Abstract
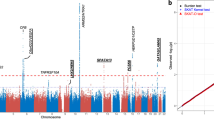
Through whole-genome sequencing of 2,230 Icelanders, we detected a rare nonsynonymous SNP (minor allele frequency = 0.55%) in the C3 gene encoding a p.Lys155Gln substitution in complement factor 3, which, following imputation into a set of Icelandic cases with age-related macular degeneration (AMD) and controls, associated with disease (odds ratio (OR) = 3.45; P = 1.1 × 10−7). This signal is independent of the previously reported common SNPs in C3 encoding p.Pro314Leu and p.Arg102Gly that associate with AMD. The association of p.Lys155Gln was replicated in AMD case-control samples of European ancestry with OR = 4.22 and P = 1.6 × 10−10, resulting in OR = 3.65 and P = 8.8 × 10−16 for all studies combined. In vitro studies have suggested that the p.Lys155Gln substitution reduces C3b binding to complement factor H, potentially creating resistance to inhibition by this factor. This resistance to inhibition in turn is predicted to result in enhanced complement activation.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout

Similar content being viewed by others
Accession codes
References
Friedman, D.S. et al. Prevalence of age-related macular degeneration in the United States. Arch. Ophthalmol. 122, 564–572 (2004).
Hindorff, L.A. et al. Potential etiologic and functional implications of genome-wide association loci for human diseases and traits. Proc. Natl. Acad. Sci. USA 106, 9362–9367 (2009).
Maller, J. et al. Common variation in three genes, including a noncoding variant in CFH, strongly influences risk of age-related macular degeneration. Nat. Genet. 38, 1055–1059 (2006).
Raychaudhuri, S. et al. A rare penetrant mutation in CFH confers high risk of age-related macular degeneration. Nat. Genet. 43, 1232–1236 (2011).
Fritsche, L.G. et al. Seven new loci associated with age-related macular degeneration. Nat. Genet. 45, 433–439 (2013).
Raychaudhuri, S. et al. Associations of CFHR1-CFHR3 deletion and a CFH SNP to age-related macular degeneration are not independent. Nat. Genet. 42, 553–555 (2010).
Yates, J.R. et al. Complement C3 variant and the risk of age-related macular degeneration. N. Engl. J. Med. 357, 553–561 (2007).
Tennessen, J.A. et al. Evolution and functional impact of rare coding variation from deep sequencing of human exomes. Science 337, 64–69 (2012).
Zhan, X. et al. Targeted sequencing, augmented with public resources, identifies a rare complement 3 allele associated with AMD. Nat. Genet. 10.1038/ng.2578 (15 September 2013).
Wu, J. et al. Structure of complement fragment C3b–factor H and implications for host protection by complement regulators. Nat. Immunol. 10, 728–733 (2009).
Miller, E. Characterization of Complement C3 Dysregulation Predisposing to Two Human Disease States, PhD thesis, Washington University (2012).
Kiemeney, L.A. et al. Sequence variant on 8q24 confers susceptibility to urinary bladder cancer. Nat. Genet. 40, 1307–1312 (2008).
Bird, A.C. et al. An international classification and grading system for age-related maculopathy and age-related macular degeneration. The International ARM Epidemiological Study Group. Surv. Ophthalmol. 39, 367–374 (1995).
van Leeuwen, R. et al. Grading of age-related maculopathy for epidemiological studies: is digital imaging as good as 35-mm film? Ophthalmology 110, 1540–1544 (2003).
Kutyavin, I.V. et al. A novel endonuclease IV post-PCR genotyping system. Nucleic Acids Res. 34, e128 (2006).
Yang, Z. et al. A variant of the HTRA1 gene increases susceptibility to age-related macular degeneration. Science 314, 992–993 (2006).
Li, H. & Durbin, R. Fast and accurate short read alignment with Burrows-Wheeler transform. Bioinformatics 25, 1754–1760 (2009).
McKenna, A. et al. The Genome Analysis Toolkit: a MapReduce framework for analyzing next-generation DNA sequencing data. Genome Res. 20, 1297–1303 (2010).
DePristo, M.A. et al. A framework for variation discovery and genotyping using next-generation DNA sequencing data. Nat. Genet. 43, 491–498 (2011).
Cingolani, P. et al. A program for annotating and predicting the effects of single nucleotide polymorphisms, SnpEff: SNPs in the genome of Drosophila melanogaster strain w1118; iso-2; iso-3. Fly (Austin) 6, 80–92 (2012).
Kong, A. et al. Detection of sharing by descent, long-range phasing and haplotype imputation. Nat. Genet. 40, 1068–1075 (2008).
Kong, A. et al. Parental origin of sequence variants associated with complex diseases. Nature 462, 868–874 (2009).
Marchini, J., Howie, B., Myers, S., McVean, G. & Donnelly, P. A new multipoint method for genome-wide association studies by imputation of genotypes. Nat. Genet. 39, 906–913 (2007).
Acknowledgements
The authors would like to thank the individuals who participated in the study and whose contributions made this work possible. We thank our valued colleagues who contributed to data collection, sample handling, genotyping and data analysis. H.L., Y.D. and K.Z. are supported in part by 973 program grant 2013CB967504, National Science Foundation of China grant 81130017, the NEI/US National Institutes of Health and a Veterans Affairs Merit Award. The Dutch replication study was supported by the Netherlands Organization for Scientific Research (Vidi Innovational Research Award 016.096.309 to A.I.d.H.) and the Foundation Fighting Blindness (grant C-GE-0811-0548-RAD04 to A.I.d.H.).
Author information
Authors and Affiliations
Contributions
The study was designed and results interpreted by H.H., P.S., H. Stefansson, G.T., U.T. and K.S. H.H., P.S. and U.T. wrote the first draft and, together with I.J., D.F.G. and K.S. wrote the final version of the manuscript. Statistical analysis was carried out by H.H., P.S., G.T., D.F.G. and A.K. O.T.M. oversaw sequencing for the Icelandic cohort, and G.M. processed the raw sequencing data. Subject recruitment, phenotyping, biological material collection and handling, and genotyping were supervised and carried out by H. Stefansson, G.T., G.B.W., T. Rafnar, A.G., G.H., E.S., F.J., H. Sigurdsson, A.I.d.H., M.R.D., F.E.S.-K., C.J.F.B., S.F., T. Ristau, S.L., J.P.H.v.d.V., C.B.H., K.Z., H.L., L.Z., Y.S., M.P., S.P., H.F., Y.D. and P.S.B. Principal investigators and corresponding authors for the replication study populations are A.I.d.H. (the EUGENDA cohort), K.Z. (the US cohort) and L.A.K. (population controls from Nijmegen).
Corresponding authors
Ethics declarations
Competing interests
The authors declare competing financial interests. H.H., P.S., G.T., H. Stefansson, I.J., G.M., D.F.G., G.B.W., O.T.M., A.K., T.R., U.T. and K.S. are employees of deCODE Genetics/Amgen.
Supplementary information
Supplementary Text and Figures
Supplementary Figure 1 and Supplementary Tables 1–12 (PDF 848 kb)
Rights and permissions
About this article
Cite this article
Helgason, H., Sulem, P., Duvvari, M. et al. A rare nonsynonymous sequence variant in C3 is associated with high risk of age-related macular degeneration. Nat Genet 45, 1371–1374 (2013). https://doi.org/10.1038/ng.2740
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ng.2740
This article is cited by
-
Age-related macular degeneration and premorbid allergic diseases: a population-based case–control study
Scientific Reports (2021)
-
A systems biology approach towards understanding and treating non-neovascular age-related macular degeneration
Nature Communications (2019)
-
Association between genetic variation of complement C3 and the susceptibility to advanced age-related macular degeneration: a meta-analysis
BMC Ophthalmology (2018)
-
Genetics and genetic testing for age-related macular degeneration
Eye (2018)