Abstract
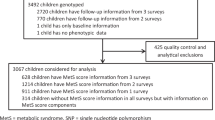
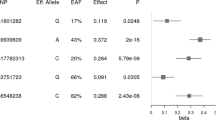
To identify common variants influencing body mass index (BMI), we analyzed genome-wide association data from 16,876 individuals of European descent. After previously reported variants in FTO, the strongest association signal (rs17782313, P = 2.9 × 10−6) mapped 188 kb downstream of MC4R (melanocortin-4 receptor), mutations of which are the leading cause of monogenic severe childhood-onset obesity. We confirmed the BMI association in 60,352 adults (per-allele effect = 0.05 Z-score units; P = 2.8 × 10−15) and 5,988 children aged 7–11 (0.13 Z-score units; P = 1.5 × 10−8). In case-control analyses (n = 10,583), the odds for severe childhood obesity reached 1.30 (P = 8.0 × 10−11). Furthermore, we observed overtransmission of the risk allele to obese offspring in 660 families (P (pedigree disequilibrium test average; PDT-avg) = 2.4 × 10−4). The SNP location and patterns of phenotypic associations are consistent with effects mediated through altered MC4R function. Our findings establish that common variants near MC4R influence fat mass, weight and obesity risk at the population level and reinforce the need for large-scale data integration to identify variants influencing continuous biomedical traits.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Jackson, R.S. et al. Obesity and impaired prohormone processing associated with mutations in the human prohormone convertase 1 gene. Nat. Genet. 16, 303–306 (1997).
Montague, C.T. et al. Congenital leptin deficiency is associated with severe early-onset obesity in humans. Nature 387, 903–908 (1997).
Clement, K. et al. A mutation in the human leptin receptor gene causes obesity and pituitary dysfunction. Nature 392, 398–401 (1998).
Krude, H. et al. Severe early-onset obesity, adrenal insufficiency and red hair pigmentation caused by POMC mutations in humans. Nat. Genet. 19, 155–157 (1998).
Vaisse, C., Clement, K., Guy-Grand, B. & Froguel, P. A frameshift mutation in human MC4R is associated with a dominant form of obesity. Nat. Genet. 20, 113–114 (1998).
Yeo, G.S.H. et al. A frameshift mutation in MC4R associated with dominantly inherited human obesity. Nat. Genet. 20, 111–112 (1998).
Frayling, T.M. et al. A common variant in the FTO gene is associated with body mass index and predisposes to childhood and adult obesity. Science 316, 889–894 (2007).
Dina, C. et al. Variation in FTO contributes to childhood obesity and severe adult obesity. Nat. Genet. 39, 724–726 (2007).
Scuteri, A. et al. Genome-wide association scan shows genetic variants in the FTO gene are associated with obesity-related traits. PLoS Genet. 3, e115 (2007).
Farooqi, I.S. et al. Dominant and recessive inheritance of morbid obesity associated with melanocortin 4 receptor deficiency. J. Clin. Invest. 106, 271–279 (2000).
Marchini, J., Howie, B., Myers, S., McVean, G. & Donnelly, P. A new multipoint method for genome-wide association studies by imputation of genotypes. Nat. Genet. 39, 906–913 (2007).
Young, E.H. et al. The V103I polymorphism of the MC4R gene and obesity: population based studies and meta-analysis of 29,563 individuals. Int. J. Obes. 31, 1437–1441 (2007).
Stutzmann, F. et al. Non-synonymous polymorphisms in melanocortin-4 receptor protect against obesity: the two facets of a Janus obesity gene. Hum. Mol. Genet. 16, 1837–1844 (2007).
Geller, F. et al. Melanocortin 4 receptor gene variant I103 is negatively associated with obesity. Am. J. Hum. Genet. 74, 572–581 (2004).
Kim, J.Y., Ahn, H.J., Ryu, J.H., Suk, K. & Park, J.H. BH3-only protein Noxa is a mediator of hypoxic cell death induced by hypoxia-inducible factor 1α. J. Exp. Med. 199, 113–124 (2004).
Farooqi, I.S. et al. Clinical spectrum of obesity and mutations in the melanocortin 4 receptor gene. N. Engl. J. Med. 348, 1085–1095 (2003).
Huszar, D. et al. Targeted disruption of the melanocortin-4 receptor results in obesity in mice. Cell 88, 131–141 (1997).
Dixon, A.L. et al. A genome-wide association study of global gene expression. Nat. Genet. 39, 1202–1207 (2007).
Stranger, B.E. et al. Population genomics of human gene expression. Nat. Genet. 39, 1217–1224 (2007).
Goring, H.H.H. et al. Discovery of expression QTLs using large-scale transcriptional profiling in human lymphocytes. Nat. Genet. 39, 1208–1216 (2007).
Myers, A.J. et al. A survey of genetic human cortical gene expression. Nat. Genet. 39, 1494–1499 (2007).
Gudmundsson, J. et al. Genome-wide association study identifies a second prostate cancer susceptibility variant at 8q24. Nat. Genet. 39, 631–637 (2007).
Zeggini, E. et al. Replication of genome-wide association signals in UK samples reveals risk loci for type 2 diabetes. Science 316, 1336–1341 (2007).
Scott, L.J. et al. A genome-wide association study of type 2 diabetes in Finns detects multiple susceptibility variants. Science 316, 1341–1345 (2007).
Diabetes Genetics Initiative of Broad Institute of Harvard and MIT, Lund University and Novartis Institutes of BioMedical Research et al. Genome-wide association analysis identifies loci for type 2 diabetes and triglyceride levels. Science 316, 1331–1336 (2007).
The Wellcome Trust Case Control Consortium. Genome-wide association study of 14,000 cases of seven common diseases and 3,000 shared controls. Nature 447, 661–678 (2007).
Day, N.E. et al. EPIC-Norfolk: study design and characteristics of the cohort. European Prospective Investigation of Cancer. Br. J. Cancer 80, 95–103 (1999).
Vollenweider, P. et al. Health examination survey of the Lausanne population: first results of the CoLaus study. Rev. Med. Suisse 2, 2528–2530 (2006).
Chen, W.M. & Abecasis, G. Family-based association tests for genomewide association scans. Am. J. Hum. Genet. 81, 913–926 (2007).
Sutton, A., Abrams, K.R., Jones, D.R., Sheldon, T.A. & Song, F. Methods for Meta-Analysis in Medical Research. (John Wiley & Sons, Chichester, UK, 2000).
Acknowledgements
We acknowledge support of the UK Medical Research Council, the Wellcome Trust, Diabetes UK, Cancer Research United Kingdom, BDA Research, UK National Health Service Research and Development, the European Commission, the Academy of Finland, the British Heart Foundation, the National Institutes of Health, the Novartis Institutes for BioMedical Research, GlaxoSmithKline and the German National Genome Research Net. Personal support was provided by NIDDK (E.K.S., H.N.L., J.N.H., F.S.C.), the Wellcome Trust (A.T.H., E.Z.), Diabetes UK (R.M.F.), the Throne-Holst Foundation (C.M.L.), the Vandervell Foundation (M.N.W.), American Diabetes Association (C.J.W.), Unilever Corporate Research (S.L.) and the British Heart foundation (N.J.S).
Author information
Authors and Affiliations
Consortia
Contributions
Writing team: R.J.F.L., C.M.L., S.L., M.I.M., N.J.W., I.B.
Project management: R.J.F.L., C.M.L., P.D., M.I.M., N.J.W., I.B.
Genome-wide association sampling, genotyping and analysis: R.J.F.L., C.M.L., S.L., E.W., J.H.Z., I.P., M.I., A.P.A., J.S.B., S.B., S.A.B., M. Bochud, M. Brown, J.M.C., D.M.E., J.G., R.G., D.H., A.S.H., A.T.H., S.E.H., T.J., J.D.M.J., A.K., K.-T.K., R.L., M.M., J.M., W.L.M., R.M., P.B.M., A.C.N., K.K.O., K.P., S.P., S.M.R., M.S.S., M.A.S., K.S., N.S., N.J.T., C.W., D.M.W., M.N.W., The Wellcome Trust Case Control Consortium, X.Y., E.Z., D.P.S., W.H.O., M.J.C., N.J.S., T.M.F., P.V., G.W., V.M., P.D., M.I.M., N.J.W., I.B., A.J.B.
Replication samples, genotyping and analysis in adults: R.J.F.L., C.M.L., S.L., R.M.F., A.P.A., A.B., S.C., C.C., G.D.S., I.D., C.D., A.S.F.D., P.E., D.M.E., P.F., C.J.G., A.T.H., M.-R.J., J.D.M.J., F.K., D.M., A.D.M., M.J.N., K.K.O., S.O., K.R.O., C.N.A.P., A.P., A.R.N., S.M.R., M.S.S., M.A.S., H.E.S., N.J.T., M.N.W., V.L.W., E.Z., W.H.O., T.M.F., M.I.M., N.J.W., I.B.
GIANT Consortium samples, genotyping and analysis: S.I.B., K.B.J., S.J.C., R.B.H., I.M.H., C.L., C.G., T.I., T.M., H.-E.W., L.Q., P K., S.E.H., D.J.H., F.B.H, E.K.S., H.N.L., B.F.V., M.R., L.G., J.N.H., M.U., P.S., S.S., G.R.A., G.A., R.N., D.S., C.J.W., A.U. J., J.T., F.S.C., M. Boehnke., K.L.M., FUSION, DGI, PLCO, NHS, SardiNIA.
Replication samples, analysis and genotyping in children: R.M.F., S.C., G.D.S., I.D., C.D., I.S.F., P.F., A.T.H., J.H., A.H., D.M., A.R.N., K.K.O., S.O., S.M.R., A.S., N.J.T., J.H.T., V.L.W., T.M.F., C.I.G.V.
Statistical analysis and informatics: R.J.F.L., C.M.L., S.L., J.H.Z., I.P., R.M.F., S.D., E.T.D., K.S.E., B.H., S.E.H., T.J., J.M., R.M., S.P., J.C.R., N.W.R., A.S., S.A.T., N.J.T., E.Z., M.I.M., N.J.W., I.B.
Corresponding authors
Ethics declarations
Competing interests
Vincent Mooser, Dawn Waterworth and Kijoung Song are full-time employees of GlaxoSmithKline.
Peter Vollenweider and Gérard Waeber received financial support from GlaxoSmithKline to build the CoLaus study.
Additional information
A full list of authors is provided in the Supplementary Note.
A full list of authors is provided at the end of the reference list.
Supplementary information
Supplementary Text and Figures
Supplementary Note, Supplementary Tables 1–7 and Supplementary Figures 1–5 (PDF 1490 kb)
Rights and permissions
About this article
Cite this article
Loos, R., Lindgren, C., Li, S. et al. Common variants near MC4R are associated with fat mass, weight and risk of obesity. Nat Genet 40, 768–775 (2008). https://doi.org/10.1038/ng.140
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ng.140
This article is cited by
-
Combined transcriptome and metabolome analysis reveals breed-specific regulatory mechanisms in Dorper and Tan sheep
BMC Genomics (2024)
-
Targeting the central melanocortin system for the treatment of metabolic disorders
Nature Reviews Endocrinology (2023)
-
Genetic association of the rs17782313 polymorphism with antipsychotic-induced weight gain
Psychopharmacology (2023)
-
Mechanisms and risk factors of metabolic syndrome in children and adolescents
Endocrine (2023)
-
The relationship of genetic risk score with cardiometabolic risk factors: a cross-sectional study
BMC Cardiovascular Disorders (2022)