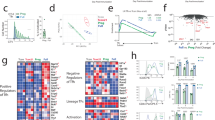
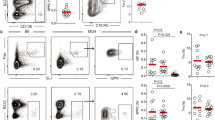
Abstract
High-affinity antibodies are critical for host protection and underlie successful vaccines. The generation of such antibodies requires T cell–dependent help, which mediates germinal center reactions in which mutation and selection of B cells occurs. Using an interleukin 4–reporter system, we show here that CD4+ follicular helper T cells constituted essentially all of the cytokine-secreting T cells in lymph nodes and were functionally distinct from T cells secreting the same cytokine in peripheral tissues. Follicular helper T cells with different cytokine profiles could be isolated as conjugates with B cells undergoing cytokine-specific immunoglobulin class switching with evidence of somatic hypermutation. Our findings support a model in which B cells compete for cytokines produced by follicular helper T cells that shape the affinity and isotype of the antibody response.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
MacLennan, I.C. Germinal centers. Annu. Rev. Immunol. 12, 117–139 (1994).
Kelsoe, G. Life and death in germinal centers (redux). Immunity 4, 107–111 (1996).
Allen, C.D., Okada, T. & Cyster, J.G. Germinal-center organization and cellular dynamics. Immunity 27, 190–202 (2007).
Fang, Y., Xu, C., Fu, Y.X., Holers, V.M. & Molina, H. Expression of complement receptors 1 and 2 on follicular dendritic cells is necessary for the generation of a strong antigen-specific IgG response. J. Immunol. 160, 5273–5279 (1998).
Hannum, L.G., Haberman, A.M., Anderson, S.M. & Shlomchik, M.J. Germinal center initiation, variable gene region hypermutation, and mutant B cell selection without detectable immune complexes on follicular dendritic cells. J. Exp. Med. 192, 931–942 (2000).
Huntington, N.D. et al. CD45 links the B cell receptor with cell survival and is required for the persistence of germinal centers. Nat. Immunol. 7, 190–198 (2006).
Allen, D., Simon, T., Sablitzky, F., Rajewsky, K. & Cumano, A. Antibody engineering for the analysis of affinity maturation of an anti-hapten response. EMBO J. 7, 1995–2001 (1988).
Meyer-Hermann, M.E., Maini, P.K. & Iber, D. An analysis of B cell selection mechanisms in germinal centers. Math. Med. Biol. 23, 255–277 (2006).
Allen, C.D., Okada, T., Tang, H.L. & Cyster, J.G. Imaging of germinal center selection events during affinity maturation. Science 315, 528–531 (2007).
Schwickert, T.A. et al. In vivo imaging of germinal centres reveals a dynamic open structure. Nature 446, 83–87 (2007).
King, C., Tangye, S.G. & Mackay, C.R. T follicular helper (TFH) cells in normal and dysregulated immune responses. Annu. Rev. Immunol. 26, 741–766 (2008).
Fazilleau, N., McHeyzer-Williams, L.J. & McHeyzer-Williams,, M.G. Local development of effector and memory T helper cells. Curr. Opin. Immunol. 19, 259–267 (2007).
Garside, P. et al. Visualization of specific B and T lymphocyte interactions in the lymph node. Science 281, 96–99 (1998).
Takahashi, Y., Dutta, P.R., Cerasoli, D.M. & Kelsoe, G. In situ studies of the primary immune response to (4-hydroxy-3-nitrophenyl)acetyl. V. Affinity maturation develops in two stages of clonal selection. J. Exp. Med. 187, 885–895 (1998).
Chtanova, T. et al. T follicular helper cells express a distinctive transcriptional profile, reflecting their role as non-Th1/Th2 effector cells that provide help for B cells. J. Immunol. 173, 68–78 (2004).
Vogelzang, A. et al. A fundamental role for interleukin-21 in the generation of T follicular helper cells. Immunity 29, 127–137 (2008).
Nurieva, R.I. et al. Generation of T follicular helper cells is mediated by interleukin-21 but independent of T helper 1, 2, or 17 cell lineages. Immunity 29, 138–149 (2008).
Mohrs, K., Wakil, A.E., Killeen, N., Locksley, R.M. & Mohrs, M. A two-step process for cytokine production revealed by IL-4 dual-reporter mice. Immunity 23, 419–429 (2005).
Vinuesa, C.G., Tangye, S.G., Moser, B. & Mackay, C.R. Follicular B helper T cells in antibody responses and autoimmunity. Nat. Rev. Immunol. 5, 853–865 (2005).
Loke, P. et al. Inducible costimulator is required for type 2 antibody isotype switching but not T helper cell type 2 responses in chronic nematode infection. Proc. Natl. Acad. Sci. USA. 102, 9872–9877 (2005).
Tafuri, A. et al. ICOS is essential for effective T-helper-cell responses. Nature 409, 105–109 (2001).
Dong, C., Temann, U.A. & Flavell, R.A. Cutting edge: critical role of inducible costimulator in germinal center reactions. J. Immunol. 166, 3659–3662 (2001).
Finkelman, F.D. et al. Interleukin-4- and interleukin-13-mediated host protection against intestinal nematode parasites. Immunol. Rev. 201, 139–155 (2004).
Voehringer, D., Shinkai, K. & Locksley, R.M. Type 2 immunity reflects orchestrated recruitment of cells committed to IL-4 production. Immunity 20, 267–277 (2004).
Okada, T. et al. Antigen-engaged B cells undergo chemotaxis toward the T zone and form motile conjugates with helper T cells. PLoS Biol. 3, e150 (2005).
Casamayor-Palleja, M., Feuillard, J., Ball, J., Drew, M. & MacLennan, I.C. Centrocytes rapidly adopt a memory B cell phenotype on co-culture with autologous germinal centre T cell-enriched preparations. Int. Immunol. 8, 737–744 (1996).
Muramatsu, M. et al. Class switch recombination and hypermutation require activation-induced cytidine deaminase (AID), a potential RNA editing enzyme. Cell 102, 553–563 (2000).
Muramatsu, M. et al. Specific expression of activation-induced cytidine deaminase (AID), a novel member of the RNA-editing deaminase family in germinal center B cells. J. Biol. Chem. 274, 18470–18476 (1999).
Reiner, S.L., Zheng, S., Wang, Z.E., Stowring, L. & Locksley, R.M. Leishmania promastigotes evade interleukin 12 (IL-12) induction by macrophages and stimulate a broad range of cytokines from CD4+ T cells during initiation of infection. J. Exp. Med. 179, 447–456 (1994).
Bothwell, A.L. et al. Heavy chain variable region contribution to the NPb family of antibodies: somatic mutation evident in a γ2a variable region. Cell 24, 625–637 (1981).
Rajewsky, K., Forster, I. & Cumano, A. Evolutionary and somatic selection of the antibody repertoire in the mouse. Science 238, 1088–1094 (1987).
Jolly, C.J., Klix, N. & Neuberger, M.S. Rapid methods for the analysis of immunoglobulin gene hypermutation: application to transgenic and gene targeted mice. Nucleic Acids Res. 25, 1913–1919 (1997).
Dorsett, Y. et al. MicroRNA-155 suppresses activation-induced cytidine deaminase-mediated Myc-Igh translocation. Immunity 28, 630–638 (2008).
Reiter, R. & Pfeffer, K. Impaired germinal centre formation and humoral immune response in the absence of CD28 and interleukin-4. Immunology 106, 222–228 (2002).
Vajdy, M., Kosco-Vilbois, M.H., Kopf, M., Kohler, G. & Lycke, N. Impaired mucosal immune responses in interleukin 4-targeted mice. J. Exp. Med. 181, 41–53 (1995).
Andoh, A., Masuda, A., Yamakawa, M., Kumazawa, Y. & Kasajima, T. Absence of interleukin-4 enhances germinal center reaction in secondary immune response. Immunol. Lett. 73, 35–41 (2000).
Itano, A.A. & Jenkins, M.K. Antigen presentation to naive CD4 T cells in the lymph node. Nat. Immunol. 4, 733–739 (2003).
Nimmerjahn, F. & Ravetch, J.V. Fcγ receptors: old friends and new family members. Immunity 24, 19–28 (2006).
Reichert, P., Reinhardt, R.L., Ingulli, E. & Jenkins, M.K. Cutting edge: in vivo identification of TCR redistribution and polarized IL-2 production by naive CD4 T cells. J. Immunol. 166, 4278–4281 (2001).
Maldonado, R.A., Irvine, D.J., Schreiber, R. & Glimcher, L.H. A role for the immunological synapse in lineage commitment of CD4 lymphocytes. Nature 431, 527–532 (2004).
Phan, T.G. et al. High affinity germinal center B cells are actively selected into the plasma cell compartment. J. Exp. Med. 203, 2419–2424 (2006).
Tarlinton, D.M. Evolution in miniature: selection, survival and distribution of antigen reactive cells in the germinal centre. Immunol. Cell Biol. 86, 133–138 (2008).
Jacob, J., Przylepa, J., Miller, C. & Kelsoe, G. In situ studies of the primary immune response to (4-hydroxy-3-nitrophenyl)acetyl. III. The kinetics of V region mutation and selection in germinal center B cells. J. Exp. Med. 178, 1293–1307 (1993).
Dal Porto, J.M., Haberman, A.M., Shlomchik, M.J. & Kelsoe, G. Antigen drives very low affinity B cells to become plasmacytes and enter germinal centers. J. Immunol. 161, 5373–5381 (1998).
Zhou, C., Saxon, A. & Zhang, K. Human activation-induced cytidine deaminase is induced by IL-4 and negatively regulated by CD45: implication of CD45 as a Janus kinase phosphatase in antibody diversification. J. Immunol. 170, 1887–1893 (2003).
Dedeoglu, F., Horwitz, B., Chaudhuri, J., Alt, F.W. & Geha, R.S. Induction of activation-induced cytidine deaminase gene expression by IL-4 and CD40 ligation is dependent on STAT6 and NFκB. Int. Immunol. 16, 395–404 (2004).
de Leval, L. et al. The gene expression profile of nodal peripheral T-cell lymphoma demonstrates a molecular link between angioimmunoblastic T-cell lymphoma (AITL) and follicular helper T (TFH) cells. Blood 109, 4952–4963 (2007).
Zangani, M.M. et al. Lymphomas can develop from B cells chronically helped by idiotype-specific T cells. J. Exp. Med. 204, 1181–1191 (2007).
Shinkai, K., Mohrs, M. & Locksley, R.M. Helper T cells regulate type-2 innate immunity in vivo. Nature 420, 825–829 (2002).
Lalor, P.A., Nossal, G.J., Sanderson, R.D. & McHeyzer-Williams, M.G. Functional and molecular characterization of single, (4-hydroxy-3-nitrophenyl)acetyl (NP)-specific, IgG1+ B cells from antibody-secreting and memory B cell pathways in the C57BL/6 immune response to NP. Eur. J. Immunol. 22, 3001–3011 (1992).
Acknowledgements
We thank G. Kelsoe (Duke University), M. Nussenzweig (Rockefeller University), K. McBride (Rockefeller University) and A. N. McKenzie (Trinity College, Dublin) for reagents and suggestions; J. Cyster, A. DeFranco and C. Allen for review and comments; and N. Flores, L. Stowring and C. McArthur for technical expertise. Supported by the National Institutes of Allergy and Infectious Diseases (AI026918 and AI077439), the Howard Hughes Medical Institute, the Sandler Asthma Basic Research Center of the University of California San Francisco and the Juvenile Diabetes Research Foundation–Irvington Institute (R.L.R.).
Author information
Authors and Affiliations
Contributions
R.L.R. and R.M.L. conceived the work; R.L.R. designed, did and analyzed experiments for Figures 1, 2, 3, 4, 5 and 7; R.L.R. helped do and analyze Figure 6; H.-E.L. designed, did and analyzed experiments for Figures 5 and 6; R.M.L. designed experiments; R.L.R and R.M.L. wrote the manuscript; and all authors read and provided comments on the manuscript.
Corresponding author
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–11 and Supplementary Tables 1–2 (PDF 11972 kb)
Rights and permissions
About this article
Cite this article
Reinhardt, R., Liang, HE. & Locksley, R. Cytokine-secreting follicular T cells shape the antibody repertoire. Nat Immunol 10, 385–393 (2009). https://doi.org/10.1038/ni.1715
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ni.1715
This article is cited by
-
Mechanismus der allergischen Sensibilisierung und wozu hat ihn die Evolution eigentlich geschaffen?
Deutsche Zeitschrift für Akupunktur (2024)
-
Engaging an HIV vaccine target through the acquisition of low B cell affinity
Nature Communications (2023)
-
Interferon-γ couples CD8+ T cell avidity and differentiation during infection
Nature Communications (2023)
-
Continually recruited naïve T cells contribute to the follicular helper and regulatory T cell pools in germinal centers
Nature Communications (2023)
-
Pathogen-associated T follicular helper cell plasticity is critical in anti-viral immunity
Science China Life Sciences (2022)