Abstract
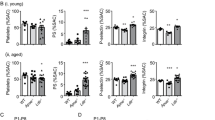
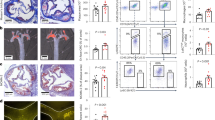
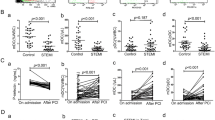
We studied whether circulating activated platelets and platelet–leukocyte aggregates cause the development of atherosclerotic lesions in apolipoprotein-E–deficient (Apoe−/−) mice. Circulating activated platelets bound to leukocytes, preferentially monocytes, to form platelet–monocyte/leukocyte aggregates. Activated platelets and platelet–leukocyte aggregates interacted with atherosclerotic lesions. The interactions of activated platelets with monocytes and atherosclerotic arteries led to delivery of the platelet-derived chemokines CCL5 (regulated on activation, normal T cell expressed and secreted, RANTES) and CXCL4 (platelet factor 4) to the monocyte surface and endothelium of atherosclerotic arteries. The presence of activated platelets promoted leukocyte binding of vascular cell adhesion molecule-1 (VCAM-1) and increased their adhesiveness to inflamed or atherosclerotic endothelium. Injection of activated wild-type, but not P-selectin–deficient, platelets increased monocyte arrest on the surface of atherosclerotic lesions and the size of atherosclerotic lesions in Apoe−/− mice. Our results indicate that circulating activated platelets and platelet–leukocyte/monocyte aggregates promote formation of atherosclerotic lesions. This role of activated platelets in atherosclerosis is attributed to platelet P-selectin–mediated delivery of platelet-derived proinflammatory factors to monocytes/leukocytes and the vessel wall.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Ross, R. Atherosclerosis—an inflammatory disease. N. Engl. J. Med. 340, 115–126 (1999).
Ross, R. & Glomset, J.A. Atherosclerosis and the arterial smooth muscle cell. Proliferation of smooth muscle is a key event in the genesis of the lesions of atherosclerosis. Science 180, 1332–1339 (1973).
Friedman, R.J. et al. The effect of thrombocytopenia on experimental arteriosclerotic lesion formation in rabbits. Smooth muscle cell proliferation and re-endothelialization. J. Clin. Invest. 60, 1191–1201 (1977).
Mendelsohn, M.E.F. & Loscalzo, J. Role of platelets in cholesteryl ester formation by U-937 cells. J. Clin. Invest. 81, 62–68 (1988).
Johnson, R.C. et al. Absence of P-selectin delays fatty streak formation in mice. J. Clin. Invest. 99, 1037–1043 (1997).
Cybulsky, M.I. et al. A major role for VCAM-1, but not ICAM-1, in early atherosclerosis. J. Clin. Invest. 107, 1255–1262 (2001).
Gu, L. et al. Absence of monocyte chemoattractant protein-1 reduces atherosclerosis in low density lipoprotein receptor-deficient mice. Mol. Cell 2, 275–281 (1998).
Boring, L., Gosling, J., Cleary, M. & Charo, I.F. Decreased lesion formation in CCR2(−/−) mice reveals a role for chemokines in the initiation of atherosclerosis. Nature 394, 894–897 (1998).
Fitzgerald, D.J., Roy, L., Catella, F. & Fitzgerald, G.A. Platelet activation in unstable coronary disease. N. Engl. J. Med. 315, 983–989 (1986).
Trip, M.D., Cats, V.M., van Capelle, F.J. & Vreeken, J. Platelet hyperreactivity and prognosis in survivors of myocardial infarction. N. Engl. J. Med. 322, 1549–1554 (1990).
Becker, R.C., Tracy, R.P., Bovill, E.G., Mann, K.G. & Ault, K. The clinical use of flow cytometry for assessing platelet activation in acute coronary syndromes. TIMI-III Thrombosis and Anticoagulation Group. Coron. Artery Dis. 5, 339–345 (1994).
van Zanten, G.H. et al. Increased platelet deposition on atherosclerotic coronary arteries. J. Clin. Invest. 93, 615–632 (1994).
Furman, M.I. et al. Increased platelet reactivity and circulating monocyte-platelet aggregates in patients with stable coronary artery disease. J. Am. College Cardiol. 31, 352–358 (1998).
Broijersen, A., Hamsten, A., Eriksson, M., Angelin, B. & Hjemdahl, P. Platelet activity in vivo in hyperlipoproteinemia—importance of combined hyperlipidemia. Thromb. Haemost. 79, 268–275 (1998).
Broijersen, A., Karpe, F., Hamsten, A., Goodall, A.H. & Hjemdahl, P. Alimentary lipemia enhances the membrane expression of platelet P-selectin without affecting other markers of platelet activation. Atherosclerosis 137, 107–113 (1998).
Hamburger, S.A. & McEver, R.P. GMP-140 mediates adhesion of stimulated platelets to neutrophils. Blood 75, 550–554 (1990).
Rinder, C.S. et al. Cardiopulmonary bypass induces leukocyte-platelet adhesion. Blood 79, 1201–1205 (1992).
Theilmeier, G. et al. Circulating activated platelets assist THP-1 monocytoid/endothelial cell interaction under shear stress. Blood 94, 2725–2734 (1999).
Diacovo, T.G., Puri, K.D., Warnock, R.A., Springer, T.A. & von Andrian, U.H. Platelet-mediated lymphocyte delivery to high endothelial venules. Science 273, 252–255 (1996).
Henn, V. et al. CD40 ligand on activated platelets triggers an inflammatory reaction of endothelial cells. Nature 391, 591–594 (1998).
Hawrylowicz, C.M., Howells, G.L. & Feldmann, M. Platelet-derived interleukin 1 induces human endothelial adhesion molecule expression and cytokine production. J. Exp. Med. 174, 785–790 (1991).
von Hundelshausen, P. et al. RANTES deposition by platelets triggers monocyte arrest on inflamed and atherosclerotic endothelium. Circulation 103, 1772–1777 (2001).
Ramos, C.L. et al. Direct demonstration of P-selectin- and VCAM-1-dependent mononuclear cell rolling in early atherosclerotic lesions of apolipoprotein E-deficient mice. Circ. Res. 84, 1237–1244 (1999).
Ziegler-Heitbrock, H.W.F. et al. Establishment of a human cell line (Mono Mac 6) with characteristics of mature monocytes. Int. J. Cancer 41, 456–461 (1988).
Jung, S. et al. Analysis of fractalkine receptor CX(3)CR1 function by targeted deletion and green fluorescent protein reporter gene insertion. Mol. Cell. Biol. 20, 4106–4114 (2000).
Frenette, P.S. et al. Platelet-endothelial interactions in inflamed mesenteric venules. Blood 91, 1318–1324 (1998).
Andre, P.F. et al. Platelets adhere to and translocate on von Willebrand factor presented by endothelium in stimulated veins. Blood 96, 3322–3328 (2000).
Theilmeier, G.F. et al. Endothelial von Willebrand factor recruits platelets to atherosclerosis-prone sites in response to hypercholesterolemia. Blood 99, 4486–4493 (2002).
Romo, G.M. et al. The glycoprotein Ib-IX-V complex is a platelet counterreceptor for P-selectin. J. Exp. Med. 190, 803–814 (1999).
Massberg, S.F. et al. A critical role of platelet adhesion in the initiation of atherosclerotic lesion formation. J. Exp. Med. 196, 887–896 (2002).
Bombeli, T., Schwartz, B.R. & Harlan, J.M. Adhesion of activated platelets to endothelial cells—evidence for a gpIIb/IIIa-dependent bridging mechanism and novel roles for endothelial intercellular adhesion molecule 1 (ICAM-1), αvβ3 integrin, and gpIb-α. J. Exp. Med. 187, 329–339 (1998).
Rinder, H.M.F., Bonan, J.L.F., Rinder, C.S.F., Ault, K.A.F. & Smith, B.R. Activated and unactivated platelet adhesion to monocytes and neutrophils. Blood 78, 1760–1769 (1991).
Berger, G., Hartwell, D.W. & Wagner, D.D. P-selectin and platelet clearance. Blood 92, 4446–4452 (1998).
Newman, P.M. & Chong, B.H. Heparin-induced thrombocytopenia: new evidence for the dynamic binding of purified anti-PF4-heparin antibodies to platelets and the resultant platelet activation. Blood 96, 182–187 (2000).
Horuk, R. et al. A receptor for the malarial parasite Plasmodium vivax: the erythrocyte chemokine receptor. Science 261, 1182–1184 (1993).
Neote, K., Darbonne, W., Ogez, J., Horuk, R. & Schall, T.J. Identification of a promiscuous inflammatory peptide receptor on the surface of red blood cells. J. Biol. Chem. 268, 12247–12249 (1993).
Mannaioni, P.F., Di Bello, M.G. & Masini, E. Platelets and inflammation: role of platelet-derived growth factor, adhesion molecules and histamine. Inflamm. Res. 46, 4–18 (1997).
Burger, P.C. & Wagner, D.D. Platelet P-selectin facilitates atherosclerotic lesion development. Blood (in press, 2002).
Schober, A.F. et al. Deposition of platelet RANTES triggering monocyte recruitment requires P-selectin and is involved in neointima formation after arterial injury. Circulation 106, 1523–1529 (2002).
Constantin, G.F. et al. Chemokines trigger immediate β2 integrin affinity and mobility changes: differential regulation and roles in lymphocyte arrest under flow. Immunity 13, 759–769 (2000).
Grabovsky, V. et al. Subsecond induction of α4 integrin clustering by immobilized chemokines stimulates leukocyte tethering and rolling on endothelial vascular cell adhesion molecule 1 under flow conditions. J. Exp. Med. 192, 495–506 (2000).
Chan, J.R.F., Hyduk, S.J.F. & Cybulsky, M.I. Chemoattractants induce a rapid and transient upregulation of monocyte α4 integrin affinity for vascular cell adhesion molecule 1 which mediates arrest: an early step in the process of emigration. J. Exp. Med. 193, 1149–1158 (2001).
Piccardoni, P. et al. Platelet/polymorphonuclear leukocyte adhesion: a new role for SRC kinases in Mac-1 adhesive function triggered by P-selectin. Blood 98, 108–116 (2001).
Frenette, P.S., Johnson, R.C., Hynes, M.R. & Wagner, D.D. Platelets roll on stimulated endothelium in vivo: an interaction mediated by endothelial P-selectin. Proc. Natl. Acad. Sci. USA 92, 7450–7454 (1995).
Nunnari, J.J., Zand, T., Joris, I. & Majno, G. Quantitation of oil red O staining of the aorta in hypercholesterolemic rats. Exp. Mol. Pathol. 51, 1–8 (1989).
Acknowledgements
We thank M. Kirkpatrick for mouse husbandry; A.L. Beaudet for Selp−/− mice; R.P. McEver for P-selectin mAbs; D.D. Wagner and H. Ni for advice on murine platelet isolation; and J.A. Redick for technical assistance in scanning electron microscopy and confocal microscopy. This work was supported by NIH grant HL-58108 to K.L., by DFG grant We-1913/2 to C.W. and AHA post-doctoral fellowship award 0120404U to Y.H.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Huo, Y., Schober, A., Forlow, S. et al. Circulating activated platelets exacerbate atherosclerosis in mice deficient in apolipoprotein E. Nat Med 9, 61–67 (2003). https://doi.org/10.1038/nm810
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nm810
This article is cited by
-
Alkaline ceramidase 1–mediated platelet ceramide catabolism mitigates vascular inflammation and abdominal aortic aneurysm formation
Nature Cardiovascular Research (2023)
-
Current concepts and novel targets for antiplatelet therapy
Nature Reviews Cardiology (2023)
-
Residual risks and evolving atherosclerotic plaques
Molecular and Cellular Biochemistry (2023)
-
Immune cell-mediated features of non-alcoholic steatohepatitis
Nature Reviews Immunology (2022)
-
Sex differences at the platelet–vascular interface
Internal and Emergency Medicine (2022)