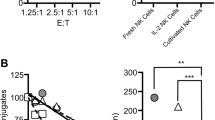
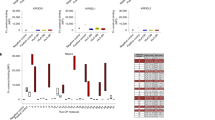
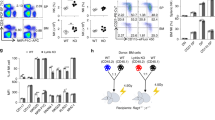
Abstract
Receptors encoded by the natural killer (NK) cell gene complex (such as NKG2D) govern the reactivity of NK cells. However, the function and ligand(s) of the NK cell gene complex–encoded human NK cell receptor NKp80 remain elusive. Here we demonstrate that NKp80 binds to the genetically linked 'orphan' receptor AICL, which, like NKp80, is absent from rodents. We defined AICL as a myeloid-specific activating receptor that is upregulated by Toll-like receptor stimulation. AICL-NKp80 interactions promoted NK cell–mediated cytolysis of malignant myeloid cells. In addition, during crosstalk between NK cells and monocytes, NKp80 stimulated the release of proinflammatory cytokines from both cell types. Thus, by specifically bridging NK cells and myeloid cells, NKp80-AICL interactions may contribute to the initiation and maintenance of immune responses at sites of inflammation.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Trinchieri, G. Biology of natural killer cells. Adv. Immunol. 47, 187–376 (1989).
Karre, K. NK cells, MHC class I molecules and the missing self. Scand. J. Immunol. 55, 221–228 (2002).
Lanier, L.L. NK cell recognition. Annu. Rev. Immunol. 23, 225–274 (2005).
Raulet, D.H. Interplay of natural killer cells and their receptors with the adaptive immune response. Nat. Immunol. 5, 996–1002 (2004).
Degli-Esposti, M.A. & Smyth, M.J. Close encounters of different kinds: dendritic cells and NK cells take centre stage. Nat. Rev. Immunol. 5, 112–124 (2005).
Martin-Fontecha, A. et al. Induced recruitment of NK cells to lymph nodes provides IFN-γ for TH1 priming. Nat. Immunol. 5, 1260–1265 (2004).
Ferlazzo, G. et al. Human dendritic cells activate resting natural killer (NK) cells and are recognized via the NKp30 receptor by activated NK cells. J. Exp. Med. 195, 343–351 (2002).
Fernandez, N.C. et al. Dendritic cells directly trigger NK cell functions: cross-talk relevant in innate anti-tumor immune responses in vivo. Nat. Med. 5, 405–411 (1999).
Gerosa, F. et al. Reciprocal activating interaction between natural killer cells and dendritic cells. J. Exp. Med. 195, 327–333 (2002).
Piccioli, D., Sbrana, S., Melandri, E. & Valiante, N.M. Contact-dependent stimulation and inhibition of dendritic cells by natural killer cells. J. Exp. Med. 195, 335–341 (2002).
Baratin, M. et al. Natural killer cell and macrophage cooperation in MyD88-dependent innate responses to Plasmodium falciparum. Proc. Natl. Acad. Sci. USA 102, 14747–14752 (2005).
Dalbeth, N. et al. CD56bright NK cells are enriched at inflammatory sites and can engage with monocytes in a reciprocal program of activation. J. Immunol. 173, 6418–6426 (2004).
Moretta, A. et al. Activating receptors and coreceptors involved in human natural killer cell-mediated cytolysis. Annu. Rev. Immunol. 19, 197–223 (2001).
Bauer, S. et al. Activation of NK cells and T cells by NKG2D, a receptor for stress-inducible MICA. Science 285, 727–729 (1999).
Roda-Navarro, P. et al. Human KLRF1, a novel member of the killer cell lectin-like receptor gene family: molecular characterization, genomic structure, physical mapping to the NK gene complex and expression analysis. Eur. J. Immunol. 30, 568–576 (2000).
Vitale, M. et al. Identification of NKp80, a novel triggering molecule expressed by human NK cells. Eur. J. Immunol. 31, 233–242 (2001).
Yokoyama, W.M. & Plougastel, B.F. Immune functions encoded by the natural killer gene complex. Nat. Rev. Immunol. 3, 304–316 (2003).
Cosman, D. et al. ULBPs, novel MHC class I-related molecules, bind to CMV glycoprotein UL16 and stimulate NK cytotoxicity through the NKG2D receptor. Immunity 14, 123–133 (2001).
Raulet, D.H. Roles of the NKG2D immunoreceptor and its ligands. Nat. Rev. Immunol. 3, 781–790 (2003).
Vivier, E., Tomasello, E. & Paul, P. Lymphocyte activation via NKG2D: towards a new paradigm in immune recognition? Curr. Opin. Immunol. 14, 306–311 (2002).
Oppenheim, D.E. et al. Sustained localized expression of ligand for the activating NKG2D receptor impairs natural cytotoxicity in vivo and reduces tumor immunosurveillance. Nat. Immunol. 6, 928–937 (2005).
Smyth, M.J. et al. NKG2D function protects the host from tumor initiation. J. Exp. Med. 202, 583–588 (2005).
Biassoni, R. et al. Molecular and functional characterization of NKG2D, NKp80, and NKG2C triggering NK cell receptors in rhesus and cynomolgus macaques: monitoring of NK cell function during simian HIV infection. J. Immunol. 174, 5695–5705 (2005).
Mavilio, D. et al. Identification of NKG2A and NKp80 as specific natural killer cell markers in rhesus and pigtailed monkeys. Blood 106, 1718–1725 (2005).
Cooper, M.A. et al. Human natural killer cells: a unique innate immunoregulatory role for the CD56bright subset. Blood 97, 3146–3151 (2001).
Carlyle, J.R. et al. Missing self-recognition of Ocil/Clr-b by inhibitory NKR-P1 natural killer cell receptors. Proc. Natl. Acad. Sci. USA 101, 3527–3532 (2004).
Iizuka, K., Naidenko, O.V., Plougastel, B.F., Fremont, D.H. & Yokoyama, W.M. Genetically linked C-type lectin-related ligands for the NKRP1 family of natural killer cell receptors. Nat. Immunol. 4, 801–807 (2003).
Aldemir, H. et al. Cutting edge: lectin-like transcript 1 is a ligand for the CD161 receptor. J. Immunol. 175, 7791–7795 (2005).
Rosen, D.B. et al. Cutting edge: lectin-like transcript-1 is a ligand for the inhibitory human NKR-P1A receptor. J. Immunol. 175, 7796–7799 (2005).
Hamann, J., Montgomery, K.T., Lau, S., Kucherlapati, R. & van Lier, R.A. AICL: a new activation-induced antigen encoded by the human NK gene complex. Immunogenetics 45, 295–300 (1997).
Belge, K.U. et al. The proinflammatory CD14+CD16+DR++ monocytes are a major source of TNF. J. Immunol. 168, 3536–3542 (2002).
Bouchon, A., Dietrich, J. & Colonna, M. Cutting edge: inflammatory responses can be triggered by TREM-1, a novel receptor expressed on neutrophils and monocytes. J. Immunol. 164, 4991–4995 (2000).
Pende, D. et al. Expression of the DNAM-1 ligands, Nectin-2 (CD112) and poliovirus receptor (CD155), on dendritic cells: relevance for natural killer-dendritic cell interaction. Blood 107, 2030–2036 (2006).
LeNaour, F. et al. Profiling changes in gene expression during differentiation and maturation of monocyte-derived dendritic cells using both oligonucleotide microarrays and proteomics. J. Biol. Chem. 276, 17920–17931 (2001).
Shiow, L.R. et al. CD69 acts downstream of interferon-alpha/beta to inhibit S1P1 and lymphocyte egress from lymphoid organs. Nature 440, 540–544 (2006).
Vankayalapati, R. et al. Role of NK cell-activating receptors and their ligands in the lysis of mononuclear phagocytes infected with an intracellular bacterium. J. Immunol. 175, 4611–4617 (2005).
Li, P. et al. Complex structure of the activating immunoreceptor NKG2D and its MHC class I–like ligand MICA. Nat. Immunol. 2, 443–451 (2001).
Korbel, D.S., Newman, K.C., Almeida, C.R., Davis, D.M. & Riley, E.M. Heterogeneous human NK cell responses to Plasmodium falciparum-infected erythrocytes. J. Immunol. 175, 7466–7473 (2005).
Zhang, R., Zheng, X., Li, B., Wei, H. & Tian, Z. Human NK cells positively regulate γδ T cells in response to Mycobacterium tuberculosis. J. Immunol. 176, 2610–2616 (2006).
Radsak, M.P. et al. The heat shock protein Gp96 binds to human neutrophils and monocytes and stimulates effector functions. Blood 101, 2810–2815 (2003).
Welte, S.A. et al. Selective intracellular retention of virally induced NKG2D ligands by the human cytomegalovirus UL16 glycoprotein. Eur. J. Immunol. 33, 194–203 (2003).
Aida, Y. & Pabst, M.J. Removal of endotoxin from protein solutions by phase separation using Triton X-114. J. Immunol. Methods 132, 191–195 (1990).
Alter, G., Malenfant, J.M. & Altfeld, M. CD107a as a functional marker for the identification of natural killer cell activity. J. Immunol. Methods 294, 15–22 (2004).
Waldhauer, I. & Steinle, A. Proteolytic release of soluble UL16-binding protein 2 from tumor cells. Cancer Res. 66, 2520–2526 (2006).
Acknowledgements
We thank W. Ruschmeier for technical assistance, J. Bukur for CD161-ED, J. Pfeiffer for real-time PCR, A. Kelp for enhanced green fluorescent protein constructs, S. Stevanovic for mass spectrometry, and H.-G. Rammensee for long-term support and critical remarks; R848 was from S. Bauer (Philipps University Marburg). Supported by Deutsche Forschungsgemeinschaft (STE 828/3-1 and SFB 685 TP A1).
Author information
Authors and Affiliations
Contributions
S.W. generated monoclonal antibodies, produced recombinant NKp80, AICL and LLT1, designed and did most of the experiments and participated in writing the manuscript; S.K. analyzed the degranulation and cytokine secretion of NK cells stimulated by plate-bound antibodies; I.W. did immunoblots; and A.S. did surface plasmon resonance measurements, conceptualized the work and wrote the manuscript.
Corresponding author
Ethics declarations
Competing interests
S.W. and A.S. have applied for a patent (submitted 6 September 2006 by the University of Tübingen) based on the results in this manuscript.
Supplementary information
Supplementary Fig. 1
Recombinant soluble ectodomains (ED) of various C-type lectin-like receptors. (PDF 129 kb)
Supplementary Fig. 2
Specificity of NKp80-specific antibodies 5D12, 10E4, and 12D11. (PDF 71 kb)
Supplementary Fig. 3
Abundance of AICL and NKp80 transcripts in leukocyte subpopulations. (PDF 30 kb)
Supplementary Fig. 4
AICL-specific antibodies. (PDF 96 kb)
Supplementary Fig. 5
Frequencies of IFN-γ-producing NK cells after 12 h co-culture with autologous CD14dimCD16+ monocytes in the presence of LPS. (PDF 84 kb)
Supplementary Table 1
AICL surface expression on human primary cells and tumor cell lines. (PDF 31 kb)
Rights and permissions
About this article
Cite this article
Welte, S., Kuttruff, S., Waldhauer, I. et al. Mutual activation of natural killer cells and monocytes mediated by NKp80-AICL interaction. Nat Immunol 7, 1334–1342 (2006). https://doi.org/10.1038/ni1402
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ni1402
This article is cited by
-
Natural killer cells: a promising immunotherapy for cancer
Journal of Translational Medicine (2022)
-
Structure of the human NK cell NKR-P1:LLT1 receptor:ligand complex reveals clustering in the immune synapse
Nature Communications (2022)
-
The NK cell–cancer cycle: advances and new challenges in NK cell–based immunotherapies
Nature Immunology (2020)
-
Transcriptional shifts account for divergent resource allocation in feed efficient broiler chickens
Scientific Reports (2018)
-
Recognition of host Clr-b by the inhibitory NKR-P1B receptor provides a basis for missing-self recognition
Nature Communications (2018)