Key Points
-
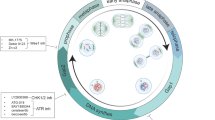
Human tumour cells are typically aneuploid, with both changes in chromosome number and structure, due to chromosome instability. One source of this instability is mitosis, during which errors in chromosome segregation can lead to gains or losses of chromosomes.
-
Members of the Aurora family of protein kinases are required for multiple aspects of mitosis. Aurora-A localizes to centrosomes/spindle poles and is required for spindle assembly, whereas Aurora-B is a chromosome passenger protein required for phosphorylation of histone H3, chromosome segregation and cytokinesis.
-
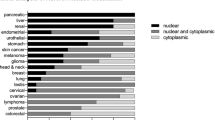
Aurora-A and -B are both overexpressed in a wide range of different human tumours. Aurora-A has also been shown to be an oncogene in in vitro transformation assays.
-
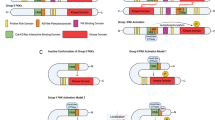
Three Aurora-kinase inhibitors have recently been described — ZM447439, Hesperadin and VX-680. All three induce similar phenotypes in cell-based assays and, interestingly, VX-680 shows antitumour activity in rodent xenograft models.
-
In mitosis, ZM447439 and Hesperadin both inhibit chromosome alignment and spindle-checkpoint function. RNAi-based experiments and overexpression of Aurora-kinase mutants indicate that these phenotypes seem to be due to inhibition of Aurora-B, not Aurora-A.
-
These drugs are not 'antimitotic' agents in that they do not directly inhibit cell-cycle progression. Rather, following an aberrant mitosis, activation of the p53-dependent post-mitotic checkpoint induces 'pseudo G1' cell-cycle arrest.
-
Although Aurora-A has received most of the attention so far in terms of a link with human cancer, Aurora-B might be the more suitable anticancer drug target, simply because inhibition of Aurora-B rapidly results in a catastrophic mitosis, which leads to cell death.
Abstract
Errors in mitosis can provide a source of the genomic instability that is typically associated with tumorigenesis. Many mitotic regulators are aberrantly expressed in tumour cells. These proteins could therefore make useful therapeutic targets. The kinases Aurora-A, -B and -C represent a family of such targets and several small-molecule inhibitors have been shown to block their function. Not only have these inhibitors advanced our understanding of mitosis, but, importantly, their in vivo antitumour activity has recently been reported. What have these studies taught us about the therapeutic potential of inhibiting this family of kinases?
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Lengauer, C., Kinzler, K. W. & Vogelstein, B. Genetic instabilities in human cancers. Nature 396, 643–649 (1998).
Adams, R. R., Carmena, M. & Earnshaw, W. C. Chromosomal passengers and the (aurora) ABCs of mitosis. Trends Cell Biol. 11, 49–54 (2001).
Carmena, M. & Earnshaw, W. C. The cellular geography of aurora kinases. Nature Rev. Mol. Cell Biol. 4, 842–854 (2003).
Nigg, E. A. Mitotic kinases as regulators of cell division and its checkpoints. Nature Rev. Mol. Cell Biol. 2, 21–32 (2001).
Bischoff, J. R. et al. A homologue of Drosophila aurora kinase is oncogenic and amplified in human colorectal cancers. EMBO J. 17, 3052–3065 (1998). Describes the identification of the mammalian Aurora-A and -B proteins, demonstrates that both are overexpressed in human tumours and that Aurora-A is an oncogene.
Sen, S., Zhou, H. & White, R. A. A putative serine/threonine kinase encoding gene BTAK on chromosome 20q13 is amplified and overexpressed in human breast cancer cell lines. Oncogene 14, 2195–2200 (1997).
Warner, S. L., Bearss, D. J., Han, H. & Von Hoff, D. D. Targeting Aurora-2 kinase in cancer. Mol. Cancer Ther. 2, 589–595 (2003).
Hauf, S. et al. The small molecule Hesperadin reveals a role for Aurora B in correcting kinetochore-microtubule attachment and in maintaining the spindle assembly checkpoint. J. Cell Biol. 161, 281–294 (2003). First description of Hesperadin, including a careful analysis showing how inhibition of Aurora-kinase activity in human cells results in syntelic orientation of chromosomes.
Ditchfield, C. et al. Aurora B couples chromosome alignment with anaphase by targeting BubR1, Mad2, and Cenp-E to kinetochores. J. Cell Biol. 161, 267–280 (2003). First description of ZM447439, including a careful analysis showing how inhibition of Aurora-kinase activity in human cells compromises the spindle checkpoint and induces endoreduplication in p53-deficient cells.
Harrington, E. A. et al. VX-680, a potent and selective small-molecule inhibitor of the Aurora kinases, suppresses tumor growth in vivo. Nature Med. 10, 262–267 (2004). First description of VX-680, including a demonstration of the antitumour activity of an Aurora-kinase inhibitor in rodent xenograft models.
Hirota, T. et al. Aurora-A and an interacting activator, the LIM protein Ajuba, are required for mitotic commitment in human cells. Cell. 114, 585–598 (2003).
Anand, S., PenrhynLowe, S. & Venkitaraman, A. R. AURORA-A amplification overrides the mitotic spindle assembly checkpoint, inducing resistance to Taxol. Cancer Cell 3, 51–62 (2003).
Meraldi, P., Honda, R. & Nigg, E. A. Aurora-A overexpression reveals tetraploidization as a major route to centrosome amplification in p53−/− cells. EMBO J. 21, 483–492 (2002). Many tumour cells, including ones with p53 deficiencies, have abnormal numbers of centrosomes. This paper shows that this is not necessarily because the mechanisms that control centrosome amplification are dysregulated, but because cytokinesis often fails when mitotic regulators, including Aurora-A, are overexpressed, leading to a tetraploid cell with four centrosomes.
Kimura, M., Matsuda, Y., Yoshioka, T. & Okano, Y. Cell cycle-dependent expression and centrosome localization of a third human aurora/Ipl1-related protein kinase, AIK3. J. Biol. Chem. 274, 7334–7340 (1999).
Li, X. et al. Direct association with inner centromere protein (INCENP) activates the novel chromosomal passenger protein, Aurora-C. J. Biol. Chem. 16 Aug 2004 (doi: 10.1074/jbc.M403029200).
Meraldi, P., Honda, R. & Nigg, E. A. Aurora kinases link chromosome segregation and cell division to cancer susceptibility. Curr. Opin. Genet. Dev. 14, 29–36 (2004).
Gassmann, R. et al. Borealin: a novel chromosomal passenger required for stability of the bipolar mitotic spindle. J. Cell Biol. 166, 179–191 (2004).
Gritsko, T. M. et al. Activation and overexpression of centrosome kinase BTAK/Aurora-A in human ovarian cancer. Clin. Cancer Res. 9, 1420–1426 (2003).
Katayama, H. et al. Mitotic kinase expression and colorectal cancer progression. J. Natl Cancer Inst. 91, 1160–1162 (1999).
Li, D. et al. Overexpression of oncogenic STK15/BTAK/Aurora A kinase in human pancreatic cancer. Clin. Cancer Res.. 9, 991–997 (2003).
Miyoshi, Y., Iwao, K., Egawa, C. & Noguchi, S. Association of centrosomal kinase STK15/BTAK mRNA expression with chromosomal instability in human breast cancers. Int. J. Cancer 92, 370–373 (2001).
Sakakura, C. et al. Tumour-amplified kinase BTAK is amplified and overexpressed in gastric cancers with possible involvement in aneuploid formation. Br. J. Cancer 84, 824–831 (2001).
Sen, S. et al. Amplification/overexpression of a mitotic kinase gene in human bladder cancer. J. Natl Cancer Inst. 94, 1320–1329 (2002).
Margolis, R. L., Lohez, O. D. & Andreassen, P. R. G1 tetraploidy checkpoint and the suppression of tumorigenesis. J. Cell. Biochem. 88, 673–683 (2003).
Tanaka, T. U. et al. Evidence that the Ipl1-Sli15 (Aurora kinase-INCENP) complex promotes chromosome bi-orientation by altering kinetochore-spindle pole connections. Cell 108, 317–329 (2002). Demonstrates that the budding yeast Aurora kinase Ipl1 destabilizes kinetochore–microtubule interactions that do not yield tension across the centromere.
Nicklas, R. B. How cells get the right chromosomes. Science 275, 632–637 (1997).
Musacchio, A. & Hardwick, K. G. The spindle checkpoint: structural insights into dynamic signalling. Nature Rev. Mol. Cell Biol. 3, 731–741 (2002).
Taylor, S. S., Scott, M. I. & Holland, A. J. The spindle checkpoint: a quality control mechanism which ensures accurate chromosome segregation. Chromosome Res. 12, 599–616 (2004).
Biggins, S. & Murray, A. W. The budding yeast protein kinase Ipl1/Aurora allows the absence of tension to activate the spindle checkpoint. Genes Dev. 15, 3118–3129 (2001). Demonstrates that the budding yeast Aurora kinase Ipl1 activates the spindle checkpoint in the absence of tension across the centromere.
Roghi, C. et al. The Xenopus protein kinase pEg2 associates with the centrosome in a cell cycle-dependent manner, binds to the spindle microtubules and is involved in bipolar mitotic spindle assembly. J. Cell Sci. 111, 557–572 (1998).
Glover, D. M., Leibowitz, M. H., McLean, D. A. & Parry, H. Mutations in aurora prevent centrosome separation leading to the formation of monopolar spindles. Cell 81, 95–105 (1995).
Marumoto, T. et al. Aurora-A kinase maintains the fidelity of early and late mitotic events in HeLa cells. J. Biol. Chem. 278, 51786–51795 (2003).
MurataHori, M. & Wang, Y. L. The kinase activity of aurora B is required for kinetochore-microtubule interactions during mitosis. Curr. Biol. 12, 894–899 (2002).
Kallio, M. J., McCleland, M. L., Stukenberg, P. T. & Gorbsky, G. J. Inhibition of aurora B kinase blocks chromosome segregation, overrides the spindle checkpoint, and perturbs microtubule dynamics in mitosis. Curr. Biol. 12, 900–905 (2002).
Carvalho, A., Carmena, M., Sambade, C., Earnshaw, W. C. & Wheatley, S. P. Survivin is required for stable checkpoint activation in taxol-treated HeLa cells. J. Cell Sci. 116, 14–98 (2003).
Lens, S. M. et al. Survivin is required for a sustained spindle checkpoint arrest in response to lack of tension. EMBO J. 22, 2934–2947 (2003). References 35 and 36 show that repression of the Aurora-B binding partner survivin results in a similar phenotype to that induced by Hesperadin and ZM447439.
Johnson, V. L., Scott, M. I., Holt, S. V., Hussein, D. & Taylor, S. S. Bub1 is required for kinetochore localization of BubR1, Cenp-E, Cenp-F and Mad2, and chromosome congression. J. Cell Sci. 117, 8–89 (2004).
Dutertre, S. & Prigent, C. Aurora-A overexpression leads to override of the microtubule-kinetochore attachment checkpoint. Mol. Interv. 3, 127–130 (2003).
Adams, R. R., Maiato, H., Earnshaw, W. C. & Carmena, M. Essential roles of Drosophila inner centromere protein (INCENP) and aurora B in histone H3 phosphorylation, metaphase chromosome alignment, kinetochore disjunction, and chromosome segregation. J. Cell Biol. 153, 865–880 (2001).
Giet, R. & Glover, D. M. Drosophila aurora B kinase is required for histone H3 phosphorylation and condensin recruitment during chromosome condensation and to organize the central spindle during cytokinesis. J. Cell Biol. 152, 669–682 (2001).
Terada, Y. et al. AIM-1: a mammalian midbody-associated protein required for cytokinesis. EMBO J. 17, 667–676 (1998).
Hamada, M. et al. Aurora2/BTAK/STK15 is involved in cell cycle checkpoint and cell survival of aggressive non-Hodgkin's lymphoma. Br. J. Haematol. 121, 439–447 (2003).
Zhou, H. et al. Tumour amplified kinase STK15/BTAK induces centrosome amplification, aneuploidy and transformation. Nature Genet. 20, 189–193 (1998). Reports that overexpression of Aurora-A in mouse fibroblasts induces centrosome duplication–distribution abnormalities, aneuploidy and transformation. Furthermore, overexpression of Aurora-A in human breast epithelial cells revealed similar centrosome abnormalities, as well as induction of aneuploidy.
EwartToland, A. et al. Identification of Stk6/STK15 as a candidate low-penetrance tumor-susceptibility gene in mouse and human. Nature Genet. 34, 403–412 (2003). Quantitative trait loci mapping of a locus that confers susceptibility to skin tumours in mice identifies Aurora-A. Comparative analysis of human samples identifies a polymorphism in Aurora-A.
Katayama, H. et al. Phosphorylation by aurora kinase A induces Mdm2-mediated destabilization and inhibition of p53. Nature Genet. 36, 55–62 (2004).
Walter, A. O., Seghezzi, W., Korver, W., Sheung, J. & Lees, E. The mitotic serine/threonine kinase Aurora2/AIK is regulated by phosphorylation and degradation. Oncogene 19, 4906–4916 (2000).
Sitnicka, E. et al. Key role of flt3 ligand in regulation of the common lymphoid progenitor but not in maintenance of the hematopoietic stem cell pool. Immunity 17, 463–472 (2002).
Mayer, T. U. et al. Small molecule inhibitor of mitotic spindle bipolarity identified in a phenotype-based screen. Science 286, 971–974 (1999).
Sakowicz, R. et al. Antitumor activity of a kinesin inhibitor. Cancer Res. 64, 3276–3280 (2004).
Giet, R. et al. Drosophila Aurora A kinase is required to localize D-TACC to centrosomes and to regulate astral microtubules. J. Cell Biol. 156, 437–451 (2002).
Conte, N. et al. TACC1–chTOG–Aurora A protein complex in breast cancer. Oncogene 22, 8102–8116 (2003).
Delaval, B. et al. Aurora B–TACC1 protein complex in cytokinesis. Oncogene 23, 4516–4522 (2004).
Acknowledgements
We would like to thank S. Green, D. Johnstone, D. Roberts, H. Robson and E. Brown (all from AstraZeneca) for helpful comments on the manuscript. We would also like to thank C. Ditchfield and the AstraZeneca Bioinformatics group for sharing unpublished data.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
Nicholas Keen is an employee of AstraZeneca UK Ltd, part of the AstraZeneca group of companies, and is also a shareholder in the holding company Astrazeneca PLC.
Related links
Related links
DATABASES
Entrez Gene
National Cancer Institute
FURTHER INFORMATION
Glossary
- MICROTUBULE
-
A hollow tube made of α- and β-tubulin subunits, which forms part of the interphase cytoskeleton and the mitotic spindle.
- CHECKPOINT
-
A surveillance mechanism that prevents cell-cycle progression in response to events that, if uncorrected, can result in genome damage.
- CENTROSOME
-
Microtubule-organizing centre typically found at the centre of the cell and from which the interphase microtubule cytoskeleton radiates. Following duplication in S phase, the two centrosomes form the spindle poles in mitosis.
- SPINDLE CHECKPOINT
-
A cell-cycle checkpoint that prevents anaphase until all the chromosomes are correctly bi-oriented on the mitotic-spindle apparatus.
- CYTOKINESIS
-
The process of cell division, which is normally coupled to mitosis.
- CHROMOSOME PASSENGER
-
A protein that localizes to the chromosomes, and more specifically the centromeres, in the early phases of mitosis, but then relocates to the spindle midzone following anaphase onset.
- ANAPHASE
-
Movement of the sister chromatids to opposite spindle poles following loss of sister-chromatid cohesion at the metaphase–anaphase transition.
- CENTROMERE
-
A specialized region of the chromosome, typically seen as a primary constriction, which assembles the two kinetochores and maintains sister-chromatid cohesion until the onset of anaphase.
- KINETOCHORE
-
Proteinacious structure that assembles at the centromere of each sister chromatid. During mitosis, kinetochores attach and move chromosomes on the microtubule spindle. When unattached, they activate the spindle checkpoint.
- POLYPLOID
-
Most normal cells have a 2N (diploid) DNA content. A polyploid cell carries multiple chromosomes, resulting from chromosome replication without nuclear or cell division.
- ENDOREDUPLICATION
-
The accumulation of multiple copies of a cell's DNA, which results when replication is not coordinated to the cell cycle.
- PACLITAXEL
-
A naturally occurring compound, originally purified from the pacific yew tree, that stabilizes microtubules and has antitumour activity.
- NOCODAZOLE
-
A microtubule toxin that prevents microtubule assembly.
- SECOND- OR THIRD-GENERATION INHIBITORS
-
Usually describes drugs that were modified from an original class of compounds, using feedback from clinical studies to address any toxicity issues or to optimize biological properties.
- THERAPEUTIC INDEX
-
The therapeutic index is a comparison of the amount of a medication that causes the therapeutic effect to the amount that causes toxic effects. A drug with a high therapeutic index produces its therapeutic effects at significantly lower doses than those where limiting toxicology is apparent.
Rights and permissions
About this article
Cite this article
Keen, N., Taylor, S. Aurora-kinase inhibitors as anticancer agents. Nat Rev Cancer 4, 927–936 (2004). https://doi.org/10.1038/nrc1502
Issue Date:
DOI: https://doi.org/10.1038/nrc1502
This article is cited by
-
Computational screening identifies depsidones as promising Aurora A kinase inhibitors: extra precision docking and molecular dynamics studies
Network Modeling Analysis in Health Informatics and Bioinformatics (2024)
-
SYT7 regulates the progression of chronic lymphocytic leukemia through interacting and regulating KNTC1
Biomarker Research (2023)
-
Computational completion of the Aurora interaction region of N-Myc in the Aurora a kinase complex
Scientific Reports (2023)
-
The genomic stability regulator PTIP is required for proper chromosome segregation in mitosis
Cell Division (2022)
-
The deubiquitinating enzyme complex BRISC regulates Aurora B activation via lysine-63-linked ubiquitination in mitosis
Communications Biology (2022)