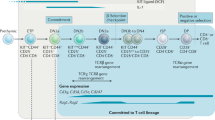
Abstract
To become accessible for rearrangement, the immunoglobulin κ locus must undergo a series of epigenetic changes. This begins in pro–B cells with the relocation of both immunoglobulin κ alleles from the periphery to the center of the nucleus. In pre–B cells, one allele became preferentially packaged into an active chromatin structure characterized by histone acetylation and methylation of histone H3 lysine 4, while the other allele was recruited to heterochromatin, where it was associated with heterochromatin protein-γ and Ikaros. These events in cis made only one allele accessible to trans-acting factors, such as RelB, which mediated DNA demethylation, to facilitate rearrangement. These results suggest that early B lymphoid epigenetic changes generate differential structures that serve as the basis for allelic exclusion.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Krangel, M.S. Gene segment selection in V(D)J recombination: accessibility and beyond. Nat. Immunol. 4, 624–630 (2003).
Bergman, Y., Fisher, A.G. & Cedar, H. Epigenetic mechanisms that regulate antigen receptor gene expression. Curr. Opin. Immunol. 15, 176–181 (2003).
Schatz, D.G., Oettinger, M.A. & Baltimore, D. The V(D)J recombination activating gene, RAG-1. Cell 59, 1035–1048 (1989).
Oettinger, M.A., Schatz, D.G., Gorka, C. & Baltimore, D. RAG-1 and RAG-2, adjacent genes that synergistically activate V(D)J recombination. Science 248, 1517–1523 (1990).
Gellert, M. A new view of V(D)J recombination. Genes Cells 1, 269–275 (1996).
Corcoran, A.E., Riddell, A., Krooshoop, D. & Venkitaraman, A.R. Impaired immunoglobulin gene rearrangement in mice lacking the IL-7 receptor. Nature 391, 904–907 (1998).
Lewis, S.A. The mechanism of V(D)J joining: lessons from molecular, immunological and comparative analyses. Adv. Immunol. 56, 27–150 (1994).
Sleckman, B.P., Gorman, J.R. & Alt, F.W. Accessibility control of antigen-receptor variable-region gene assembly: role of cis-acting elements. Annu. Rev. Immunol. 14, 459–481 (1996).
Brown, K.E. et al. Association of transcriptionally silent genes with Ikaros complexes at centromeric heterochromatin. Cell 91, 845–854 (1997).
Brown, K.E., Baxter, J., Graf, D., Merkenschlager, M. & Fisher, A.G. Dynamic repositioning of genes in the nucleus of lymphocytes preparing for cell division. Mol. Cell 3, 207–217 (1999).
Skok, J.A. et al. Nonequivalent nuclear location of immunoglobulin alleles in B lymphocytes. Nat. Immunol. 2, 848–854 (2001).
Mather, E.L. & Perry, R.P. Methylation status and DNaseI sensitivity of immunoglobulin genes: changes associated with rearrangement. Proc. Natl. Acad. Sci. USA 80, 4689–4693 (1983).
Goldmit, M., Schlissel, M., Cedar, H. & Bergman, Y. Differential accessibility at the kappa chain locus plays a role in allelic exclusion. EMBO J. 21, 5255–5261 (2002).
Mostoslavsky, R. et al. κ chain monoallelic demethylation and the establishment of allelic exclusion. Genes Dev. 12, 1801–1811 (1998).
Chowdhury, D. & Sen, R. Stepwise activation of the immunoglobulin μ heavy chain gene locus. EMBO J. 20, 6394–6403 (2001).
Maës, J. et al. Chromatin remodeling at the Ig loci prior to V(D)J recombination. J. Immunol. 167, 866–874 (2001).
Morshead, K.B., Ciccone, D.N., Taverna, S.D., Allis, C.D. & Oettinger, M.A. Antigen receptor loci poised for V(D)J rearrangement are broadly associated with BRG1 and flanked by peaks of histone H3 dimethylated at lysine 4. Proc. Natl. Acad. Sci. USA 100, 11577–11582 (2003).
Kosak, S.T. et al. Subnuclear compartmentalization of immunoglobulin loci during lymphocyte development. Science 296, 158–162 (2002).
Kirillov, A. et al. A role for nuclear NF-κB in B-cell-specific demethylation of the Igκ locus. Nat. Genet. 13, 435–441 (1996).
Rolink, A., Kudo, A., Karasuyama, H., Kikuchi, Y. & Melchers, F. Long-term proliferating early pre-B cell lines and clones with the potential to develop to surface Ig-positive, mitogen reactive B cells in vitro and in vivo. EMBO J. 10, 327–336 (1991).
Mostoslavsky, R. et al. Asynchronous replication and allelic exclusion in the immune system. Nature 414, 221–225 (2001).
Azuara, V. et al. Heritable gene silencing in lymphocytes delays chromatid resolution without affecting the timing of DNA replication. Nat. Cell Biol. 5, 668–674 (2003).
Maison, C. & Almouzni, G. HP1 and the dynamics of heterochromatin maintenance. Nat. Rev. Mol. Cell Biol. 5, 296–304 (2004).
Dillon, N. & Festenstein, R. Unravelling heterochromatin: competition between positive and negative factors regulates accessibility. Trends Genet. 18, 252–258 (2002).
Sabbattini, P. et al. Binding of Ikaros to the λ5 promoter silences transcription through a mechanism that does not require heterochromatin formation. EMBO J. 20, 2812–2822 (2001).
Mostoslavsky, R. et al. Demethylation and the establishment of κ allelic exclusion. Cold Spring Harb. Symp. Quant. Biol. 64, 197–206 (1999).
Hashimshony, T., Zhang, J., Keshet, I., Bustin, M. & Cedar, H. The role of DNA methylation in setting up chromatin structure during development. Nat. Genet. 34, 187–192 (2003).
Smale, S.T. The establishment and maintenance of lymphocyte identity through gene silencing. Nat. Immunol. 4, 607–615 (2003).
Fisher, A.G. & Merkenschlager, M. Gene silencing, cell fate and nuclear organisation. Curr. Opin. Genet. Dev. 12, 193–197 (2002).
Martin, D.J. van Ness, B.G. Initiation and processing of two kappa immunoglobulin germ line transcripts in mouse B cells. Mol. Cell. Biol. 10, 1950–1958 (1990).
Singh, N., Bergman, Y., Cedar, H. & Chess, A. Biallelic germline transcription at the κ immunoglobulin locus. J. Exp. Med. 197, 743–750 (2003).
Georgopoulos, K. Haematopoietic cell-fate decisions, chromatin regulation and ikaros. Nat. Rev. Immunol. 2, 162–174 (2002).
Smale, S.T. & Fisher, A.G. Chromatin structure and gene regulation in the immune system. Annu. Rev. Immunol. 20, 427–462 (2002).
Sun, L., Liu, A. & Georgopoulos, K. Zinc finger-mediated protein interactions modulate Ikaros activity, a molecular control of lymphocyte development. EMBO J. 15, 5358–5369 (1996).
Cobb, B.S. et al. Targeting of Ikaros to pericentromeric heterochromatin by direct DNA binding. Genes Dev. 14, 2146–2160 (2000).
Strom, L., Lundgren, M. & Severinson, E. Binding of Ikaros to germline Ig heavy chain γ1 and ε promoters. Mol. Immunol. 39, 771–782 (2003).
Harker, N. et al. The CD8α gene locus is regulated by the Ikaros family of proteins. Mol. Cell 10, 1403–1415 (2002).
Trinh, L.A. et al. Down-regulation of TDT transcription in CD4+CD8+ thymocytes by Ikaros proteins in direct competition with an Ets activator. Genes Dev. 15, 1817–1832 (2001).
McBlane, F. & Boyes, J. Stimulation of V(D)J recombination by histone acetylation. Curr. Biol. 10, 483–486 (2000).
Su, I.H. et al. Ezh2 controls B cell development through histone H3 methylation and Igh rearrangement. Nat. Immunol. 4, 124–131 (2003).
Johnson, K., Angelin-Duclos, C., Park, S. & Calame, K.L. Changes in histone acetylation are associated with differences in accessibility of V(H) gene segments to V-DJ recombination during B-cell ontogeny and development. Mol. Cell. Biol. 23, 2438–2450 (2003).
Su, R.C. et al. Dynamic assembly of silent chromatin during thymocyte maturation. Nat. Genet. 36, 502–506 (2004).
Liang, H-E., Hsu, L-Y., Cado, D. & Schlissel, M.S. Variegated transcriptional activation of the immunoglobulin κ locus in pre-B cells contributes to the allelic exclusion of light-chain expression. Cell 118, 19–29 (2004).
Southern, E.M. Detection of specific sequences among DNA fragments separated by gel electrophoresis. J. Mol. Biol. 98, 503–517 (1975).
Hebbes, T.R., Clayton, A.L., Thorne, A.W. & Crane-Robinson, C. Core histone hyperacetylation co-maps with generalized DNase I sensitivity in the chicken β-globin chromosomal domain. EMBO J. 13, 1823–1830 (1994).
Ji, Y., Zhang, J., Lee, A.I., Cedar, H. & Bergman, Y. A multistep mechanism for the activation of rearrangement in the immune system. Proc. Natl. Acad. Sci. USA 100, 7557–7562 (2003).
Lichtenstein, M., Keini, G., Cedar, H. & Bergman, Y. B-cell specific demethylation: a novel role for the intronic κ-chain enhancer sequence. Cell 76, 913–923 (1994).
Acknowledgements
We thank T. Wirth (Ulm University, Ulm, Germany) for the RelB expression plasmid, and L. Lande for help in completing this project. Supported by the Israel Academy of Sciences (H.C. and Y.B.), NIH (H.C. and Y.B.), Israel Cancer Research Foundation (H.C.), European Community 5th Framework Quality of Life Program (Y.B.), Wellcome Trust University Award (J.S.) and a Horvitz Fellowship (M.G.).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Fig. 1
Flow cytometric analysis of wild-type pro-B, IL-7–dependent pre-B and ex vivo small pre-B cells. (PDF 119 kb)
Supplementary Fig. 2
Igk rearrangement in pre-B cells. (PDF 77 kb)
Rights and permissions
About this article
Cite this article
Goldmit, M., Ji, Y., Skok, J. et al. Epigenetic ontogeny of the Igk locus during B cell development. Nat Immunol 6, 198–203 (2005). https://doi.org/10.1038/ni1154
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ni1154
This article is cited by
-
Clonally stable Vκ allelic choice instructs Igκ repertoire
Nature Communications (2017)
-
Dissecting Epigenetic Dysregulation of Primary Antibody Deficiencies
Journal of Clinical Immunology (2016)
-
Multiple factors influence the contribution of individual immunoglobulin light chain genes to the naïve antibody repertoire
BMC Immunology (2014)
-
Histone methylation and V(D)J recombination
International Journal of Hematology (2014)
-
Transcription-coupled eviction of histones H2A/H2B governs V(D)J recombination
The EMBO Journal (2013)