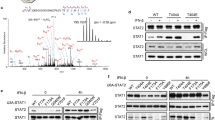
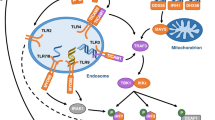
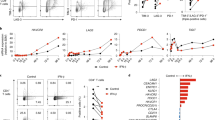
Abstract
Antiviral responses must be tightly regulated to defend rapidly against infection while minimizing inflammatory damage. Type 1 interferons (IFN-I) are crucial mediators of antiviral responses1 and their transcription is regulated by a variety of transcription factors2; principal among these is the family of interferon regulatory factors (IRFs)3. The IRF gene regulatory networks are complex and contain multiple feedback loops. The tools of systems biology are well suited to elucidate the complex interactions that give rise to precise coordination of the interferon response. Here we have used an unbiased systems approach to predict that a member of the forkhead family of transcription factors, FOXO3, is a negative regulator of a subset of antiviral genes. This prediction was validated using macrophages isolated from Foxo3-null mice. Genome-wide location analysis combined with gene deletion studies identified the Irf7 gene as a critical target of FOXO3. FOXO3 was identified as a negative regulator of Irf7 transcription and we have further demonstrated that FOXO3, IRF7 and IFN-I form a coherent feed-forward regulatory circuit. Our data suggest that the FOXO3–IRF7 regulatory circuit represents a novel mechanism for establishing the requisite set points in the interferon pathway that balances the beneficial effects and deleterious sequelae of the antiviral response.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Liu, S. Y., Sanchez, D. J. & Cheng, G. New developments in the induction and antiviral effectors of type I interferon. Curr. Opin. Immunol. 23, 57–64 (2011)
Panne, D., Maniatis, T. & Harrison, S. C. An atomic model of the interferon-beta enhanceosome. Cell 129, 1111–1123 (2007)
Tamura, T., Yanai, H., Savitsky, D. & Taniguchi, T. The IRF family transcription factors in immunity and oncogenesis. Annu. Rev. Immunol. 26, 535–584 (2008)
Barbalat, R., Ewald, S. E., Mouchess, M. L. & Barton, G. M. Nucleic acid recognition by the innate immune system. Annu. Rev. Immunol. 29, 185–214 (2011)
Benayoun, B. A., Caburet, S. & Veitia, R. A. Forkhead transcription factors: key players in health and disease. Trends Genet. 27, 224–232 (2011)
Schoggins, J. W. et al. A diverse range of gene products are effectors of the type I interferon antiviral response. Nature 472, 481–485 (2011)
Honda, K. et al. IRF-7 is the master regulator of type-I interferon-dependent immune responses. Nature 434, 772–777 (2005)
Shahbazian, M. D. & Grunstein, M. Functions of site-specific histone acetylation and deacetylation. Annu. Rev. Biochem. 76, 75–100 (2007)
Szklarczyk, D. et al. The STRING database in 2011: functional interaction networks of proteins, globally integrated and scored. Nucleic Acids Res. 39, D561–D568 (2011)
Khan, N. et al. Determination of the class and isoform selectivity of small-molecule histone deacetylase inhibitors. Biochem. J. 409, 581–589 (2008)
Ning, S., Huye, L. E. & Pagano, J. S. Regulation of the transcriptional activity of the IRF7 promoter by a pathway independent of interferon signaling. J. Biol. Chem. 280, 12262–12270 (2005)
Brunet, A. et al. Akt promotes cell survival by phosphorylating and inhibiting a Forkhead transcription factor. Cell 96, 857–868 (1999)
Plas, D. R. & Thompson, C. B. Akt activation promotes degradation of tuberin and FOXO3a via the proteasome. J. Biol. Chem. 278, 12361–12366 (2003)
Dejean, A. S. et al. Transcription factor Foxo3 controls the magnitude of T cell immune responses by modulating the function of dendritic cells. Nature Immunol. 10, 504–513 (2009)
Fallarino, F. et al. CTLA-4–Ig activates Forkhead transcription factors and protects dendritic cells from oxidative stress in nonobese diabetic mice. J. Exp. Med. 200, 1051–1062 (2004)
Wang, S. T. et al. RNA interference-mediated silencing of Foxo3 in antigen-presenting cells as a strategy for the enhancement of DNA vaccine potency. Gene Ther. 18, 372–383 (2011)
Ning, S., Pagano, J. S. & Barber, G. N. IRF7: activation, regulation, modification and function. Genes Immun. 10.1038/gene.2011.21.
Kumagai, Y. et al. Alveolar macrophages are the primary interferon-α producer in pulmonary infection with RNA viruses. Immunity 27, 240–252 (2007)
Lyles, D. S. & Rupprecht, C. E. in Fields Virology 5th edn, Vol. 1 (eds Howley, P. M. & Knipe, D. M. ) 1363–1408 (2007)
Sullivan, J. A., Kim, E. H., Plisch, E. H., Peng, S. L. & Suresh, M. FOXO3 regulates CD8 T cell memory by T cell-intrinsic mechanisms. PLoS Pathog. 8, e1002533 (2012)
Gilchrist, M. et al. Systems biology approaches identify ATF3 as a negative regulator of Toll-like receptor 4. Nature 441, 173–178 (2006)
Müller, U. et al. Functional role of type I and type II interferons in antiviral defense. Science 264, 1918–1921 (1994)
Honda, K. et al. IRF-7 is the master regulator of type-I interferon-dependent immune responses. Nature 434, 772–777 (2005)
Castrillon, D. H., Miao, L., Kollipara, R., Horner, J. W. & DePinho, R. A. Suppression of ovarian follicle activation in mice by the transcription factor Foxo3a. Science 301, 215–218 (2003)
Irizarry, R. A. et al. Exploration, normalization, and summaries of high density oligonucleotide array probe level data. Biostatistics 4, 249–264 (2003)
Gentleman, R. C. e. t. a. l. Bioconductor: open software development for computational biology and bioinformatics. Genome Biol. 5, research80 (2004)
Parkinson, H. et al. ArrayExpress–a public database of microarray experiments and gene expression profiles. Nucleic Acids Res. 35, D747–D750 (2007)
Chiang, D. Y., Brown, P. O. & Eisen, M. B. Visualizing associations between genome sequences and gene expression data using genome-mean expression profiles. Bioinformatics 17, S49–S55 (2001)
Dollar, P. Piotr Dollar’s Image and Video Toolbox for Matlab (University of California, San Diego, 2006)
Ramsey, S. A. et al. Uncovering a macrophage transcriptional program by integrating evidence from motif scanning and expression dynamics. PLOS Comput. Biol. 4, e1000021 (2008)
Subramanian, A. et al. Gene set enrichment analysis: a knowledge-based approach for interpreting genome-wide expression profiles. Proc. Natl Acad. Sci. USA 102, 15545–15550 (2005)
Gilchrist, M., McCauley, S. D. & Befus, A. D. Expression, localization, and regulation of NOS in human mast cell lines: effects on leukotriene production. Blood 104, 462–469 (2004)
Thijs, G. et al. INCLUSive: integrated clustering, upstream sequence retrieval and motif sampling. Bioinformatics 18, 331–332 (2002)
Biobase. TRANSFAC Professional v9. 3 (http://www.biobase.de)
Ramsey, S. A. et al. Genome-wide histone acetylation data improve prediction of mammalian transcription factor binding sites. Bioinformatics 26, 2071–2075 (2010)
Renault, V. M. et al. FoxO3 regulates neural stem cell homeostasis. Cell Stem Cell 5, 527–539 (2009)
McLaren, L. C., Holland, J. J. & Syverton, J. T. The mammalian cell–virus relationship. I. Attachment of poliovirus to cultivated cells of primate and non-primate origin. J. Exp. Med. 109, 475–485 (1959)
Smith, K. D. et al. Toll-like receptor 5 recognizes a conserved site on flagellin required for protofilament formation and bacterial motility. Nature Immunol. 4, 1247–1253 (2003)
Acknowledgements
We thank K. A. Kennedy and J. J. Peschon for discussions and critical reading of the manuscript; and S. A. Danziger, T. Stolyar and E. van Gaver for technical assistance. This work was supported by grants and contracts from the National Institutes of Health (R01AI025032, R01AI032972, HHSN272200700038C, HHSN272200800058C and U54GM103511).
Author information
Authors and Affiliations
Contributions
V.L. designed experiments, carried out all experimental studies and drafted the manuscript; A.V.R. performed data mining and microarray data analysis; A.E.L. provided technical assistance for experiments, including quantitative real-time PCR, ChIP and in vivo studies; F.S. carried out western blots; A.C.H. and A.R. performed ChIP-Seq data analysis; A.G.R. did genome-wide motif scanning analysis; A.B. did in vivo studies; J.D.A. supervised the computational analysis and A.A. supervised the study and wrote the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Information
This file contains Supplementary Figures 1-14 and full Legends for Supplementary Tables 1-10. (PDF 1139 kb)
Supplementary Data
This file contains Supplementary Tables 1-5 (see Supplementary Information file for full Supplementary Table Legends). (XLS 398 kb)
Supplementary Data
This file contains Supplementary Tables 6-10 (see Supplementary Information file for full Supplementary Table Legends). (XLS 508 kb)
Rights and permissions
About this article
Cite this article
Litvak, V., Ratushny, A., Lampano, A. et al. A FOXO3–IRF7 gene regulatory circuit limits inflammatory sequelae of antiviral responses. Nature 490, 421–425 (2012). https://doi.org/10.1038/nature11428
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature11428
This article is cited by
-
Epigenomic and transcriptomic analyses reveal differences between low-grade inflammation and severe exhaustion in LPS-challenged murine monocytes
Communications Biology (2022)
-
Screening of organoids derived from patients with breast cancer implicates the repressor NCOR2 in cytotoxic stress response and antitumor immunity
Nature Cancer (2022)
-
Amplification of immunity by engineering chicken MDA5 combined with the C terminal domain (CTD) of RIG-I
Applied Microbiology and Biotechnology (2022)
-
FoxO3 might be involved in the inflammatory response of human monocytes to lipopolysaccharide through regulating expression of toll like receptor 4
Molecular Biology Reports (2022)
-
SCD2-mediated monounsaturated fatty acid metabolism regulates cGAS-STING-dependent type I IFN responses in CD4+ T cells
Communications Biology (2021)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.