I. V. et al. Gene expression profiling of familial and sporadic interstitial pneumonia. Am. J. Respir. Crit. Care Med. 175, 45–54 (2007). This study used gene expression analysis to distinguish familial from sporadic fibrotic lung disease, an approach that might facilitate more refined disease classification.
Konishi, K. et al. Gene expression profiles of acute exacerbations of idiopathic pulmonary fibrosis. Am. J. Respir. Crit. Care Med. 180, 167–175 (2009).
Boon, K. et al. Molecular phenotypes distinguish patients with relatively stable from progressive idiopathic pulmonary fibrosis (IPF). PLoS ONE. 4, e5134 (2009).
Pardo, A. et al. Up-regulation and profibrotic role of osteopontin in human idiopathic pulmonary fibrosis. PLoS Med. 2, e251 (2005).
Chaudhary, N. I. et al. Inhibition of PDGF, VEGF and FGF signalling attenuates fibrosis. Eur. Respir. J. 29, 976–985 (2007).
Roth, G. J. et al. Design, synthesis, and evaluation of indolinones as triple angiokinase inhibitors and the discovery of a highly specific 6-methoxycarbonyl-substituted indolinone (BIBF 1120). J. Med. Chem. 52, 4466–4480 (2009).
Uhal, B. D. The role of apoptosis in pulmonary fibrosis. Eur. Respir. Rev. 17, 138–144 (2008).
Basseri, S., Lhotak, S., Sharma, A. M. & Austin, R. C. The chemical chaperone 4-phenylbutyrate inhibits adipogenesis by modulating the unfolded protein response. J. Lipid Res. 50, 2486–2501 (2009).
de Almeida, S. F. et al. Chemical chaperones reduce endoplasmic reticulum stress and prevent mutant HFE aggregate formation. J. Biol. Chem. 282, 27905–27912 (2007).
Hecker, L. et al. NADPH oxidase-4 mediates myofibroblast activation and fibrogenic responses to lung injury. Nature Med. 15, 1077–1081 (2009).
Lambeth, J. D. Nox enzymes, ROS, and chronic disease: an example of antagonistic pleiotropy. Free Radic. Biol. Med. 43, 332–347 (2007).
Lambeth, J. D., Krause, K. H. & Clark, R. A. NOX enzymes as novel targets for drug development. Semin. Immunopathol. 30, 339–363 (2008).
Kotani, I. et al. Increased procoagulant and antifibrinolytic activities in the lungs with idiopathic pulmonary fibrosis. Thromb. Res. 77, 493–504 (1995).
Belperio, J. A. et al. The role of the Th2 CC chemokine ligand CCL17 in pulmonary fibrosis. J. Immunol. 173, 4692–4698 (2004).
Keane, M. P., Arenberg, D. A., Moore, B. B., Addison, C. L. & Strieter, R. M. CXC chemokines and angiogenesis/angiostasis. Proc. Assoc. Am. Physicians 110, 288–296 (1998).
Moore, B. B., Keane, M. P., Addison, C. L., Arenberg, D. A. & Strieter, R. M. CXC chemokine modulation of angiogenesis: the importance of balance between angiogenic and angiostatic members of the family. J. Investig. Med. 46, 113–120 (1998).
Keane, M. P. et al. The CXC chemokines, IL-8 and IP-10, regulate angiogenic activity in idiopathic pulmonary fibrosis. J. Immunol. 159, 1437–1443 (1997).
Munger, J. S. et al. The integrin αvβ6 binds and activates latent TGF β1: a mechanism for regulating pulmonary inflammation and fibrosis. Cell 96, 319–328 (1999).
Horan, G. S. et al. Partial inhibition of integrin αvβ6 prevents pulmonary fibrosis without exacerbating inflammation. Am. J. Respir. Crit. Care Med. 177, 56–65 (2008).
Sheppard, D. Integrin-mediated activation of transforming growth factor-β1 in pulmonary fibrosis. Chest 120, S49–S53 (2001).
Mercer, P. F. et al. Pulmonary epithelium is a prominent source of proteinase-activated receptor-1-inducible CCL2 in pulmonary fibrosis. Am. J. Respir. Crit. Care Med. 179, 414–425 (2009).
Gharaee-Kermani, M., McCullumsmith, R. E., Charo, I. F., Kunkel, S. L. & Phan, S. H. CC-chemokine receptor 2 required for bleomycin-induced pulmonary fibrosis. Cytokine 24, 266–276 (2003).
Jiang, D. et al. Regulation of pulmonary fibrosis by chemokine receptor CXCR3. J. Clin. Invest. 114, 291–299 (2004).
Johnson, M. A. et al. Randomized controlled trial comparing prednisolone alone with cyclophosphamide and low dose prednisolone in combination in cryptogenic fibrosing alveolitis. Thorax 44, 280–288 (1989).
Tomioka, H. et al. A pilot study of aerosolized N-acetylcysteine for idiopathic pulmonary fibrosis. Respirology 10, 449–455 (2005).
Taniguchi, H. et al. Pirfenidone in idiopathic pulmonary fibrosis: a Phase III clinical trial in Japan. Eur. Resp. J. 8 Dec 2009 (doi: 10.1183/09031936.00005209).
Author information
Authors and Affiliations
Ethics declarations
Competing interests
R.M.d.B. has been a paid consultant, steering-committee member and/or co-chair of a steering committee for InterMune, Actelion, Centocor, Boehringer Ingelheim, Novartis, Genzyme and Mondobiotech.
Supplementary information
Supplementary information S1 (figure)
(PDF 335 kb)
Related links
Glossary
- Ground glass pattern
-
A radiological term for a hazy pattern of increased attenuation that is seen on computed tomography scans of the lung. It is caused by any infiltrate that partially excludes air from the alveoli.
- Usual interstitial pneumonia pattern
-
A histopathological term that denotes a patchy, temporally and anatomically heterogeneous pattern of disease that includes established fibrosis and less established disease with evidence of epithelial cell injury.
- Metalloproteinase
-
An enzyme that catalyses peptide hydrolysis and requires a metal for the enzymatic reaction.
- Apoptosis
-
Programmed cell death.
- Endoplasmic reticulum stress
-
A coordinated cellular response to aberrant protein folding and transport through the endoplasmic reticulum. It can occur following oxidant injury or abnormal protein synthesis and prevents intracellular protein accumulation. If unsuccessful, cell apoptosis is triggered.
- Telomere
-
Either end of a chromosome in a eukaryotic cell.
- Cochrane review
-
A review of medical publications on a particular therapy or group of therapies using defined selection and adjudication criteria, undertaken by volunteers for the Cochrane collaboration, to produce systematic reviews of the effects of health-care interventions.
- Alkylating nitrogen mustard
-
An agent that can donate an alkyl group to another molecule — such as DNA, which would inhibit cell division.
- Pulmonary arterial hypertension
-
A condition in which patients have a mean pulmonary artery pressure of greater than 25 mm Hg at rest or 30 mm Hg during exercise.
- 6-minute walk test
-
A test that measures the distance that a patient can walk quickly on a flat, hard surface in a period of 6 minutes. It provides a global index of the integrated response required for exercise but no organ-specific information.
- Honeycomb CT change
-
A computed tomographic pattern in which clusters of cysts are present at the lung periphery and extend contiguously into the adjacent, more central regions. They usually represent areas in which lung destruction is surrounded by established fibrosis.
- Vital capacity
-
A measure of lung volume that is the difference in lung size between a maximal inspiration and a maximal expiration.
- Gas exchange for carbon monoxide
-
An index of the lungs ability to move gas from the alveolar space into the vascular compartment. Abnormalities in this measure indicate a problem in the most peripheral regions of the lungs.
- Tyrosine kinase
-
An enzyme that can transfer a phosphate group from ATP to a tyrosine residue in a protein, often as part of an intracellular signalling process.
Rights and permissions
About this article
Cite this article
du Bois, R. Strategies for treating idiopathic pulmonary fibrosis. Nat Rev Drug Discov 9, 129–140 (2010). https://doi.org/10.1038/nrd2958
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrd2958
This article is cited by
-
The targeted delivery of chitosan nanoparticles to treat indoxacarb: induced lung fibrosis in rats
Bulletin of the National Research Centre (2022)
-
Cereblon contributes to the development of pulmonary fibrosis via inactivation of adenosine monophosphate-activated protein kinase α1
Experimental & Molecular Medicine (2021)
-
Caveolin-1 gene therapy inhibits inflammasome activation to protect from bleomycin-induced pulmonary fibrosis
Scientific Reports (2019)
-
Autophagic flux blockage in alveolar epithelial cells is essential in silica nanoparticle-induced pulmonary fibrosis
Cell Death & Disease (2019)
-
Characterization of CD28null T cells in idiopathic pulmonary fibrosis
Mucosal Immunology (2019)