Abstract
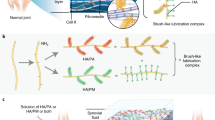
Lubrication is key for the efficient function of devices and tissues with moving surfaces, such as articulating joints, ocular surfaces and the lungs. Indeed, lubrication dysfunction leads to increased friction and degeneration of these systems. Here, we present a polymer–peptide surface coating platform to non-covalently bind hyaluronic acid (HA), a natural lubricant in the body. Tissue surfaces treated with the HA-binding system exhibited higher lubricity values, and in vivo were able to retain HA in the articular joint and to bind ocular tissue surfaces. Biomaterials-mediated strategies that locally bind and concentrate HA could provide physical and biological benefits when used to treat tissue-lubricating dysfunction and to coat medical devices.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Dorinson, A. & Ludema, K. C. Mechanics and Chemistry in Lubrication Ch. 1 (Elsevier, 1985).
Kato, K. Industrial tribology in the past and future. Tribology 6, 1–9 (2011).
Moghani, T., Butler, J. P. & Loring, S. H. Determinants of friction in soft elastohydrodynamic lubrication. J. Biomech. 42, 1069–1074 (2009).
Chawla, K., Ham, H. O., Nguyen, T. & Messersmith, P. B. Molecular resurfacing of cartilage with proteoglycan 4 (PRG4). Acta Biomater. 6, 3388–3394 (2010).
Flannery, C. R. et al. Prevention of cartilage degeneration in a rat model of osteoarthritis by intracellular treatment with recombinant lubricin. Arthritis Rheum. 60, 840–847 (2009).
McNary, S. M., Athanasiou, K. A. & Reddi, A. H. Engineering lubrication in articular cartilage. Tissue Eng. Part B Rev. 18, 88–100 (2012).
Sivan, S. et al. Liposomes act as effective biolubricants for friction reduction in human synovial joints. Langmuir 26, 1107–1116 (2010).
Hills, B. A. & Butler, B. D. Surfactants identified in synovial fluid and their ability to act as boundary lubricants. Ann. Rheum. Dis. 43, 641–648 (1984).
Schmidt, T. A., Gastelum, N. S., Nguyen, Q. T., Schumacher, B. L. & Sah, R. L. Boundary lubrication of articular cartilage: Role of synovial fluid constituents. Arthritis Rheum. 56, 882–891 (2007).
Neu, C. P., Komvopoulos, K. & Reddi, A. H. The interface of functional biotribology and regenerative medicine in synovial joints. Tissue Eng. Part B Rev. 14, 235–247 (2008).
Chen, M., Briscoe, W. H., Armes, S. P. & Klein, J. Lubrication at physiological pressures by polyzwitterionic brushes. Science 323, 1698–1701 (2009).
Schmidt, T. A. et al. Transcription, translation, and function of lubricin, a boundary lubricant, at the ocular surface. JAMA Ophthalmol. 131, 766–776 (2013).
Zmolik, J. M. & Mummert, M. E. Pep-1 as a novel probe for the in situ detection of hyaluronan. J. Histochem. Cytochem. 53, 745–751 (2005).
Mummert, M. E., Mohamadzadeh, M., Mummert, D. I., Mizumoto, N. & Takashima, A. Development of a peptide inhibitor of hyaluronan-mediated leukocyte trafficking. J. Exp. Med. 192, 769–780 (2000).
Tolg, C. et al. A RHAMM mimetic peptide blocks hyaluronan signaling and reduces inflammation and fibrogenesis and excisional skin wounds. Am. J. Pathol. 181, 1250–1270 (2012).
Zaleski, K. J. et al. Hyaluronic acid binding peptides prevent experimental staphylococcal wound infection. Antimicrob. Agents Chemother. 50, 3856–3860 (2006).
Yang, B., Zhang, L. & Turley, E. A. Identification of two hyaluronan-binding domains in the hyaluronan receptor RHAMM. J. Biol. Chem. 268, 8617–8623 (1993).
Jay, G. D., Torres, J. R., Warman, M. L., Laderer, M. C. & Breuer, K. S. The role of lubricin in the mechanical behavior of synovial fluid. Proc. Natl Acad. Sci. USA 104, 6194–6199 (2007).
Zhang, D., Johnson, L. J., Hsu, H. P. & Spector, M. Cartilaginous deposits in subchondral bone in regions of exposed bone in osteoarthritis of the human knee: Histomorphometric study of PRG4 distribution in osteoarthritic cartilage. J. Orthop. Res. 25, 873–883 (2007).
Presti, D. & Scott, J. E. Hyaluronan-mediated protective effect against cell damage caused by enzymatically produced hydroxyl (OH.) radicals is dependent on hyaluronan molecular mass. Cell Biochem. Funct. 12, 281–288 (1994).
Julovi, S. M., Yasuda, T., Shimizu, M., Hiramitsu, T. & Nakamura, T. Inhibition of interleukin-1β-stimulated production of matrix metalloproteinases by hyaluronan via CD44 in human articular cartilage. Arthritis Rheum. 50, 516–525 (2004).
Fraser, J. R. E., Laurent, T. C. & Laurent, U. B. G. Hyaluronan: Its nature, distribution, functions and turnover. J. Intern. Med. 242, 27–33, (1997).
Buckwalter, J. A. & Mankin, H. J. Articular cartilage. Part II: Degeneration and osteoarthrosis, repair, regeneration, and transplantation. J. Bone Joint Surg. Am. 79, 612–632 (1997).
Morrell, K. C., Hodge, W. A., Krebs, D. E. & Mann, R. W. Corroboration of in vivo cartilage pressures with implications for synovial joint tribology and osteoarthritis causation. Proc. Natl Acad. Sci. USA 102, 14819–14824 (2005).
Greene, G. W. et al. Adaptive mechanically controlled lubrication mechanism found in articular joints. Proc. Natl Acad. Sci. USA 108, 5255–5259 (2011).
Das, S. et al. Synergistic interactions between grafted hyaluronic acid and lubricin provide enhanced wear protection and lubrication. Biomacromolecules 14, 1669–1677 (2013).
Amiel, D. et al. Long-term effect of sodium hyaluronate (Hyalgan®) on osteoarthritis progression in a rabbit model. Osteoarthritis Cartilage 11, 636–643 (2003).
Yoshimi, T. et al. Effects of high-molecular-weight sodium hyaluronate on experimental osteoarthrosis induced by the resection of rabbit anterior cruciate ligament. Clin. Orthop. Relat. Res. 298, 296–304 (1994).
Elmory, S. et al. Chondroprotective effects of high-molecular-weight cross-linked hyaluronic acid in a rabbit knee osteoarthritis model. Osteoarthritis Cartilage 22, 121–127 (2014).
Yu, C-J. et al. Proteomic analysis of osteoarthritic chondrocyte reveals the hyaluronic acid regulated proteins involved in chondroprotective effect under oxidative stress. J. Proteomics 99, 40–53 (2014).
Moreland, L. W. Intra-articular hyaluronan (hyaluronic acid) and hylans for the treatment of osteoarthritis: Mechanisms of action. Arthritis Res. Ther. 5, 54–67 (2003).
Sharma, B., Williams, C. G., Khan, M., Manson, P. & Elisseeff, J .H. In vivo chondrogenesis of mesenchymal stem cells in photopolymerized hydrogel. Plast. Reconstr. Surg. 119, 112–120 (2007).
Jackson, D. W. & Simon, T. M. Intra-articular distribution and residence time of Hylan A and B: A study in the goat knee. Osteoarthritis Cartilage 14, 1248–1257 (2006).
Zhang, W. et al. OARSI recommendations for the management of hip and knee osteoarthritis: Part III: Changes in evidence following systematic cumulative update of research published through January 2009. Osteoarthritis Cartilage 18, 476–499 (2010).
Strauss, E. J., Hart, J. A., Miller, M. D., Altman, R. D. & Rosen, J. E. Hyaluronic acid viscosupplementation and osteoarthritis: Current uses and future directions. Am. J. Sports Med. 37, 1636–1644 (2009).
Brandt, K. D., Smith, G. N. & Simon, L. S. Intraarticular injection of hyaluronan as treatment for knee osteoarthritis: What is the evidence. Arthritis Rheum. 43, 1192–1203 (2000).
Sharma, B. et al. Human cartilage repair with a photoreactive adhesive-hydrogel composite. Sci. Transl. Med. 5, 167ra6 (2013).
Wang, D. A. et al. Multifunctional chondroitin sulphate for cartilage tissue-biomaterial integration. Nature Mater. 6, 385–392 (2007).
Messman, J. M., Lokitz, B. S., Pickel, J. M. & Kilbey, S. M. Highly tailorable materials based on 2-vinyl-4,4-dimethyl azlactone: (co)polymerization, synthetic manipulation and characterization. Macromolecules 42, 3933–3941 (2009).
Brown, T. J., Laurent, U. B. G. & Fraser, J. R. E. Turnover of hyaluronan in synovial joints: Elimination of labeled hyaluronan from the knee joint of the rabbit. Exp. Physiol. 76, 125–134 (1991).
Smith, G. N., Mickler, E. A., Myers, S. L. & Brandt, K. D. Effect of intraarticular hyaluronan injection on synovial fluid hyaluronan in the early stage of canine post-traumatic osteoarthritis. J. Rheum. 28, 1341–1346 (2001).
Caligaris, M., Canal, C. E., Ahmad, C. S., Gardner, T. R. & Ateshian, G. A. Investigation of the frictional response of osteoarthritic human tibiofemoral joints and the potential beneficial tribological effect of healthy synovial fluid. Osteoarthritis Cartilage 17, 1327–1332 (2009).
Lee, S. S. et al. Frictional response of normal and osteoarthritic articular cartilage in human femoral head. Proc. Inst. Mech. Eng. H. 227, 129–137 (2013).
Desrochers, J., Amrein, M. W. & Matyas, J. R. Microscale surface friction of articular cartilage in early osteoarthritis. J. Mech. Behav. Biomed. Mater. 25, 11–22 (2013).
Rah, M. J. A review of hyaluronan and its ophthalmic applications. Optometry 82, 38–43 (2011).
Fonn, D. Targeting contact lens induced dryness and discomfort: What properties will make lenses more comfortable. Optom. Vis. Sci. 84, 279–285 (2007).
Hargittai, I. & Hargittai, M. Molecular structure of hyaluronan: An introduction. Struct. Chem. 19, 697–717 (2008).
Inoue, M. & Katakami, C. The effect of hyaluronic acid on corneal epithelial cell proliferation. Invest. Opthalmol. Vis. Sci. 34, 2313–2315 (1993).
Pauloin, T., Dutot, M., Joly, F., Warnet, J. M. & Rat, P. High molecular weight hyaluronan decreases UVB-induced apoptosis and inflammation in human epithelial corneal cells. Mol. Vis. 15, 577–583 (2009).
Rothenfluh, D. A., Bermudez, H., O’Neil, C. P. & Hubbell, J. A. Biofunctional polymer nanoparticles for intra-articular targeting and retention in cartilage. Nature Mater. 7, 248–254 (2008).
Löster, K., Zeilinger, K., Schuppan, D. & Reutter, W. The cysteine-rich region of dipeptidyl peptidase IV (CD26) is the collagen-binding site. Biochem. Biophys. Res. Commun. 217, 341–348 (1995).
Sistiabudi, R. & Ivanisevic, A. Collagen-binding peptide interaction with retinal tissue surfaces. Langmuir 24, 1591–1594 (2008).
Schmidt, T. A. & Sah, R. L. Effect of synovial fluid on boundary lubrication of articular cartilage. Osteoarthritis Cartilage 15, 35–47 (2007).
Jones, A. R. et al. Binding and localization of recombinant lubricin to articular cartilage surfaces. J. Orthop. Res. 25, 283–292 (2007).
Acknowledgements
We thank F. Guilak (Duke University) for helpful discussions on the friction testing. A.S. was supported by the Arthritis Research Foundation Award 5885 and S.A.U. was supported in part by the National Institutes of Health (NIH) under the Ruth L. Kirschstein National Research Service Award AG328232. Funding sources gratefully acknowledged are NIH R01AR054005, DoD-PRORP grant, the Wallace H. Coulter Foundation, the Ort Philanthropic Fund for supporting the rheometer and the Jules Stein Professorship from the Research to Prevent Blindness Foundation. We gratefully acknowledge the Johns Hopkins A.B. Mass Spectrometry/Proteomic Facility for providing access to the matrix-assisted laser-desorption ionization time-of-flight spectrometer, the Johns Hopkins Department of Chemistry Instrumentation Facility for providing access to the peptide synthesizer, and the Johns Hopkins Department of Materials Science and the Fairbrother research group for use of the surface analysis laboratory.
Author information
Authors and Affiliations
Contributions
The experiments were designed by A.S., M.C., S.A.U., P.M. and J.H.E., and carried out by A.S., M.C., S.A.U. and K.A.W. Data analyses were performed by A.S., M.C. and J.H.E. The manuscript was written by A.S. and J.H.E.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Information
Supplementary Information (PDF 966 kb)
Rights and permissions
About this article
Cite this article
Singh, A., Corvelli, M., Unterman, S. et al. Enhanced lubrication on tissue and biomaterial surfaces through peptide-mediated binding of hyaluronic acid. Nature Mater 13, 988–995 (2014). https://doi.org/10.1038/nmat4048
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nmat4048
This article is cited by
-
Surface hydrophobization of hydrogels via interface dynamics-induced network reconfiguration
Nature Communications (2024)
-
A pre-market interventional, single-arm clinical investigation of a new topical lotion based on hyaluronic acid and peptides, EGYFILTM, for the treatment of pain and stiffness in soft tissues
BMC Musculoskeletal Disorders (2023)
-
Tribology of enzymatically degraded cartilage mimicking early osteoarthritis
Friction (2023)
-
An overview of functional biolubricants
Friction (2023)
-
Study on the Grafting Polyelectrolyte on Cross-Linked Polyethylene Surface and Its Biotribological Behaviors
Journal of Bio- and Tribo-Corrosion (2023)