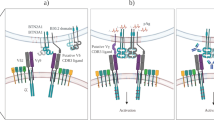
Abstract
Peripheral T-cell lymphomas (TCLs) are uncommon neoplasms, accounting for about 12% of all lymphoid tumors worldwide. TCLs in which γδ T-cell receptors are expressed (γδ TCLs) are extremely aggressive and rare (<1% of lymphoid neoplasms). γδ TCLs originate from γδ T cells, a small subset of peripheral T cells with direct antigen recognition capability acting at the interface between innate and adaptive immunity. Two distinct γδ TCL entities are recognized: hepatosplenic T-cell lymphoma (HSTL) and primary cutaneous γδ T-cell lymphoma (PCGD-TCL). HSTL is a well-characterized extranodal lymphoma that has a disguised onset, secondary to intrasinusoidal infiltration of the spleen, liver and bone marrow, has a rapidly progressive course that is poorly responsive to chemotherapy, and often ensues in the setting of immune system suppression. PCGD-TCL can present with prominent epidermal involvement or with a panniculitis-like clinical picture that can be complicated by a concurrent hemophagocytic syndrome; the disease shows biological and phenotypic overlap with other extranodal γδ TCLs that involve the respiratory or gastrointestinal tract mucosa. The regular application of phenotypic and molecular techniques is crucial for the diagnosis of γδ TCLs. In this Review, we discuss the clinical and biological features, the diagnostic challenges and the therapeutic perspectives of HSTL and PCGD-TCL.
Key Points
-
T-cell lymphomas that bear γδ T-cell receptors are heterogeneous biological entities that share a very poor prognosis irrespective of their site of origin
-
The actual incidence of γδ lymphomas is probably underestimated because of difficulties in the identification of γδ T-cell receptors in routine biopsy specimens
-
In young patients who present with hepatosplenomegaly, jaundice, altered liver function parameters, thrombocytopenia, and fever, hepatosplenic T-cell lymphoma should be included in the differential diagnosis
-
The response of γδ T-cell lymphomas to standard chemotherapy regimens is poor
-
Cytarabine combined with platinum-containing chemotherapies consolidated with stem cell transplantation seems to be the best available therapeutic strategy for eligible patients on diagnosis
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Armitage, J., Vose, J. & Weisenburger, D. International peripheral T-cell and natural killer/T-cell lymphoma study: pathology findings and clinical outcomes. J. Clin. Oncol. 26, 4124–4130 (2008).
Swerdlow, S. H et al. (eds) World Health Organization Classification of Tumors of Hematopoietic and Lymphoid Tissues (IARC, Lyon, 2008).
Piccaluga, P. P. et al. Gene expression analysis of peripheral T cell lymphoma, unspecified, reveals distinct profiles and new potential therapeutic targets. J. Clin. Invest. 117, 823–834 (2007).
Agostinelli, C. et al. Peripheral T cell lymphoma, not otherwise specified: the stuff of genes, dreams and therapies. J. Clin. Pathol. 61, 1160–1167 (2008).
Ciofani, M., Knowles, G. C., Wiest, D. L., von Boehmer, H. & Zúñiga-Pflücker, J. C. Stage-specific and differential Notch dependency at the αβ and γδ T lineage bifurcation. Immunity 25, 105–116 (2006).
Denning, S. M. & Haynes, B. F. Differentiation of human T cells. Clin. Lab. Med. 8, 1–14 (1988).
Havran, W. L. & Boismenu, R. Activation and function of γδ T cells. Curr. Opin. Immunol. 6, 442–446 (1994).
Sayers, T. J. et al. The restricted expression of granzyme M in human lymphocytes. J. Immunol. 166, 765–771 (2001).
Chattopadhyay, P. K. et al. The cytolytic enzymes granyzme A, granzyme B, and perforin: expression patterns, cell distribution, and their relationship to cell maturity and bright CD57 expression. J. Leukoc. Biol. 85, 88–97 (2009).
Inghirami, G., Zhu, B. Y., Chess, L. & Knowles, D. M. Flow cytometric and immunohistochemical characterization of the γ/δ T-lymphocyte population in normal human lymphoid tissue and peripheral blood. Am. J. Pathol. 136, 357–367 (1990).
Falini, B. et al. Distribution of T cells bearing different forms of the T cell receptor γ/δ in normal and pathological human tissues. J. Immunol. 143, 2480–2488 (1989).
Moser, B. & Eberl, M. γδ T cells: novel initiators of adaptive immunity. Immunol. Rev. 215, 89–102 (2007).
Groh, V., Steinle, A., Bauer S. & Spies, T. Recognition of stress-induced MHC molecules by intestinal epithelial γδ T cells. Science 279, 1737–1740 (1998).
Brandes, M., Willimann, K. & Moser, B. Professional antigen-presentation function by human γδ T cells. Science 309, 264–268 (2005).
Schild, H. et al. The nature of major histocompatibility complex recognition by γδ T cells. Cell 76, 29–37 (1994).
Chien, Y. H. & Konigshofer, Y. Antigen recognition by γδ T cells. Immunol. Rev. 215, 46–58 (2007).
Gong, G. et al. Phosphoantigen-activated Vγ 2Vδ 2 T cells antagonize IL-2-induced CD4+CD25+Foxp3+ T regulatory cells in mycobacterial infection. Blood 113, 837–845 (2009).
Shibata, K., Yamada, H., Hara, H., Kishihara, K. & Yoshikai, Y. Resident Vδ1+ γδ T cells control early infiltration of neutrophils after Escherichia coli infection via IL-17 production. J. Immunol. 178, 4466–4472 (2007).
Toth, B. et al. The role of γδ T cells in the regulation of neutrophil-mediated tissue damage after thermal injury. J. Leukoc. Biol. 76, 545–552 (2004).
Jensen, K. D. Thymic selection determines γδ T cell effector fate: antigen-naive cells make interleukin-17 and antigen-experienced cells make interferon γ. Immunity 29, 90–100 (2008).
Przybylski, G. K. et al. Hepatosplenic and subcutaneous panniculitis-like γ/δ T cell lymphomas are derived from different Vδ subsets of γ/δ T lymphocytes. J. Mol. Diagn. 2, 11–19 (2000).
Tey, S. K., Marlton, P. V., Hawley, C. M., Norris, D. & Gill, D. S. Post-transplant hepatosplenic T-cell lymphoma successfully treated with HyperCVAD regimen. Am. J. Hematol. 83, 330–333 (2008).
Khan, W. A. et al. Hepatosplenic γ/δ T-cell lymphoma in immunocompromised patients. Report of two cases and review of literature. Am. J. Clin. Pathol. 116, 41–50 (2001).
Knowles, D. M. et al. Correlative morphologic and molecular genetic analysis demonstrates three distinct categories of posttransplantation lymphoproliferative disorders. Blood 85, 552–565 (1995).
Vilmer, E. et al. Prominent expansion of circulating lymphocytes bearing γ T-cell receptors, with preferential expression of variable γ genes after allogeneic bone marrow transplantation. Blood 72, 841–849 (1988).
Kronenberg, M. & Havran, W. L. Frontline T cells: γδ T cells and intraepithelial lymphocytes. Immunol. Rev. 215, 5–7 (2007).
Ross, C. W., Schnitzer, B., Sheldon, S., Braun, D. K. & Hanson, C. A. γ/δ T-cell posttransplantation lymphoproliferative disorder primarily in the spleen. Am. J. Clin. Pathol. 102, 310–315 (1994).
Belhadj, K. et al. Hepatosplenic γδ T-cell lymphoma is a rare clinicopathologic entity with poor outcome: report on a series of 21 patients. Blood 102, 4261–4269 (2003).
Mackey, A. C., Green, L., Liang, L. C., Dinndorf, P. & Avigan, M. Hepatosplenic T cell lymphoma associated with infliximab use in young patients treated for inflammatory bowel disease. J. Pediatr. Gastroenterol. Nutr. 44, 265–267 (2007).
Shale, M., Kanfer, E., Panaccione, R. & Ghosh, S. Hepatosplenic T cell lymphoma in inflammatory bowel disease. Gut 57, 1639–1641 (2008).
US Food and Drug Administration The Adverse Event Reporting System (AERS): Latest Quarterly Data Files [online], (2009).
Toro, J. R. et al. γ δ T-cell lymphoma of the skin: a clinical, microscopic, and molecular study. Arch. Dermatol. 136, 1024–1032 (2000).
Arnulf, B. et al. Nonhepatosplenic γδ T-cell lymphoma: a subset of cytotoxic lymphomas with mucosal or skin localization. Blood 91, 1723–1731 (1998).
Farcet, J. P. et al. Hepatosplenic T-cell lymphoma: sinusal/sinusoidal localization of malignant cells expressing the T-cell receptor γ δ. Blood 75, 2213–2219 (1990).
Gaulard, P., Belhadj, K. & Reyes, F. γδ T-cell lymphomas. Semin. Hematol. 40, 233–243 (2003).
Falchook, G. S. et al. Hepatosplenic γ-δ T-cell lymphoma: clinicopathological features and treatment. Ann. Oncol. 20, 1080–1085 (2009).
Weidmann, E. Hepatosplenic T cell lymphoma. A review on 45 cases since the first report describing the disease as a distinct lymphoma entity in 1990. Leukemia 14, 991–997 (2000).
Florena, A. M., Iannitto, E., Quintini, G. & Franco, V. Bone marrow biopsy in hemophagocytic syndrome. Virchows Arch. 441, 335–344 (2002).
Burg, G. et al. A subcutaneous δ-positive T-cell lymphoma that produces interferon γ. N. Engl. J. Med. 325, 1078–1081 (1991).
Nosari, A. et al. Hepato-splenic γδ T-cell lymphoma: a rare entity mimicking the hemophagocytic syndrome. Am. J. Hematol. 60, 61–65 (1999).
Mazodier, K. et al. Severe imbalance of IL-18/IL-18BP in patients with secondary hemophagocytic syndrome. Blood 106, 3483–3489 (2005).
Wang, C. Y., Su, W. P. & Kurtin, P. J. Subcutaneous panniculitic T-cell lymphoma. Int. J. Dermatol. 35, 1–8 (1996).
Kumar, S. et al. Subcutaneous panniculitic T-cell lymphoma is a tumor of cytotoxic T lymphocytes. Hum. Pathol. 29, 397–403 (1998).
Toro, J. R. et al. γ-δ T-cell phenotype is associated with significantly decreased survival in cutaneous T-cell lymphoma. Blood 101, 3407–3412 (2003).
Willemze, R. et al. EORTC Cutaneous Lymphoma Group. Subcutaneous panniculitis-like T-cell lymphoma: definition, classification, and prognostic factors: an EORTC Cutaneous Lymphoma Group Study of 83 cases. Blood 111, 838–845 (2008).
Vega, F., Medeiros, L. J. & Gaulard, P. Hepatosplenic and other γδ T-cell lymphomas. Am. J. Clin. Pathol. 127, 869–880 (2007).
Salhany, K. E. et al. Hepatosplenic γδ T-cell lymphoma: ultrastructural, immunophenotypic, and functional evidence for cytotoxic T lymphocyte differentiation. Hum. Pathol. 28, 674–685 (1997).
Sallah, S. et al. γ/δ T-cell hepatosplenic lymphoma: review of the literature, diagnosis by flow cytometry and concomitant autoimmune hemolytic anemia. Ann. Hematol. 74, 139–142 (1997).
Falini, B. et al. T-lymphoblastic lymphomas expressing the non-disulfide-linked form of the T-cell receptor γ/δ: characterization with monoclonal antibodies and genotypic analysis. Blood 74, 2501–2507 (1989).
Ascani, S. et al. T-cell prolymphocytic leukaemia: does the expression of CD8+ phenotype justify the identification of a new subtype? Description of two cases and review of the literature. Ann. Oncol. 10, 649–653 (1999).
Massone, C. et al. Subcutaneous, blastic natural killer (NK), NK/T-cell, and other cytotoxic lymphomas of the skin: a morphologic, immunophenotypic, and molecular study of 50 patients. Am. J. Surg. Pathol. 28, 719–735 (2004).
Go, R. S. & Wester, S. M. Immunophenotypic and molecular features, clinical outcomes, treatments, and prognostic factors associated with subcutaneous panniculitis-like T-cell lymphoma: a systematic analysis of 156 patients reported in the literature. Cancer 101, 1404–1413 (2004).
Gaulard, P. et al. Expression of the α/β and γ/δ T-cell receptors in 57 cases of peripheral T-cell lymphomas. Identification of a subset of γ/δ T-cell lymphomas. Am. J. Pathol. 137, 617–628 (1990).
Cooke, C. B. et al. Hepatosplenic T-cell lymphoma: a distinct clinicopathologic entity of cytotoxic γ δ T-cell origin. Blood 88, 4265–4274 (1996).
de Wolf-Peeters, C. & Achten, R. γδ T-cell lymphomas: a homogeneous entity? Histopathology 36, 294–305 (2000).
Allory, Y. et al. Bone marrow involvement in lymphomas with hemophagocytic syndrome at presentation: a clinicopathologic study of 11 patients in a Western institution. Am. J. Surg. Pathol. 25, 865–874 (2001).
Gaulard, P. et al. Peripheral T-cell lymphoma presenting as predominant liver disease: a report of three cases. Hepatology 6, 864–868 (1986).
Florena, A. M. et al. Immunophenotypic profile and role of adhesion molecules in splenic marginal zone lymphoma with bone marrow involvement. Leuk. Lymphoma 47, 49–57 (2006).
Mastovich, S., Ratech, H., Ware, R. E., Moore, J. O. & Borowitz, M. J. Hepatosplenic T-cell lymphoma: an unusual case of a γ δ T-cell lymphoma with a blast-like terminal transformation. Hum. Pathol. 25, 102–108 (1994).
Vega, F. et al. Hepatosplenic γ/δ T-cell lymphoma in bone marrow. A sinusoidal neoplasm with blastic cytologic features. Am. J. Clin. Pathol. 116, 410–419 (2001).
Boulland, M. L., Kanavaros, P., Wechsler, J., Casiraghi, O. & Gaulard, P. Cytotoxic protein expression in natural killer cell lymphomas and in α β and γ δ peripheral T-cell lymphomas. J. Pathol. 183, 432–439 (1997).
Suarez, F. et al. Hepatosplenic αβ T-cell lymphoma: an unusual case with clinical, histologic, and cytogenetic features of γδ hepatosplenic T-cell lymphoma. Am. J. Surg. Pathol. 24, 1027–1032 (2000).
Macon, W. R. Hepatosplenic αβ T-cell lymphomas: a report of 14 cases and comparison with hepatosplenic γδ T-cell lymphomas. Am. J. Surg. Pathol. 25, 285–296 (2001).
Wang, C. C. et al. Consistent presence of isochromosome 7q in hepatosplenic T γ/δ lymphoma: a new cytogenetic-clinicopathologic entity. Genes Chromosomes Cancer 12, 161–164 (1995).
Wlodarska. I. Fluorescence in situ hybridization study of chromosome 7 aberrations in hepatosplenic T-cell lymphoma: isochromosome 7q as a common abnormality accumulating in forms with features of cytologic progression. Genes Chromosomes Cancer 33, 243–251 (2002).
Jonveaux, P., Daniel, M. T., Martel, V., Maarek, O. & Berger, R. Isochromosome 7q and trisomy 8 are consistent primary, non-random chromosomal abnormalities associated with hepatosplenic T γ/δ lymphoma. Leukemia 10, 1453–1455 (1996).
Tamaska, J. et al. Hepatosplenic γδ T-cell lymphoma with ring chromosome 7, an isochromosome 7q equivalent clonal chromosomal aberration. Virchows Arch. 449, 479–483 (2006).
Alonsozana, E. L. et al. Isochromosome 7q: the primary cytogenetic abnormality in hepatosplenic γδ T cell lymphoma. Leukemia 11, 1367–1372 (1997).
Miyazaki, K. et al. Gene expression profiling of peripheral T-cell lymphoma including γδ T-cell lymphoma. Blood 113, 1071–1074 (2009).
Aldinucci, D. et al. In vitro and in vivo effects of 2′-deoxycoformycin (Pentostatin) on tumour cells from human γδ+ T-cell malignancies. Br. J. Haematol. 110, 188–196 (2000).
Iannitto, E., Barbera, V., Quintini, G., Cirrincione, S. & Leone, M. Hepatosplenic γδ T-cell lymphoma: complete response induced by treatment with pentostatin. Br. J. Haematol. 117, 995–996 (2002).
Corazzelli, G. et al. Pentostatin (2′-deoxycoformycin) for the treatment of hepatosplenic γδ T-cell lymphomas. Haematologica 90, ECR14 (2005).
Macor, P. & Tedesco, F. Complement as effector system in cancer immunotherapy. Immunol. Lett. 111, 6–13 (2007).
Macor, P. et al. In vivo targeting of human neutralizing antibodies against CD55 and CD59 to lymphoma cells increases the antitumor activity of rituximab. Cancer Res. 67, 10556–10563 (2007).
Gallamini, A. et al. Alemtuzumab (Campath-1H) and CHOP chemotherapy as first-line treatment of peripheral T-cell lymphoma: results of a GITIL (Gruppo Italiano Terapie Innovative nei Linfomi) prospective multicenter trial. Blood 110, 2316–2323 (2007).
Mittal, S., Milner, B. J., Johnston, P. W. & Culligan, D. J. A case of hepatosplenic γ-δ T-cell lymphoma with a transient response to fludarabine and alemtuzumab. Eur. J. Haematol. 76, 531–534 (2006).
Jaeger, G. et al. Hepatosplenic γδ T-cell lymphoma successfully treated with a combination of alemtuzumab and cladribine. Ann. Oncol. 19, 1025–1026 (2008).
Ferrarini, M. et al. NF-κB modulates sensitivity to apoptosis, proinflammatory and migratory potential in short- versus long-term cultured human γ δ lymphocytes. J. Immunol. 181, 5857–5864 (2008).
Koenecke, C. et al. In vivo application of mAb directed against the γδ TCR does not deplete but generates “invisible” γδ T cells. Eur. J. Immunol. 39, 372–379 (2009).
Hathaway, T., Subtil, A., Kuo, P. & Foss, F. Efficacy of denileukin diftitox in subcutaneous panniculitis-like T-cell lymphoma. Clin. Lymphoma Myeloma 7, 541–545 (2007).
Vidulich, K., Jones, D. & Duvic, M. Cutaneous γ/δ T-cell lymphoma treated with radiation and denileukin diftitox. Clin. Lymphoma Myeloma 8, 55–58 (2008).
Bonder, C. S., Clark, S. R., Norman, M. U., Johnson, P & Kubes, P. Use of CD44 by CD4+ TH1 and TH2 lymphocytes to roll and adhere. Blood 107, 4798–4806 (2006).
Bonneville, M. & Scotet, E. Human Vγ9Vδ2 T cells: promising new leads for immunotherapy of infections and tumors. Curr. Opin. Immunol. 18, 539–546 (2006).
Alexander, A. A. et al. Isopentenyl pyrophosphate-activated CD56+ γδ T lymphocytes display potent antitumor activity toward human squamous cell carcinoma. Clin. Cancer Res. 14, 4232–4240 (2008).
Thedrez, A. et al. IL-21-mediated potentiation of antitumor cytolytic and proinflammatory responses of human Vγ 9V δ2 T cells for adoptive immunotherapy. J. Immunol. 182, 3423–3431 (2009).
Martinet, L. et al. A regulatory cross-talk between Vγ9Vδ2 T lymphocytes and mesenchymal stem cells. Eur. J. Immunol. 39, 752–762 (2009).
Henter, J. I., Elinder, G. & Ost, A. Diagnostic guidelines for hemophagocytic lymphohistiocytosis. The FHL Study Group of the Histiocyte Society. Semin. Oncol. 18, 29–33 (1991).
Acknowledgements
The authors are indebted to Dr. Mario Colombo of the IRCSS Istituto Nazionale dei Tumori, Milan, for helpful discussions. This manuscript was supported by grants from the Gruppo Italiano Studio Linfomi, Associazione Italiana per la Ricerca sul Cancro (Milan), BolognAIL (Bologna), Centro Interdipartimentale di Ricerca sul Cancro “G. Prodi” (Bologna), Fondazione Cassa di Risparmio in Bologna (Bologna).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Tripodo, C., Iannitto, E., Florena, A. et al. Gamma-delta T-cell lymphomas. Nat Rev Clin Oncol 6, 707–717 (2009). https://doi.org/10.1038/nrclinonc.2009.169
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrclinonc.2009.169
This article is cited by
-
Chimeric antigen receptor T cells for gamma–delta T cell malignancies
Leukemia (2022)
-
IRF4 drives clonal evolution and lineage choice in a zebrafish model of T-cell lymphoma
Nature Communications (2022)
-
Clinical, Diagnostic and Prognostic Characteristics of Primary Cutaneous Gamma Delta T-cell Lymphomas
Clinical Hematology International (2022)
-
γδ T cells: pleiotropic immune effectors with therapeutic potential in cancer
Nature Reviews Cancer (2019)