Abstract
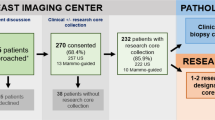
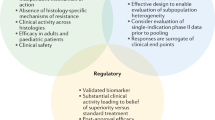
Increasingly, clinical trials incorporate translational research questions aimed at identifying biomarkers of response or resistance to agents under investigation. Biomarker assays can require tissue samples to be collected through a research biopsy before therapy, during treatment, or at the time of tumor progression. Such biopsy samples will generally not provide a direct benefit to the patient and, given the risks associated with any surgical procedure, ethical concerns have been raised when the participant's enrollment on a clinical trial depends on their consent to undergo a research biopsy. In this Perspectives article, we present the rationale for mandatory research biopsies and offer suggestions for standardization to ensure that high-quality, patient-centered, clinical trials continue to be designed with scientific and ethical rigor.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Mandrekar, S. J. & Sargent, D. J. Genomic advances and their impact on clinical trial design. Genome Med. 1, 69 (2009).
Mandrekar, S. J. & Sargent, D. J. Clinical trial designs for predictive biomarker validation: theoretical considerations and practical challenges. J. Clin. Oncol. 27, 4027–4034 (2009).
Mandrekar, S. J. & Sargent, D. J. Clinical trial designs for predictive biomarker validation: one size does not fit all. J. Biopharm. Stat. 19, 530–542 (2009).
Peppercorn, J. et al. Ethics of mandatory research biopsy for correlative end points within clinical trials in oncology. J. Clin. Oncol. 28, 2635–2640 (2010).
Slamon, D. J. et al. Use of chemotherapy plus a monoclonal antibody against HER2 for metastatic breast cancer that overexpresses HER2. N. Engl. J. Med. 344, 783–792 (2001).
Kwak, E. L. et al. Anaplastic lymphoma kinase inhibition in non-small-cell lung cancer. N. Engl. J. Med. 363, 1693–1703 (2010).
Tsao, M. S. et al. Erlotinib in lung cancer—molecular and clinical predictors of outcome. N. Engl. J. Med. 353, 133–144 (2005).
van Cutsem, E. et al. Cetuximab and chemotherapy as initial treatment for metastatic colorectal cancer. N. Engl. J. Med. 360, 1408–1417 (2009).
Demetri, G. D. et al. Efficacy and safety of imatinib mesylate in advanced gastrointestinal stromal tumors. N. Engl. J. Med. 347, 472–480 (2002).
Sørlie, T. et al. Gene expression patterns of breast carcinomas distinguish tumor subclasses with clinical implications. Proc. Natl Acad. Sci. USA 98, 10869–10874 (2001).
US National Library of Medicine. ClinicalTrials.gov [online].
Locatelli, M. A. et al. Should liver metastases of breast cancer be biopsied to improve treatment choice? J. Clin. Oncol. Annual Meeting Proceedings (Post-Meeting Edition). 28 (2010).
Berney, D. M., Montironi, R. & Egevad, L. Pathology in prostate research: optimizing tissue quality. Acta Oncol. 50 (Suppl. 1), 53–55 (2011).
Harris, L. N. et al. Predictors of resistance to preoperative trastuzumab and vinorelbine for HER2-positive early breast cancer. Clin. Cancer Res. 13, 1198–1207 (2007).
Krop, I. & Winer, E. Ten years of HER2-directed therapy: still questions after all these years. Breast Cancer Res. Treat. 113, 207–209 (2009).
Shapiro, G. et al. Phase I dose-escalation study of XL147, a PI3K inhibitor administered orally to patients with solid tumors [abstract 3500]. J. Clin. Oncol. 25 (2009).
Dowsett, M. et al. Prognostic value of Ki67 expression after short-term presurgical endocrine therapy for primary breast cancer. J. Natl Cancer Inst. 99, 167–170 (2007).
Winer, E. What can we learn? The scientific justification for consideration of mandatory research biopsy within some clinical trials. ASCO Annual Meeting (Chicago, IL, 2010).
Turke, A. B. et al. Preexistence and clonal selection of MET amplification in EGFR mutant NSCLC. Cancer Cell 17, 77–88 (2010).
US National Library of Medicine. ClinicalTrials.gov [online], (2010).
Brown, A., Wendler, D., Camphausen, K., Miller, F. & Citrin, D. Performing nondiagnostic research biopsies in irradiated tissue: a review of scientific, clinical, and ethical considerations. J. Clin. Oncol. 26, 3987–3994 (2008).
Dowlati, A. et al. Sequential tumor biopsies in early phase clinical trials of anticancer agents for pharmacodynamic evaluation. Clin. Cancer Res. 7, 2971–2976 (2001).
Agulnik, M., Oza, A., Pond, G. & Siu, L. Impact and perceptions of mandatory tumor biopsies for correlative studies in clinical trials of novel anticancer agents. J. Clin. Oncol. 24, 4801–4807 (2006).
Kim, E. et al. The BATTLE trial (Biomarker-integrated Approaches of Targeted Therapy for Lung Cancer Elimination): personalizing therapy for lung cancer [abstract LB-1]. Proc. 101st Annual Meeting of the AACR. (Washington, DC, 2010).
Nazarian, L. N. et al. Safety and efficacy of sonographically guided random core biopsy for diffuse liver disease. J. Ultrasound Med. 19, 537–541 (2000).
Helft, P. & Daugherty, C. Are we taking without giving in return? The ethics of research-related biopsies and the benefits of clinical trial participation. J. Clin. Oncol. 24, 4793–4795 (2006).
Cannistra, S. Performance of biopsies in clinical research. J. Clin. Oncol. 25, 1454–1455 (2007).
US Department of Health and Human Services. Protection of Human Subjects. Code Fed. Regul. Public Welfare. Title 45 (Section 46). (2005).
Weinfurt, K. P. et al. Expectations of benefit in early-phase clinical trials: implications for assessing the adequacy of informed consent. Med. Decis. Making 28, 575–581 (2008).
Wright, J. R. et al. Why cancer patients enter randomized clinical trials: exploring the factors that influence their decision. J. Clin. Oncol. 22, 4312–4318 (2004).
Catania, C. et al. Participation in clinical trials as viewed by the patient: understanding cultural and emotional aspects which influence choice. Oncology 74, 177–187 (2008).
Nurgat, Z. A. et al. Patient motivations surrounding participation in phase I and phase II clinical trials of cancer chemotherapy. Br. J. Cancer 92, 1001–1005 (2005).
Stadtmauer, E. A. et al. Conventional-dose chemotherapy compared with high-dose chemotherapy plus autologous hematopoietic stem-cell transplantation for metastatic breast cancer. Philadelphia Bone Marrow Transplant Group. N. Engl. J. Med. 342, 1069–1076 (2000).
Joffe, S. et al. Quality of informed consent in cancer clinical trials: a cross-sectional survey. Lancet 358, 1772–1777 (2001).
Flory, J. & Emanuel, E. Interventions to improve research participants'understanding in informed consent for research: a systematic review. JAMA 292, 1593–1601 (2004).
Katz, M. Are we asking too much? Ethical concerns over mandatory research biopsy: concerns over coercion. ASCO Annual Meeting (Chicago, IL, 2010).
Brown, R. F., Butow, P. N., Butt, D. G., Moore, A. R. & Tattersall, M. H. Developing ethical strategies to assist oncologists in seeking informed consent to cancer clinical trials. Soc. Sci. Med. 58, 379–390 (2004).
Joffe, S. & Weeks, J. C. Views of American oncologists about the purposes of clinical trials. J. Natl Cancer Inst. 94, 1847–1853 (2002).
Kerick, M., Timmermann, B. & Schweiger, M. R. [High-throughput sequencing of frozen and paraffin-embedded tumor and normal tissue]. Pathologe 31, 255–257 (2010).
Hillman, B. J. & Goldsmith, J. C. The uncritical use of high-tech medical imaging. N. Engl. J. Med. 363, 4–6 (2010).
Kuang, Y. et al. Noninvasive detection of EGFR T790M in gefitinib or erlotinib resistant non-small cell lung cancer. Clin. Cancer Res. 15, 2630–1636 (2009).
US National Library of Medicine. ClinicalTrials.gov [online].
US National Library of Medicine ClinicalTrials.gov [online].
Emanuel, E., Wendler, D. & Grady, C. What makes clinical research ethical? JAMA 283, 2701–2711 (2000).
Agrawal, M. & Emanuel, E. Ethics of phase 1 oncology studies: reexamining the arguments and data. JAMA 290, 1075–1082 (2003).
Author information
Authors and Affiliations
Contributions
All authors contributed to researching the data for the article, discussions of the content, writing the article and editing the manuscript before submission.
Corresponding author
Ethics declarations
Competing interests
All authors receive grant support from Genentech.
Rights and permissions
About this article
Cite this article
Olson, E., Lin, N., Krop, I. et al. The ethical use of mandatory research biopsies. Nat Rev Clin Oncol 8, 620–625 (2011). https://doi.org/10.1038/nrclinonc.2011.114
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrclinonc.2011.114
This article is cited by
-
Applications of liquid biopsy in the Pharmacological Audit Trail for anticancer drug development
Nature Reviews Clinical Oncology (2021)
-
Patients’ willingness to participate in clinical trials and their views on aspects of cancer research: results of a prospective patient survey
Trials (2016)
-
Implementing personalized cancer genomics in clinical trials
Nature Reviews Drug Discovery (2013)
-
Reporting practices of pharmacodynamic studies involving invasive research procedures in cancer trials
British Journal of Cancer (2013)
-
Prospective clinical experience with research biopsies in breast cancer patients
Breast Cancer Research and Treatment (2013)