Abstract
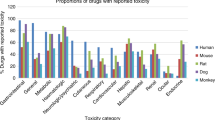
Selecting a pharmacologically relevant animal species for testing the safety and toxicity of novel monoclonal antibody (mAb) therapies to support clinical testing can be challenging. Frequently, the species of choice is the primate. With the increased number of mAbs in the pharmaceutical pipeline, this has significant implications for primate use, and so raises several important scientific, ethical and economic issues. Here, following a recent international workshop held to debate this topic, we discuss issues in the preclinical testing of mAbs, with a particular focus on species relevance and primate use, and provide suggestions for how these issues might be addressed.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout

Similar content being viewed by others
References
Douthwaite, J. & Jermutus, L. Exploiting directed evolution for the discovery of biologicals. Curr. Opin. Drug Discov. Dev. 8, 268–275 (2006).
Reichert, J. M. & Dewitz, M. C. Anti-infective monoclonal antibodies: perils and promise of development. Nature Rev. Drug Discov. 5, 191–195 (2006).
Reichert J. M., Rosensweig, C. J., Faden, L. B. & Dewitz, M. C. Monoclonal antibody successes in the clinic. Nature Biotechnol. 23, 1073–1078 (2005).
Reichert, J & Pavlou, A. Monoclonal antibodies market. Nature Rev. Drug Discov. 3, 383–384 (2004).
Suntharalingam, G. et al. Cytokine storm in a phase 1 trial of the anti-CD28 monoclonal antibody TGN1412. N. Engl. J. Med. 355, 1018–1028 (2006).
Nguyen, D. H., Hurtado-Ziola, N., Gagneux, P. & Varki, A. Loss of Siglec expression on T lymphocytes during human evolution. Proc. Natl Acad. Sci. USA 103, 7765–7770 (2006).
European Medicines Agency (EMEA). European Public Assessment Report for Remicade. EMEA web site [online], <http://www.emea.eu.int/humandocs/Humans/EPAR/remicade/remicade.htm>(2005).
Sharma, A. et al. Comparative pharmacodynamics of keliximab and clenoliximab in transgenic mice bearing human CD4. J. Pharmacol. Exp. Ther. 293, 33–41 (2000).
Clarke, J. et al. Evaluation of a surrogate antibody for preclinical safety testing of an anti-CD11a monoclonal antibody. Regul. Toxicol. Pharmacol. 40, 219–226 (2004).
Podolin, P. L., Webb, E. F., Reddy, M., Truneh, A. & Griswold, D. E. Inhibition of contact sensitivity in human CD4+ transgenic mice by human CD4-specific monoclonal antibodies: CD4+ T-cell depletion is not required. Immunolology 99, 287–295 (2000).
Herzyk, D. J. et al. Immunomodulatory effects of anti-CD4 antibody in host resistance against infections and tumors in human CD4 transgenic mice. Infect. Immun. 69, 1032–1043 (2001).
Yocum, D. E. et al. Clinical and immunological effects of a primatized anti-CD4 monoclonal antibody in active rheumatoid arthritis: Results of a phase I single dose, dose escalating trial. J. Rheumatol. 25, 1257–1262 (1998).
Kon, O. M. et al. The effects of an anti-CD4 monoclonal antibody, keliximab, on peripheral blood CD4+ T-cells in asthma. Eur. Respir. J. 18, 45–52 (2001).
Mould, D. R. et al. A population pharmacokinetic-pharmacodynamic analysis of single doses of clenoliximab in patients with rheumatoid arthritis. Clin. Pharmacol. Ther. 66, 246–257 (1999).
Bugelski, P.J. et al. Preclinical development of keliximab, a primatized anti-CD4 monoclonal antibody, in human CD4 transgenic mice: characterization of the model and safety studies. Hum. Exp. Toxicol. 19, 1–14 (2000).
Wu, B., Joshi, A., Ren, S. & Ng, C. The application of mechanism-based PK/PD modelling in pharmacodynamic-based dose selection of muM17, a surrogate monoclonal antibody for efalizumab. J. Pharm. Sci. 95, 1258–1268 (2006).
Food and Drug Administration (FDA). Pharmacology review of infliximab BLA. FDA web site [online], <http://www.fda.gov/cder/biologics/review/inflcen082498r4.pdf>(1998).
BioIndustry Association (BIA) & Association of the British Pharmaceutical Industry (ABPI). Early Stage Clinical Trial Taskforce: Joint ABPI/BIA report. ABPI web site [online], <http://www.abpi.org.uk/information/pdfs/BIAABPI_taskforce2.pdf>(2006).
European Medicines Agency (EMEA). European Public Assessment Report for Raptiva. EMEA web site [online], <http://www.emea.eu.int/humandocs/Humans/EPAR/raptiva/raptivaM.htm>(2004).
Shankar, G., Shores, E., Wagner, C. & Mire-Sluis, A. Scientific and regulatory considerations on the immunogenicity of biologics. Trends Biotechnol. 24, 274–280 (2006).
Committee for Medicinal Products for Human Use (CHMP). Concept paper on guideline on immunogenicity assessment of therapeutic proteins. EMEA web site [online], <http://www.emea.eu.int/pdfs/human/biosimilar/24651105en.pdf>(2005).
Snodin, D. J. & Ryle, P. R. Understanding and applying regulatory guidance on the nonclinical development of biotechnology-derived pharmaceuticals. BioDrugs 20, 25–52 (2006).
Schweiterman, W. Regulating biopharmaceuticals under CDER versus CBER: an insider's perspective. Drug Discov. Today 11, 945–951 (2006).
Robinson, S. et al. A European pharmaceutical industry initiative to challenge the requirement for conventional acute toxicity studies. Proceedings of the 9th FELASA symposium web site [online], <http://www.lal.org.uk/pdffiles/FELASA/Section1.pdf>(2006).
Acknowledgements
The National Centre for the Replacement, Refinement and Reduction of Animals in Research expresses its sincere thanks to all speakers and delegates at the workshop. Particular thanks to V. Robinson and P. Round for help in developing the concepts in this article.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Related links
Rights and permissions
About this article
Cite this article
Chapman, K., Pullen, N., Graham, M. et al. Preclinical safety testing of monoclonal antibodies: the significance of species relevance. Nat Rev Drug Discov 6, 120–126 (2007). https://doi.org/10.1038/nrd2242
Issue Date:
DOI: https://doi.org/10.1038/nrd2242
This article is cited by
-
Safety Profile Based on Concordance of Nonclinical Toxicity and Clinical Adverse Drug Reactions for Blood Cancer Drugs Approved in Japan
Drugs in R&D (2017)
-
Primate autoimmune disease models; lost for translation?
Clinical & Translational Immunology (2016)
-
Drug Development of Therapeutic Monoclonal Antibodies
BioDrugs (2016)
-
Muscle wasting associated with pathologic change is a risk factor for the exacerbation of joint swelling in collagen-induced arthritis in cynomolgus monkeys
BMC Musculoskeletal Disorders (2013)
-
Novel genetically-humanized mouse model established to evaluate efficacy of therapeutic agents to human interleukin-6 receptor
Scientific Reports (2013)