Abstract
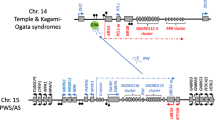
Prader-Willi syndrome (PWS) is a neurogenetic disorder that results from the absence of a normal paternal contribution to the 15q11–13 region1–3. The clinical manifestations of PWS are a transient severe hypotonia in the newborn period, with mental retardation, hypogonadism and obesity observed later in development4. Five transcripts with exclusive expression from the paternal allele have been isolated, but none of these has been shown to be involved in PWS5,6. In this study, we report the isolation and characterization of NDN, a new human imprinted gene. NDN is exclusively expressed from the paternal allele in the tissues analysed and is located in the PWS region. It encodes a putative protein homologous to the mouse brain-specific NECDIN protein7, NDN; as in mouse, expression in brain is restricted to post-mitotic neurons. NDN displays several characteristics of an imprinted locus, including allelic DNA methylation and asynchronous DNA replication. A complete lack of NDN expression in PWS brain and f ibroblasts indicates that the gene is expressed exclusively from the paternal allele in these tissues and suggests a possible role of this new gene in PWS.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Nicholls, R.D., Knoll, J.H.M., Butler, M.G., Karam, S. & Lalande, M. Genetic imprinting suggested by maternal heterodisomy in non-deletion Prader-Willi syndrome. Nature. 342, 281–285 (1989).
Robinson, W.P. et al. Molecular, cytogenetic, and clinical investigations of Prader-Willi syndrome patients. Am. J. Hum. Genet. 49, 1219–1234 (1991).
Mascari, M.J. et al. Molecular diagnosis of Prader-Willi syndrome. N. Engl. J. Med. 326, 1599–1607 (1992).
Holm, V., Cassidy, S., Butler, M., Hanchett, J. & Greenberg, F. Prader-Willi syndrome: consensus diagnostic criteria. Pediatrics. 91, 398–402 (1993).
Lalande, M. Parental imprinting and human disease. Annu. Rev. Genet. 30, 173–195 (1997).
Ning, Y. et al. Identification of a novel paternally expressed transcript adjacent to snRPN in the Prader-Willi region. Genome Res. 6, 742–746 (1996).
Maruyama, K., Usami, M., Aizawa, T. & Yoshikawa, K. A novel brain-specific mRNA encoding nuclear protein (Necdin) expressed in neurally differentiated embryonal carcinoma cells. Biochem. Biophys. Res. Com. 178, 291–296 (1991).
Jay, P. et al. Isolation and regional mapping of cDNAs expressed during early human development. Genomics 39, 104–108 (1997).
Mutirangura, A. et al. A complete YAC contig of the Prader-Willi/Angelman chromosome region (15q11–q13) and refined localization of the SNRPN gene. Genomics 18, 546–552 (1993).
Horsthemke, B., Dittrich, B. & Buiting, K. Parent-of-origin-specific DNA methylation and imprinting mutations on human chromosome 15. in Genomic Imprinting: Causes and Consequences (eds Ohlsson, R. et al.) 295–308 (Cambridge University Press, Cambridge, UK, 1995).
LaSalle, J. & Lalande, M. Domain organization of allele-specific replication within the GABRB gene cluster requires a biparental 15q11–13 contribution. Nature Genet. 9, 386–395 (1995).
Vergnaud, G. et al. The use of synthetic tandem repeats to isolate new VNTR loci: cloning of a human hypermutable sequence. Genomics 11, 135–144 (1991).
Aizawa, T., Maruyama, K., Kondo, H. & Yoshikawa, K. Expression of NECDIN, an embryonal carcinoma-derived nuclear protein, in developing mouse brain. Dev. Brain Res. 68, 265–274 (1992).
Maruyama, E. Biochemical characterization of mouse brain necdin. Biochem. J. 314, 895–901 (1996).
Uetsuki, T., Tagaki, K., Sugiura, H. & Yoshikawa, K. Structure and expression of the mouse Necdin gene. J. Biol. Chem. 12, 918–924 (1996).
Hayashi, Y., Matsuyama, K., Tagaki, K., Sugiura, H. & Yoshikawa, K. Arrest of cell growth by necdin, a nuclear protein expressed in postmitotic neurons. Biochem. Biophys. Res. Com. 213, 317–324 (1995).
Watrin, F. et al. The mouse Necdin gene is expressed from the paternal allele only and lies in the 7C region of the mouse chromosome 7, a region synteny to the Prader-Willi syndrome region, fur. J. Hum. Genet. (in the press).
Glenn, C.C. et al. Gene structure, DNA methylation, and imprinted expression of the human SNRPN gene. Am. J. Hum. Genet. 58, 335–346 (1996).
Sun, Y. et al. Breakage in the SNRPN locus in a balanced 46,XY,t(15;19) Prader-Willi syndrome patient. Hum. Mol. Genet. 5, 517–524 (1996).
Schulze, A. et al. Exclusion of SNRPN as a major determinant of Prader-Willi syndrome by a translocation breakpoint. Nature Genet. 12, 452–454 (1996).
Huntriss, D.J., Barr, J.A., Horn, D.A., Williams, D.G. & Latchman, D.S. Mice lacking Snrpn expression show normal regulation of neuronal alternative splicing events. Mol. Biol. Rep. 20, 19–25 (1994).
Clayton-Smith, J. Further evidence for dominant inheritance at the chromosome 15q11-q13 locus in familial Angelman syndrome. Am. J. Med. Genet. 44, 256–260 (1992).
Mutirangura, A., Kuwano, A., Ledbetter, S.A., Chinault, A.C. & Ledbetter, D.H. Dinucleotide repeat polymorphism at the D15S11 locus in the Angelman/Prader–Willi region (AS/PWS) of chromosome 15. Hum. Mol. Genet. 1, 139 (1992).
Glatt, K., Sinnett, D. & Lalande, M. The human g-aminobutyric acid receptor subunit b3 and a5 gene cluster in chromosome 15q11–q13 is rich in highly polymorphic (CA)n repeats. Genomics. 19, 157–160 (1994).
Rougeulle, C., Glatt, H.,& Lalande, M. The Angelman syndrome candidate gene, UBE3A/E6-AP, is imprinted in brain. Nature Genet. 17, 14–15 (1997).
Walker, A.P. et al. A YAC contig in Xp21 containning the adrenal hypoplasia congenita and glycerol kinase deficiency genes. Hum. Mol. Genet. 1, 579–585 (1992).
Ausubel, F.M. et al. eds. Current Protocols in Molecular Biology, 3rd Ed. John Wiley, New York, (1995).
Pinkel, D., Staume, T. & Gray, J.W. Cytogenetic analysis using quantitative, high sensitivity, fluorescence hybridization. Proc. Natl. Acad. Sci. USA. 83, 2934–2938 (1986).
Bergé-Lefranc, J.-L. et al. Characterization of the human jumonji. gene. Hum. Mol. Genet. 5, 1637–1641 (1996).
Gastaldi, M. et al. Increase in mRNAs encoding neonatal II and III sodium channel alpha isoforms during kainate-induced seizures in adult rat hippocampus. Mol. Brain Res. 192, 222–231 (1997).
Saitho, S. et al. Minimal definition of the imprinting center and fixation of a chromosome 15q11-q13 epigenotype by imprinting mutations. Proc. Natl. Acad. Sci. USA. 93, 7811–7815 (1996).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Jay, P., Rougeulle, C., Massacrier, A. et al. The human necdin gene, NDN, is maternally imprinted and located in the Prader-Willi syndrome chromosomal region. Nat Genet 17, 357–361 (1997). https://doi.org/10.1038/ng1197-357
Issue Date:
DOI: https://doi.org/10.1038/ng1197-357
This article is cited by
-
Firing activity of locus coeruleus noradrenergic neurons decreases in necdin-deficient mice, an animal model of Prader–Willi syndrome
Journal of Neurodevelopmental Disorders (2020)
-
Relationship between phenotype and genotype of 102 Chinese newborns with Prader–Willi syndrome
Molecular Biology Reports (2019)
-
Gastro-oesophageal reflux - an important causative factor of severe tooth wear in Prader-Willi syndrome?
Orphanet Journal of Rare Diseases (2018)
-
Promotion of mitochondrial biogenesis by necdin protects neurons against mitochondrial insults
Nature Communications (2016)
-
Physical activity delays hippocampal neurodegeneration and rescues memory deficits in an Alzheimer disease mouse model
Translational Psychiatry (2016)