Abstract
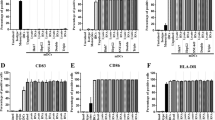
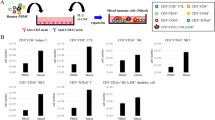
Dendritic cells (DC) generated from the peripheral blood mononuclear cells of healthy individuals or from cancer patients transfected with carcinoembryonic antigen (CEA) mRNA stimulate a potent CD8+ cytotoxic T lymphocyte (CTL) response in vitro. DCs are effectively sensitized with RNA in the absence of reagents commonly used to facilitate mammalian cell transfection. RNA encoding a chimeric CEA/LAMP-1 lysosomal targeting signal enhances the induction of CEA-specific CD4+ T cells, providing a strategy to induce T-help that may be necessary to generate and/or maintain an optimal CD8+ CTL response in vivo. CEA RNA-transfected DCs also serve as effective targets in cytotoxicity assays, thus providing a general method for inducing, as well as measuring, CEA-specific CTL responses across a broad spectrum of HLA haplotypes.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Steinman, R.M. The dendtitic cell system and its role in immunogenicity.Ann. Rev. Immunol. 9: 271–296.
Nair, S., Zhou, F., Reddy, R., Huang, L., and Rouse, B.T. 1992. Solubel proteins delivered to dendritic cells via pH–sensitive liposomes induce primary cytotoxic T lymphocyte responses in vitro. J. Exp. Med. 175: 609–612.
Nair, S. Babu, J.S., Dunham, R.G. Kanda, P., Burke, R.L., and Rouse, B.T. 1993. Induction of primary, antiviral cytotoxic, and proliferative responses with antigens administered via dendritic cells. J. Virol. 67: 4062–4069.
Rouse, R.J.D. Nair, S.K., Lydy, S.L. Bowen, J.C., and Rouse, B.T. 1994. Induction in vitro of primary cytotoxic T-lymphocyte responses with DNA encoding herpes simplex virus proteins. J. Virol. 68: 5685–5689.
Porgador, A. and Gilboa, E. 1995. Bone marrow generated dendritic cells pulsed with a class l-restricted peptide are potent inducers of cytotoxic T lymphocytes. J. Exp. Med. 182: 255–260.
Takahashi, H. Nakagawa, Y., Yokomuro, K., and Berzofsky, J. A. 1993. Induction of CDS+ cytotoxic T lymphocytes by immunization with syngeneic irradiated HIV-1 envelope derived peptide-pulsed cells. Int. Immunol. 5: 849–857.
Zitvogel, L., Mayordomo, J.I., Tjandrawan, T., DeLeo, A.B., Clarke, M.R., Lotze, M.T., and Storkus, W.J. 1996. Therapy of murine tumors with tumor peptide-pulsed dendritic cells: dependence on T cells, B7 costimulation, and T helper cell 1-associated cytokines. J. Exp. Med. 183: 87–97.
Paglia, P., Chiodoni, C., Rodolfo, M., and Colombo, M.P. 1996. Murine dendritic cells loaded in vitro with soluble protein prime CTL against tumor antigen in vivo. J. Exp. Med. 183: 317–322.
Flamand, V., Sornasse, T., Thielemans, K., Demanet, C., Bakkus, H., Bazin, F. et al. 1994. Murine dendritic cells pulsed in vitro with tumor antigen induce tumor resistance in vivo. Eur. J. of Immunol. 24: 605–610.
Porgador, A., Snyder, D., and Gilboa, E. 1996. Induction of antitumor immunity using bone marrow-generated dendritic cells. J. Immumol. 156: 2918–2926.
Mayordomo, J.I., Zorina, T., Storkus, W.J., Zitvogel, L., Celluzzi, C., Falo, L.D. et al. 1995. Bone marrow-derived dendritic cells pulsed with synthetic tumor peptides elicit protective and therapeutic antitumor immunity. Nat. Med. 1: 1297–1302.
Hsu, F.J., Benike, C., Fagnoni, F., Liles, T.M., Czerwinski, D., Taidi, B. et al. 1996. Vaccination of patients with B-cell lymphoma using autologous antigen-pulsed dendritic cells. Nat. Med. 2: 52–58.
Ossevoort, M.A., Feltkamp, M.C.W., van Veen, K.J.H., Melief, C.J.M., and Kast, W.M. 1995. Dendritic cells as carriers for a cytotoxic T-lymphocyte epitope-based peptide vaccine in protection against a human papillomavirus type 16-induced tumor. J. Immunotherapy 18: 86–94.
Nair, S.K., Snyder, D., Rouse, B.T., and Gilboa, E. 1997. Regression of tumors in mice vaccinated with professional antigen presenting cells pulsed with tumor extracts. Int. J. Cancer 70: 706–715.
Boczkowski, D., Nair, S., Snyder, D., and Gilboa, E. 1996. Dendritic cells pulsed with RNA are potent antigen presenting cells in vitro and in vivo. J. Exp. Med. 184: 465–472.
Tsang, K.Y., Zaremba, S., Nieroda, C.A., and Schlom, J. 1995. Generation of human cytotoxic T cells specific for human carcinoembryonic antigen epitopes from patients immunized with recombinant vaccinia-CEA vaccine. J. Natl. Cancer Inst. 87: 982–989.
Celis, E.V., Tsai, S., Crimi, C., DeMars, R., Wentworth, P.A., Chestnut, R.W. et al. 1994. Induction of anti-tumor cytotoxic T lymphocytes in normal humans using primary cultures and synthetic peptide epitopes. Proc. Natl. Acad. Sci. USA 91: 2105–2109.
Gagliardi, M.C., Petrillo, G.D., Salemi, S., Boffa, L., Longobardi, M.G., Dellabona, P. et al. 1995. Presentation of peptides by cultured monocytes or activated T cells allows specific priming of human cytotoxic T lymphocytes in vitro. Int. Immunol. 7: 1741–1752.
Macatonia, S.E., Patterson, S., and Knight, S.C. 1991. Primary proliferative and cytotoxic T-cell responses to HIV induced in vitro by human dendritic cells. Immunology 74: 399–406.
Mehta-Damani, A., Markowicz, S., and Engleman, E.G. 1994. Generation of antigen-specific CD8+ CTLs from naive precursors. J. Immunol 153: 996–1003.
Bakker, A.B.H., Marland, G., de Boer, A.J., Huijbens, R.J.F., Danen, E.H.J., Adema, G.J., and Figdor, C.G. 1995. Generation of antimelanoma cytotoxic T lymphocytes from healthy donors after presentation of melanoma-associated antigen-derived epitopes by dendritic cells in vitro. Cancer Res 55: 5330–5334.
Reeves, M., Royal, R.E., Lam, J.S., Rosenberg, S.A and Hwu, P. 1996. Retroviral transduction of human dendritic cells with a tumor-associated antigen gene. Cancer Res 56: 5672–5677.
Wong, C.W., Morse, M. and Nair, S.K. 1998. Induction of primary, human antigen-specific cytotoxic T lymphocytes in vitro using dendritic cells pulsed with peptides. J. Immunotherapy 21: 32–40.
Stern, P.L. 1996. Immunity to human papillomavirus-associated cervical neoplasia. Adv. Cancer Res 69: 175–211.
Chalfie, M., Tu, Y., Euskirchen, G., Ward, W.W., and Prasher, D.C. 1994. Green fluorescent protein as a marker for gene expression. Science 263: 802–805.
Felgner, J., Bennett, F., and Felgner, P.L. 1993. Cationic lipid-mediated delivery of poiynucleotides. Methods 5: 67–75.
Sallusto, F., Cella, M., Danieli, C., and Lanzavecchia, A. 1995. Dendritic cells use macropinocytosis and the mannose receptor to concentrate macromolecules in the major histocompatibility complex class II compartment: downregulation by cytokines and bacterial products. J. Exp. Med 182: 389–400.
Lanzavecchia, A. 1996. Mechanisms of antigen uptake for presentation. Curr. Opin. Immunol. 8: 348–354.
Morse, M.A., Zhou, L.-J., Tedder, T.F., Lyerly, K. and Smith, C. 1997. Generation of dendritic cells in vitro from peripheral blood mononuclear cells with GM-CSF, IL-4, and TNFα for use in cancer immunotherapy. Ann. Surgery 226: 6–16.
Keene, J. and Forman, J. 1982. Helper activity is required for the in vivo generation of cytotoxic T lymphocytes. J. Exp. Med 155: 768–782.
von Herrath, M.G., Yokoyama, M., Dockter, J., Oldstone, M.B.A., and Whitton, J.L. 1996. CD4-Deficient mice have reduced levels of memory cytotoxic T lymphocytes after immunization and show diminished resistance to subsequent virus challenge. J. Virol 70: 1072–1079.
Bennett, S.R.M., Carbone, F.R., Karamalis, F., Miller, J.F.A.P., and Heath, W.R. 1997. Induction of a CD8+ cytotoxic T lymphocyte response by cross-priming requires cognate CD4+ T cell help. J. Exp. Med. 186: 65–70.
Rock, K.L. and K, Clark 1996. Analysis of the role of MHC class II presentation in the stimulation of cytotoxic T lymphocytes by antigens targeted into the exogenous antigen-MHC class I presentation pathway. J. Immunol 156: 3721–3726.
Wu, T.-C., Guarnier, F.G., Staveley-O'Carroll, K.F., Viscidi, R.P., Levitsky, H.I., Hedrick, K.R. et al. 1995. Engineering an intracellular pathway for major histocompatibility complex class II presentation of antigens. Proc. Natl. Acad. Sci. USA 92: 11671–11675.
Lin, K.-Y., Guarnier, F.G., Staveley-O'Carroll, K.F., Viscidi, R.P., Levitsky, H.I., Hedrick, K.R. et al. 1996. Treatment of established tumors with a novel vaccine that enhances major histocompatibility class II presentation of tumor antigen. Cancer Res. 56: 21–26.
Babu, J.S., Nair, S., Kanda, P., and Rouse, B.T. 1995. Priming for virus-specific CD8+ but not CD4+ cytotoxic T lymphocytes with synthetic lipopeptide is influenced by acylation units and liposome encapsulation. Vaccine 13: 1669–1676.
Siliciano, R.F., Lawton, T., Knall, C., Karr, R.W., Berman, P., Gregory, T. et al. 1988. Analysis of host-virus interactions in AIDS with human T cell clones specific for HIV gp120: effect of HIV genomic heterogeneity and a mechanism for CD4+ cell depletion. Cell 54: 561–575.
Kern, D.E., Klarnet, J.P., Jensen, M.C.V., and Greenberg, P.D. 1986. Requirement for recognition of class II molecules and processed tumor antigen for optimal generation of syngeneic tumor-specific class I restricted CTL. J. Immunol. 136: 4303–4315.
Lechler, R., Aichinger, G., and Lightstone, L. 1996. The endogenous pathway of MHC class II antigen presentation. Immunol. Rev. 151: 51–79.
Salter, R.D., Howell, D.N., and Cresswell, P. 1985. Genes regulating HLA class I antigen expression in T-B lymphoblast hybrids. Immunogenetics 21: 235–246.
Romani, N., Gruner, S., Brang, D., Kampgen, E., Lenz, A., Trockenbacher, B. et al. 1994. Proliferating dendritic cell progenitors in human blood. J. Exp. Med. 180: 83–93.
Volgmann, T., Klein-Struckmeier, A., and Mohr, H. 1989. A fluorescence-based assay for quantitation of lymphokine-activated killer cell activity. J. Immunol. Methods 119: 45–51.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Nair, S., Boczkowski, D., Morse, M. et al. Induction of primary carcinoembryonic antigen (CEA)-specific cytotoxic T lymphocytes in vitro using human dendritic cells transfected with RNA. Nat Biotechnol 16, 364–369 (1998). https://doi.org/10.1038/nbt0498-364
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/nbt0498-364
This article is cited by
-
Self-replicating messenger RNA based cancer immunotherapy
Cancer Gene Therapy (2023)
-
mRNA therapeutics in cancer immunotherapy
Molecular Cancer (2021)
-
Clinical and immunological control of experimental autoimmune encephalomyelitis by tolerogenic dendritic cells loaded with MOG-encoding mRNA
Journal of Neuroinflammation (2019)
-
Calcein Release from Cells In Vitro via Reversible and Irreversible Electroporation
The Journal of Membrane Biology (2018)
-
White paper on microbial anti-cancer therapy and prevention
Journal for ImmunoTherapy of Cancer (2018)