Abstract
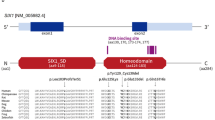
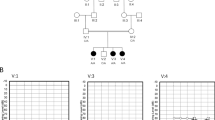
Antenatal Bartter syndrome (aBS) comprises a heterogeneous group of autosomal recessive salt-losing nephropathies1. Identification of three genes that code for renal transporters and channels as responsible for aBS2,3,4,5,6,7 has resulted in new insights into renal salt handling, diuretic action and blood-pressure regulation8. A gene locus of a fourth variant of aBS called BSND, which in contrast to the other forms is associated with sensorineural deafness (SND) and renal failure, has been mapped to chromosome 1p9. We report here the identification by positional cloning, in a region not covered by the human genome sequencing projects, of a new gene, BSND, as the cause of BSND. We examined ten families with BSND and detected seven different mutations in BSND that probably result in loss of function. In accordance with the phenotype, BSND is expressed in the thin limb and the thick ascending limb of the loop of Henle in the kidney and in the dark cells of the inner ear. The gene encodes a hitherto unknown protein with two putative transmembrane α-helices and thus might function as a regulator for ion-transport proteins involved in aBS, or else as a new transporter or channel itself.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Rodriguez-Soriano, J. Bartter and related syndromes: the puzzle is almost solved. Pediatr. Nephrol. 12, 315–327 (1998).
Simon, D.B. et al. Bartter's syndrome, hypokalaemic alkalosis with hypercalciuria, is caused by mutations in the Na-K-2Cl cotransporter NKCC2. Nature Genet. 13, 183–188 (1996).
Simon, D.B. et al. Genetic heterogeneity of Bartter's syndrome revealed by mutations in the K+ channel, ROMK. Nature Genet. 14, 152–156 (1996).
International Collaborative Study Group for Bartter-Like Syndromes. Mutations in the gene encoding the inwardly-rectifying renal potassium channel, ROMK, cause the antenatal variant of Bartter syndrome: evidence for genetic heterogeneity. Hum. Mol. Genet. 6, 17–26 (1997).
Simon, D.B. et al. Mutations in the chloride channel gene, CLCNKB, cause Bartter's syndrome type III. Nature Genet. 17, 171–178 (1997).
Vargas-Poussou, R. et al. Novel molecular variants of the Na-K-2Cl cotransporter gene are responsible for antenatal Bartter syndrome. Am. J. Hum. Genet. 62, 1332–1340 (1998).
Konrad, M. et al. Novel molecular variants in the gene for the basolateral chloride channel CLCNKB as a cause of classic Bartter syndrome. J. Am. Soc. Nephrol. 11, 1449–1549 (2000).
Lifton, R.P. Molecular genetics of human blood pressure variation. Science 272, 676–680 (1996).
Brennan, T.M. et al. Linkage of infantile Bartter syndrome with sensorineural deafness to chromosome 1p. Am. J. Hum. Genet. 62, 355–361 (1998).
Vollmer, M. et al. Antenatal Bartter syndrome with sensorineural deafness: refinement of the locus on chromosome 1p31. Nephrol. Dial. Transplant. 15, 970–974 (2000).
Lander, E.S. & Botstein, D. Homozygosity mapping: a way to map human recessive traits with the DNA of inbred children. Science 236, 1567–1570 (1987).
Ioannou, P.A. et al. A new bacteriophage P1–derived vector for the propagation of large human DNA fragments. Nature Genet. 6, 84–89 (1994).
The International Human Genome Mapping Consortium. A physical map of the human genome. Nature 409, 934–941 (2001).
Venter, J.C. et al. The sequence of the human genome. Science 291, 1304–1351 (2001).
Altschul, S.F. et al. Gapped BLAST and PSI-BLAST: a new generation of protein database search programs. Nucleic Acids Res. 25, 3389–3402 (1997).
Igarashi, P., Vanden Heuvel, G.B., Payne, J.A. & Forbush, B. III. Cloning, embryonic expression, and alternative splicing of a murine kidney-specific Na-K-Cl cotransporter. Am. J. Physiol. 269, F405–F418 (1995).
Kaplan, M.R. et al. Apical localization of the Na-K-Cl cotransporter, rBSC1, on rat thick ascending limbs. Kidney Int. 49, 40–47 (1996).
Yoshikawa, M. et al. Localization of rat CLC-K2 chloride channel mRNA in the kidney. Am. J. Physiol. 276, F552–F558 (1999).
Kobayashi, K., Uchida, S., Mizutani, S., Sasaki, S. & Marumo, F. Intrarenal and cellular localization of clc-k2 protein in the mouse kidney. J. Am. Soc. Nephrol. 12, 1327–1334 (2001).
Steel, K.P. & Kros, C.J. A genetic approach to understanding auditory function. Nature Genet. 27, 143–149 (2001).
Kubisch, C. et al. KCNQ4, a novel potassium channel expressed in sensory outer hair cells, is mutated in dominant deafness. Cell 96, 437–446 (1999).
Tyson, J. et al. IsK and KvLQT1: mutation in either of the two subunits of the slow component of the delayed rectifier potassium channel can cause Jervell and Lange–Nielsen syndrome. Hum. Mol. Genet. 6, 2179–2185 (1997).
Schulze-Bahr, E. et al. KCNE1 mutations cause Jervell and Lange–Nielsen syndrome. Nature Genet. 17, 267–268 (1997).
Petit, C. Genes responsible for human hereditary deafness: symphony of a thousand. Nature Genet. 14, 385–391 (1996).
Kockerling, A., Reinalter, S.C. & Seyberth, H.J. Impaired response to furosemide in hyperprostaglandin E syndrome: evidence for a tubular defect in the loop of Henle. J. Pediatr. 129, 519–528 (1996).
Delpire, E., Lu, J., England, R., Dull, C. & Thorne, T. Deafness and imbalance associated with inactivation of the secretory Na-K-2Cl co-transporter. Nature Genet. 22, 192–195 (1999).
Nicolas, M., Dememes, D., Martin, A., Kupershmidt, S. & Barhanin, J. KCNQ1/KCNE1 potassium channels in mammalian vestibular dark cells. Hear. Res. 153, 132–145 (2001).
Jeck, N. et al. Hypokalemic salt-losing tubulopathy with chronic renal failure and sensorineural deafness. Pediatrics 108, E5 (2001).
Nothwang, H.G. et al. Construction of a gene map of the nephronophthisis type 1 (NPHP1) region on human chromosome 2q12-q13. Genomics 47, 276–285 (1998).
Lescher, B., Haenig, B. & Kispert, A. sFRP-2 is a target of the Wnt-4 signaling pathway in the developing metanephric kidney. Dev. Dyn. 213, 440–451 (1998).
Acknowledgements
We thank the affected patients and their families for participating in this study, Th. Henne, R. Mallmann, W. Marg, L. Monnens, P. Niaudet, H.-J. Seyberth, J. Strehlau and U. Vossmerbäumer for contributing material from patients, A. Imm and M. Petry for excellent technical assistance, T.J. Jentsch and H.J. Groene for helpful discussion, S. Schnittger for fluorescence in situ hybridization (FISH) analysis and S. Gregory at the Sanger Centre for sequencing BAC GS1-118i5. F.H. is a Heisenberg Scholar of the DFG (Hi 381/7-2) and was supported by a grant from the Forschungsschwerpunkt-Programm 'Ionenkanal' (state of Baden-Württemberg).
Author information
Authors and Affiliations
Corresponding author
Supplementary information
Rights and permissions
About this article
Cite this article
Birkenhäger, R., Otto, E., Schürmann, M. et al. Mutation of BSND causes Bartter syndrome with sensorineural deafness and kidney failure. Nat Genet 29, 310–314 (2001). https://doi.org/10.1038/ng752
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ng752
This article is cited by
-
A novel mutation of KCNJ1 identified in an affected child with nephrolithiasis
BMC Nephrology (2022)
-
Development of the stria vascularis in the common marmoset, a primate model
Scientific Reports (2022)
-
Genetic diagnosis and treatment of hereditary renal tubular disease with hypokalemia and alkalosis
Journal of Nephrology (2022)
-
Pathophysiological aspects of the thick ascending limb and novel genetic defects: HELIX syndrome and transient antenatal Bartter syndrome
Pediatric Nephrology (2022)
-
Molecular mechanisms altering tubular calcium reabsorption
Pediatric Nephrology (2022)