Abstract
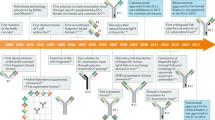
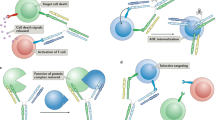
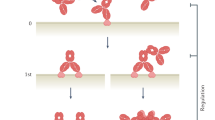
With 18 monoclonal antibody (mAb) products currently on the market and more than 100 in clinical trials, it is clear that engineered antibodies have come of age as biopharmaceuticals. In fact, by 2008, engineered antibodies are predicted to account for >30% of all revenues in the biotechnology market. Smaller recombinant antibody fragments (for example, classic monovalent antibody fragments (Fab, scFv)) and engineered variants (diabodies, triabodies, minibodies and single-domain antibodies) are now emerging as credible alternatives. These fragments retain the targeting specificity of whole mAbs but can be produced more economically and possess other unique and superior properties for a range of diagnostic and therapeutic applications. Antibody fragments have been forged into multivalent and multispecific reagents, linked to therapeutic payloads (such as radionuclides, toxins, enzymes, liposomes and viruses) and engineered for enhanced therapeutic efficacy. Recently, single antibody domains have been engineered and selected as targeting reagents against hitherto immunosilent cavities in enzymes, receptors and infectious agents. Single-domain antibodies are anticipated to significantly expand the repertoire of antibody-based reagents against the vast range of novel biomarkers being discovered through proteomics. As this review aims to show, there is tremendous potential for all antibody fragments either as robust diagnostic reagents (for example in biosensors), or as nonimmunogenic in vivo biopharmaceuticals with superior biodistribution and blood clearance properties.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout

Katie Ris


Similar content being viewed by others
References
Better, M., Chang, C.P., Robinson, R.R. & Horwitz, A.H. Escherichia coli secretion of an active chimeric antibody fragment. Science 240, 1041–1043 (1988).
Huston, J.S. et al. Protein engineering of antibody binding sites: recovery of specific activity in an anti-digoxin single-chain Fv analogue produced in Escherichia coli. Proc. Natl. Acad. Sci. USA 85, 5879–5883 (1988).
Bird, R.E. et al. Single-chain antigen-binding proteins. Science 242, 423–426 (1988).
Holliger, P., Prospero, T. & Winter, G. “Diabodies”: small bivalent and bispecific antibody fragments. Proc. Natl. Acad. Sci. USA 90, 6444–6448 (1993).
Pei, X.Y., Holliger, P., Murzin, A.G. & Williams, R.L. The 2.0-A resolution crystal structure of a trimeric antibody fragment with noncognate VH-VL domain pairs shows a rearrangement of VH CDR3. Proc. Natl. Acad. Sci. USA 94, 9637–9642 (1997).
Iliades, P., Kortt, A.A. & Hudson, P.J. Triabodies: single chain Fv fragments without a linker form trivalent trimers. FEBS Lett. 409, 437–441 (1997).
Ward, E.S., Gussow, D., Griffiths, A.D., Jones, P.T. & Winter, G. Binding activities of a repertoire of single immunoglobulin variable domains secreted from Escherichia coli. Nature 341, 544–546 (1989).
Hudson, P.J. & Souriau, C. Engineered antibodies. Nat. Med. 9, 129–134 (2003).
Woof, J.M. & Burton, D.R. Human antibody-Fc receptor interactions illuminated by crystal structures. Nat. Rev. Immunol. 4, 89–99 (2004).
Ward, E.S. et al. From sorting endosomes to exocytosis: association of Rab4 and Rab11 GTPases with the Fc receptor, FcRn, during recycling. Mol. Biol. Cell 16, 2028–2038 (2005).
Cardoso, R.M. et al. Broadly neutralizing anti-HIV antibody 4E10 recognizes a helical conformation of a highly conserved fusion-associated motif in gp41. Immunity 22, 163–173 (2005).
Zhang, M.Y. et al. Identification and characterization of a new cross-reactive human immunodeficiency virus type 1-neutralizing human monoclonal antibody. J. Virol. 78, 9233–9242 (2004).
Adams, E.J., Chien, Y.H. & Garcia, K.C. Structure of a gammadelta T cell receptor in complex with the nonclassical MHC T22. Science 308, 227–231 (2005).
De Genst, E. et al. Strong in vivo maturation compensates for structurally restricted H3 loops in antibody repertoires. J. Biol. Chem. 280, 14114–14121 (2005).
De Genst, E. et al. Chemical basis for the affinity maturation of a camel single domain antibody. J. Biol. Chem. 279, 53593–53601 (2004).
Dooley, H. & Flajnik, M.F. Shark immunity bites back: affinity maturation and memory response in the nurse shark, Ginglymostoma cirratum. Eur. J. Immunol. 35, 936–945 (2005).
Streltsov, V. & Nuttall, S. Do sharks have a new antibody lineage? Immunol. Lett. 97, 159–160 (2005).
Streltsov, V.A. et al. Structural evidence for evolution of shark Ig new antigen receptor variable domain antibodies from a cell-surface receptor. Proc. Natl. Acad. Sci. USA 101, 12444–12449 (2004).
Stanfield, R.L., Dooley, H., Flajnik, M.F. & Wilson, I.A. Crystal structure of a shark single-domain antibody V region in complex with lysozyme. Science 305, 1770–1773 (2004).
Nuttall, S.D. et al. Selection and affinity maturation of IgNAR variable domains targeting Plasmodium falciparum AMA1. Proteins 55, 187–197 (2004).
Conrath, K. et al. Antigen binding and solubility effects upon the veneering of a camel VHH in framework-2 to mimic a VH. J. Mol. Biol. 350, 112–125 (2005).
Saerens, D. et al. Single domain antibodies derived from dromedary lymph node and peripheral blood lymphocytes sensing conformational variants of prostate-specific antigen. J. Biol. Chem. 279, 51965–51972 (2004).
Pleschberger, M. et al. An S-layer heavy chain camel antibody fusion protein for generation of a nanopatterned sensing layer to detect the prostate-specific antigen by surface plasmon resonance technology. Bioconjug. Chem. 15, 664–671 (2004).
Stijlemans, B. et al. Efficient targeting of conserved cryptic epitopes of infectious agents by single domain antibodies. African trypanosomes as paradigm. J. Biol. Chem. 279, 1256–1261 (2004).
Cortez-Retamozo, V. et al. Efficient cancer therapy with a nanobody-based conjugate. Cancer Res. 64, 2853–2857 (2004).
Dottorini, T., Vaughan, C.K., Walsh, M.A., LoSurdo, P. & Sollazzo, M. Crystal structure of a human VH: requirements for maintaining a monomeric fragment. Biochemistry 43, 622–628 (2004).
Colby, D.W. et al. Development of a human light chain variable domain (V(L)) intracellular antibody specific for the amino terminus of huntingtin via yeast surface display. J. Mol. Biol. 342, 901–912 (2004).
Jespers, L., Schon, O., James, L.C., Veprintsev, D. & Winter, G. Crystal structure of HEL4, a soluble, refoldable human V(H) single domain with a germ-line scaffold. J. Mol. Biol. 337, 893–903 (2004).
Jespers, L., Schon, O., Famm, K. & Winter, G. Aggregation-resistant domain antibodies selected on phage by heat denaturation. Nat. Biotechnol. 22, 1161–1165 (2004).
Souriau, C., Chiche, L., Irving, R. & Hudson, P. New binding specificities derived from Min-23, a small cystine-stabilized peptidic scaffold. Biochemistry 44, 7143–7155 (2005).
Binz, H.K. & Pluckthun, A. Engineered proteins as specific binding reagents. Curr. Opin. Biotechnol. 16, 459–469 (2005).
Midelfort, K.S. et al. Substantial energetic improvement with minimal structural perturbation in a high affinity mutant antibody. J. Mol. Biol. 343, 685–701 (2004).
Moroncini, G. et al. Motif-grafted antibodies containing the replicative interface of cellular PrP are specific for PrPSc. Proc. Natl. Acad. Sci. USA 101, 10404–10409 (2004).
James, L.C., Roversi, P. & Tawfik, D.S. Antibody multispecificity mediated by conformational diversity. Science 299, 1362–1367 (2003).
Linsley, P.S. New look at an old costimulator. Nat. Immunol. 6, 231–232 (2005).
Teeling, J.L. et al. Characterization of new human CD20 monoclonal antibodies with potent cytolytic activity against non-Hodgkin lymphomas. Blood 104, 1793–1800 (2004).
Casey, J.L. et al. Tumour targeting of humanised cross-linked divalent-Fab′ antibody fragments: a clinical phase I/II study. Br. J. Cancer 86, 1401–1410 (2002).
Weir, A.N. et al. Formatting antibody fragments to mediate specific therapeutic functions. Biochem. Soc. Trans. 30, 512–516 (2002).
Dolezal, O. et al. Single-chain Fv multimers of the anti-neuraminidase antibody NC10: the residue at position 15 in the V(L) domain of the scFv-0 (V(L)-V(H)) molecule is primarily responsible for formation of a tetramer-trimer equilibrium. Protein Eng. 16, 47–56 (2003).
Power, B.E., Kortt, A.A. & Hudson, P.J. Generation of recombinant multimeric antibody fragments for tumor diagnosis and therapy. Methods Mol. Biol. 207, 335–350 (2003).
Arndt, M.A., Krauss, J. & Rybak, S.M. Antigen binding and stability properties of non-covalently linked anti-CD22 single-chain Fv dimers. FEBS Lett. 578, 257–261 (2004).
Griffiths, G.L. et al. Reagents and methods for PET using bispecific antibody pretargeting and 68Ga-radiolabeled bivalent hapten-peptide-chelate conjugates. J. Nucl. Med. 45, 30–39 (2004).
Olafsen, T. et al. Covalent disulfide-linked anti-CEA diabody allows site-specific conjugation and radiolabeling for tumor targeting applications. Protein Eng. Des. Sel. 17, 21–27 (2004).
Wittel, U.A. et al. The in vivo characteristics of genetically engineered divalent and tetravalent single-chain antibody constructs. Nucl. Med. Biol. 32, 157–164 (2005).
Le Gall, F., Reusch, U., Little, M. & Kipriyanov, S.M. Effect of linker sequences between the antibody variable domains on the formation, stability and biological activity of a bispecific tandem diabody. Protein Eng. Des. Sel. 17, 357–366 (2004).
Rosenthal, P.B., Waddington, L.J. & Hudson, P.J. Structure of an influenza neuraminidase-diabody complex by electron cryomicroscopy and image analysis. J. Mol. Biol. 334, 721–731 (2003).
Carmichael, J.A. et al. The crystal structure of an anti-CEA scFv diabody assembled from T84.66 scFvs in V(L)-to-V(H) orientation: implications for diabody flexibility. J. Mol. Biol. 326, 341–351 (2003).
Kenanova, V. et al. Tailoring the pharmacokinetics and positron emission tomography imaging properties of anti-carcinoembryonic antigen single-chain Fv-Fc antibody fragments. Cancer Res. 65, 622–631 (2005).
Robinson, M.K. et al. Quantitative immuno-positron emission tomography imaging of HER2-positive tumor xenografts with an iodine-124 labeled anti-HER2 diabody. Cancer Res. 65, 1471–1478 (2005).
Sundaresan, G. et al. 124I-labeled engineered anti-CEA minibodies and diabodies allow high-contrast, antigen-specific small-animal PET imaging of xenografts in athymic mice. J. Nucl. Med. 44, 1962–1969 (2003).
Jain, R.K. & Baxter, L.T. Mechanisms of heterogeneous distribution of monoclonal antibodies and other macromolecules in tumors: significance of elevated interstitial pressure. Cancer Res. 48, 7022–7032 (1988).
Rao, B.M., Lauffenburger, D.A. & Wittrup, K.D. Integrating cell-level kinetic modeling into the design of engineered protein therapeutics. Nat. Biotechnol. 23, 191–194 (2005).
Oh, P. et al. Subtractive proteomic mapping of the endothelial surface in lung and solid tumours for tissue-specific therapy. Nature 429, 629–635 (2004).
Adams, G.P. et al. A single treatment of yttrium-90-labeled CHX-A”-C6.5 diabody inhibits the growth of established human tumor xenografts in immunodeficient mice. Cancer Res. 64, 6200–6206 (2004).
Matter, C.M. et al. Molecular imaging of atherosclerotic plaques using a human antibody against the extra-domain B of fibronectin. Circ. Res. 95, 1225–1233 (2004).
Brack, S.S., Dinkelborg, L.M. & Neri, D. Molecular targeting of angiogenesis for imaging and therapy. Eur. J. Nucl. Med. Mol. Imaging 31, 1327–1341 (2004).
Olafsen, T. et al. Characterization of engineered anti-p185HER-2 (scFv-CH3)2 antibody fragments (minibodies) for tumor targeting. Protein Eng. Des. Sel. 17, 315–323 (2004).
Nielsen, U.B., Adams, G.P., Weiner, L.M. & Marks, J.D. Targeting of bivalent anti-ErbB2 diabody antibody fragments to tumor cells is independent of the intrinsic antibody affinity. Cancer Res. 60, 6434–6440 (2000).
Fujimori, K., Fisher, D.R. & Weinstein, J.N. Integrated microscopic-macroscopic pharmacology of monoclonal antibody radioconjugates: the radiation dose distribution. Cancer Res. 51, 4821–4827 (1991).
Knight, D.M. et al. Pharmacodynamic enhancement of the anti-platelet antibody fab abciximab by site-specific pegylation. Platelets 15, 409–418 (2004).
Huhalov, A. & Chester, K.A. Engineered single chain antibody fragments for radioimmunotherapy. Q. J. Nucl. Med. Mol. Imaging 48, 279–288 (2004).
Holliger, P., Wing, M., Pound, J.D., Bohlen, H. & Winter, G. Retargeting serum immunoglobulin with bispecific diabodies. Nat. Biotechnol. 15, 632–636 (1997).
Medzihradszky, K.F. et al. Glycoforms obtained by expression in Pichia pastoris improve cancer targeting potential of a recombinant antibody-enzyme fusion protein. Glycobiology 14, 27–37 (2004).
Sanz, L., Blanco, B. & Alvarez-Vallina, L. Antibodies and gene therapy: teaching old 'magic bullets' new tricks. Trends Immunol. 25, 85–91 (2004).
Afanasieva, T.A. et al. Single-chain antibody and its derivatives directed against vascular endothelial growth factor: application for antiangiogenic gene therapy. Gene Ther. 10, 1850–1859 (2003).
Blanco, B., Holliger, P., Vile, R.G. & Alvarez-Vallina, L. Induction of human T lymphocyte cytotoxicity and inhibition of tumor growth by tumor-specific diabody-based molecules secreted from gene-modified bystander cells. J. Immunol. 171, 1070–1077 (2003).
Lobato, M.N. & Rabbitts, T.H. Intracellular antibodies as specific reagents for functional ablation: future therapeutic molecules. Curr. Mol. Med. 4, 519–528 (2004).
Tanaka, T., Chung, G.T., Forster, A., Lobato, M.N. & Rabbitts, T.H. De novo production of diverse intracellular antibody libraries. Nucleic Acids Res. 31, e23 (2003).
Colby, D.W. et al. Potent inhibition of huntingtin aggregation and cytotoxicity by a disulfide bond-free single-domain intracellular antibody. Proc. Natl. Acad. Sci. USA 101, 17616–17621 (2004).
Jendreyko, N. et al. Intradiabodies, bispecific, tetravalent antibodies for the simultaneous functional knockout of two cell surface receptors. J. Biol. Chem. 278, 47812–47819 (2003).
Zhao, Y., Brown, T.L., Kohler, H. & Muller, S. MTS-conjugated-antiactive caspase 3 antibodies inhibit actinomycin D-induced apoptosis. Apoptosis 8, 631–637 (2003).
Weisbart, R.H. et al. Nuclear delivery of p53 C-terminal peptides into cancer cells using scFv fragments of a monoclonal antibody that penetrates living cells. Cancer Lett. 195, 211–219 (2003).
Carter, P. Bispecific human IgG by design. J. Immunol. Methods 248, 7–15 (2001).
Xie, Z., Guo, N., Yu, M., Hu, M. & Shen, B. A new format of bispecific antibody: highly efficient heterodimerization, expression and tumor cell lysis. J. Immunol. Methods 296, 95–101 (2005).
Zhu, Z., Presta, L.G., Zapata, G. & Carter, P. Remodeling domain interfaces to enhance heterodimer formation. Protein Sci. 6, 781–788 (1997).
Schlereth, B. et al. Eradication of tumors from a human colon cancer cell line and from ovarian cancer metastases in immunodeficient mice by a single-chain Ep-CAM-/CD3-bispecific antibody construct. Cancer Res. 65, 2882–2889 (2005).
Hoffmann, P. et al. Serial killing of tumor cells by cytotoxic T cells redirected with a CD19-/CD3-bispecific single-chain antibody construct. Int. J. Cancer 115, 98–104 (2005).
Kipriyanov, S.M. & Le Gall, F. Recent advances in the generation of bispecific antibodies for tumor immunotherapy. Curr. Opin. Drug Discov. Devel. 7, 233–242 (2004).
Schlenzka, J. et al. Combined effect of recombinant CD19 × CD16 diabody and thalidomide in a preclinical model of human B cell lymphoma. Anticancer Drugs 15, 915–919 (2004).
Haas, C., Lulei, M., Fournier, P., Arnold, A. & Schirrmacher, V. T-cell triggering by CD3- and CD28-binding molecules linked to a human virus-modified tumor cell vaccine. Vaccine 23, 2439–2453 (2005).
Lu, D. et al. A fully human recombinant IgG-like bispecific antibody to both the epidermal growth factor receptor and the insulin-like growth factor receptor for enhanced antitumor activity. J. Biol. Chem. 280, 19665–19672 (2005).
Jimenez, X. et al. A recombinant, fully human, bispecific antibody neutralizes the biological activities mediated by both vascular endothelial growth factor receptors 2 and 3. Mol. Cancer Ther. 4, 427–434 (2005).
Korn, T., Nettelbeck, D.M., Volkel, T., Muller, R. & Kontermann, R.E. Recombinant bispecific antibodies for the targeting of adenoviruses to CEA-expressing tumour cells: a comparative analysis of bacterially expressed single-chain diabody and tandem scFv. J. Gene Med. 6, 642–651 (2004).
Schnyder, A. & Huwyler, J. Drug transport to brain with targeted liposomes. NeuroRx 2, 99–107 (2005).
Biburger, M., Weth, R. & Wels, W.S. A novel bispecific tetravalent antibody fusion protein to target costimulatory activity for T-cell activation to tumor cells overexpressing ErbB2/HER2. J. Mol. Biol. 346, 1299–1311 (2005).
Sharkey, R.M. & Goldenberg, D.M. Perspectives on cancer therapy with radiolabeled monoclonal antibodies. J. Nucl. Med. 46 (Suppl. 1), 115S–127S (2005).
Zhang, M. et al. Pretargeting radioimmunotherapy of a murine model of adult T-cell leukemia with the alpha-emitting radionuclide, bismuth 213. Blood 100, 208–216 (2002).
Nakamura, T. et al. Antibody-targeted cell fusion. Nat. Biotechnol. 22, 331–336 (2004).
Nakamura, T. et al. Rescue and propagation of fully retargeted oncolytic measles viruses. Nat. Biotechnol. 23, 209–214 (2005).
Bang, S., Nagata, S., Onda, M., Kreitman, R.J. & Pastan, I. HA22 (R490A) is a recombinant immunotoxin with increased antitumor activity without an increase in animal toxicity. Clin. Cancer Res. 11, 1545–1550 (2005).
Vallera, D.A. et al. Molecular modification of a recombinant, bivalent anti-human CD3 immunotoxin (Bic3) results in reduced in vivo toxicity in mice. Leuk. Res. 29, 331–341 (2005).
Krauss, J., Arndt, M.A., Vu, B.K., Newton, D.L. & Rybak, S.M. Targeting malignant B-cell lymphoma with a humanized anti-CD22 scFv-angiogenin immunoenzymedouble dagger. Br. J. Haematol. 128, 602–609 (2005).
Halin, C. et al. Enhancement of the antitumor activity of interleukin-12 by targeted delivery to neovasculature. Nat. Biotechnol. 20, 264–269 (2002).
Smallshaw, J.E. et al. Genetic engineering of an immunotoxin to eliminate pulmonary vascular leak in mice. Nat. Biotechnol. 21, 387–391 (2003).
Forsberg, G. et al. Therapy of human non-small-cell lung carcinoma using antibody targeting of a modified superantigen. Br. J. Cancer 85, 129–136 (2001).
Enever, C., Tomlinson, I.M., Lund, J., Levens, M. & Holliger, P. Engineering high affinity superantigens by phage display. J. Mol. Biol. 347, 107–120 (2005).
Brignole, C. et al. Immune cell-mediated antitumor activities of GD2-targeted liposomal c-myb antisense oligonucleotides containing CpG motifs. J. Natl. Cancer Inst. 96, 1171–1180 (2004).
van Broekhoven, C.L., Parish, C.R., Demangel, C., Britton, W.J. & Altin, J.G. Targeting dendritic cells with antigen-containing liposomes: a highly effective procedure for induction of antitumor immunity and for tumor immunotherapy. Cancer Res. 64, 4357–4365 (2004).
Lu, M., Isogawa, M., Xu, Y. & Hilken, G. Immunization with the gene expressing woodchuck hepatitis virus nucleocapsid protein fused to cytotoxic-T-lymphocyte-associated antigen 4 leads to enhanced specific immune responses in mice and woodchucks. J. Virol. 79, 6368–6376 (2005).
Sharma, S.K. et al. Sustained tumor regression of human colorectal cancer xenografts using a multifunctional mannosylated fusion protein in antibody-directed enzyme prodrug therapy. Clin. Cancer Res. 11, 814–825 (2005).
Mayer, A. et al. Modifying an immunogenic epitope on a therapeutic protein: a step towards an improved system for antibody-directed enzyme prodrug therapy (ADEPT). Br. J. Cancer 90, 2402–2410 (2004).
Mosquera, L.A. et al. In vitro and in vivo characterization of a novel antibody-like single-chain TCR human IgG1 fusion protein. J. Immunol. 174, 4381–4388 (2005).
Michalet, X. et al. Quantum dots for live cells, in vivo imaging, and diagnostics. Science 307, 538–544 (2005).
Evans, E.J. et al. Crystal structure of a soluble CD28-Fab complex. Nat. Immunol. 6, 271–279 (2005).
Ravn, P. et al. Multivalent scFv display of phagemid repertoires for the selection of carbohydrate-specific antibodies and its application to the Thomsen-Friedenreich antigen. J. Mol. Biol. 343, 985–996 (2004).
Jespers, L., Bonnert, T.P. & Winter, G. Selection of optical biosensors from chemisynthetic antibody libraries. Protein Eng. Des. Sel. 17, 709–713 (2004).
Sepp, A., Tawfik, D.S. & Griffiths, A.D. Microbead display by in vitro compartmentalisation: selection for binding using flow cytometry. FEBS Lett. 532, 455–458 (2002).
de Wildt, R.M., Tomlinson, I.M., Ong, J.L. & Holliger, P. Isolation of receptor-ligand pairs by capture of long-lived multivalent interaction complexes. Proc. Natl. Acad. Sci. USA 99, 8530–8535 (2002).
Dolk, E. et al. Induced refolding of a temperature denatured llama heavy-chain antibody fragment by its antigen. Proteins 59, 555–564 (2005).
Dolk, E. et al. Isolation of llama antibody fragments for prevention of dandruff by phage display in shampoo. Appl. Environ. Microbiol. 71, 442–450 (2005).
Nuttall, S.D. et al. Isolation and characterization of an IgNAR variable domain specific for the human mitochondrial translocase receptor Tom70. Eur. J. Biochem. 270, 3543–3554 (2003).
Tanaka, T., Lobato, M.N. & Rabbitts, T.H. Single domain intracellular antibodies: a minimal fragment for direct in vivo selection of antigen-specific intrabodies. J. Mol. Biol. 331, 1109–1120 (2003).
Wunderlich, M., Martin, A. & Schmid, F.X. Stabilization of the cold shock protein CspB from Bacillus subtilis by evolutionary optimization of Coulombic interactions. J. Mol. Biol. 347, 1063–1076 (2005).
Dill, K. et al. Immunoassays based on electrochemical detection using microelectrode arrays. Biosens. Bioelectron. 20, 736–742 (2004).
Hamelinck, D. et al. Optimized normalization for antibody microarrays and application to serum-protein profiling. Mol. Cell Proteomics 4, 773–784 (2005).
Orntoft, T.F. & Vestergaard, E.M. Clinical aspects of altered glycosylation of glycoproteins in cancer. Electrophoresis 20, 362–371 (1999).
Prescher, J.A., Dube, D.H. & Bertozzi, C.R. Chemical remodelling of cell surfaces in living animals. Nature 430, 873–877 (2004).
Chambers, R.S. High-throughput antibody production. Curr. Opin. Chem. Biol. 9, 46–50 (2005).
Grosse-Hovest, L. et al. Supraagonistic, bispecific single-chain antibody purified from the serum of cloned, transgenic cows induces T-cell-mediated killing of glioblastoma cells in vitro and in vivo. Int. J. Cancer; published online 7 July 2005. http://www3.interscience.wiley.com/cgi-bin/abstract/110559371/ABSTRACT
Holliger, P. et al. Carcinoembryonic antigen (CEA)-specific T-cell activation in colon carcinoma induced by anti-CD3xanti-CEA bispecific diabodies and B7xantiCEA bspecific fusion proteins. Cancer Res. 59, 2909–2916 (1999).
Pattersen, E.F. et al. UCFS Chimera–a visualization system for exploratory research and analysis. J. Comput. Chem. 25, 1605–1612 (2004)
Olafsen, T. et al. Optimizing radiolabeled engineered anti-p185HER2 antibody fragments for in vivo imaging. Cancer Res. 65, 5907–5916 (2005).
Shen, S. et al. Patient-specific dosimetry of pretargeted radioimmunotherapy using CC49 fusion protein in patients with gastrointestinal malignancies. J. Nucl. Med. 46, 642–651 (2005).
Nellis, D.F. et al. Preclinical manufacture of anti-HER2 liposome-inserting, scFv-PEG-lipid conjugate. 2. Conjugate micelle identity, purity, stability, and potency analysis. Biotechnol. Prog. 21, 221–232 (2005).
Ebbinghaus, C. et al. Engineered vascular-targeting antibody-interferon-gamma fusion protein for cancer therapy. Int. J. Cancer 116, 304–313 (2005).
Wong, J.Y. et al. Pilot trial evaluating an 123I-labeled 80-kilodalton engineered anticarcinoembryonic antigen antibody fragment (cT84.66 minibody) in patients with colorectal cancer. Clin. Cancer Res. 10, 5014–5021 (2004).
Hulstein, J.J. et al. A novel nanobody that detects the gain-of-function phenotype of Von Willebrand factor in adamts13 deficiency and Von Willebrand disease type 2B. Blood 12 July 2005. http://www.bloodjournal.org/cgi/reprint/2005-03-1153v1
Acknowledgements
We thank V. Streltsov, S. Nuttall and J. Atwell for their help in assembling Figures 1 and 2 and A. Wu and G. Adams for allowing us to include their unpublished data in Figure 3. We also thank our colleagues L. Jespers and I. Tomlinson from Domantis Ltd. for sharing unpublished results and L. James (Medical Research Council) for helpful discussion.
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Holliger, P., Hudson, P. Engineered antibody fragments and the rise of single domains. Nat Biotechnol 23, 1126–1136 (2005). https://doi.org/10.1038/nbt1142
Published:
Issue Date:
DOI: https://doi.org/10.1038/nbt1142
This article is cited by
-
Next-Generation Anti-TNFα Agents: The Example of Ozoralizumab
BioDrugs (2024)
-
Gold conjugated nanobodies in a signal-enhanced lateral flow test strip for rapid detection of SARS-CoV-2 S1 antigen in saliva samples
Scientific Reports (2023)
-
Development of a MET-targeted single-chain antibody fragment as an anti-oncogene targeted therapy for breast cancer
Investigational New Drugs (2023)
-
Side-by-Side Comparison of uPAR-Targeting Optical Imaging Antibodies and Antibody Fragments for Fluorescence-Guided Surgery of Solid Tumors
Molecular Imaging and Biology (2023)
-
Engineering an autonomous VH domain to modulate intracellular pathways and to interrogate the eIF4F complex
Nature Communications (2022)