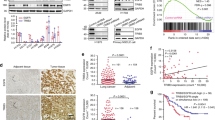
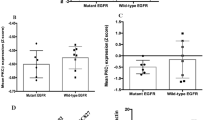
Abstract
Human lung adenocarcinomas with activating mutations in EGFR (epidermal growth factor receptor) often respond to treatment with EGFR tyrosine kinase inhibitors (TKIs), but the magnitude of tumour regression is variable and transient1,2. This heterogeneity in treatment response could result from genetic modifiers that regulate the degree to which tumour cells are dependent on mutant EGFR. Through a pooled RNA interference screen, we show that knockdown of FAS and several components of the NF-κB pathway specifically enhanced cell death induced by the EGFR TKI erlotinib in EGFR-mutant lung cancer cells. Activation of NF-κB through overexpression of c-FLIP or IKK (also known as CFLAR and IKBKB, respectively), or silencing of IκB (also known as NFKBIA), rescued EGFR-mutant lung cancer cells from EGFR TKI treatment. Genetic or pharmacologic inhibition of NF-κB enhanced erlotinib-induced apoptosis in erlotinib-sensitive and erlotinib-resistant EGFR-mutant lung cancer models. Increased expression of the NF-κB inhibitor IκB predicted for improved response and survival in EGFR-mutant lung cancer patients treated with EGFR TKI. These data identify NF-κB as a potential companion drug target, together with EGFR, in EGFR-mutant lung cancers and provide insight into the mechanisms by which tumour cells escape from oncogene dependence.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Haber, D. A. et al. Molecular targeted therapy of lung cancer: EGFR mutations and response to EGFR inhibitors. Cold Spring Harb. Symp. Quant. Biol. 70, 419–426 (2005)
Miller, V. A. et al. Molecular characteristics of bronchioloalveolar carcinoma and adenocarcinoma, bronchioloalveolar carcinoma subtype, predict response to erlotinib. J. Clin. Oncol. 26, 1472–1478 (2008)
Weinstein, I. B. & Joe, A. Oncogene addiction. Cancer Res. 68, 3077–3080 (2008)
Knight, Z. A., Lin, H. & Shokat, K. M. Targeting the cancer kinome through polypharmacology. Nature Rev. Cancer 10, 130–137 (2010)
Sos, M. L. et al. PTEN loss contributes to erlotinib resistance in EGFR-mutant lung cancer by activation of Akt and EGFR. Cancer Res. 69, 3256–3261 (2009)
Silva, J. M. et al. Profiling essential genes in human mammary cells by multiplex RNAi screening. Science 319, 617–620 (2008)
Peter, M. E. et al. The CD95 receptor: apoptosis revisited. Cell 129, 447–450 (2007)
Chen, L. et al. CD95 promotes tumour growth. Nature 465, 492–496 (2010)
O’Reilly, L. A. et al. Membrane-bound Fas ligand only is essential for Fas-induced apoptosis. Nature 461, 659–663 (2009)
Green, D. R. Cancer: a wolf in wolf’s clothing. Nature 465, 433 (2010)
Gong, Y. et al. High expression levels of total IGF-1R and sensitivity of NSCLC cells in vitro to an anti-IGF-1R antibody (R1507). PLoS ONE 4, e7273 (2009)
Sato, M. et al. Multiple oncogenic changes (K-RASV12, p53 knockdown, mutant EGFRs, p16 bypass, telomerase) are not sufficient to confer a full malignant phenotype on human bronchial epithelial cells. Cancer Res. 66, 2116–2128 (2006)
Meylan, E. et al. Requirement for NF-κB signalling in a mouse model of lung adenocarcinoma. Nature 462, 104–107 (2009)
Luo, J. L., Kamata, H. & Karin, M. IKK/NF-κB signaling: balancing life and death–a new approach to cancer therapy. J. Clin. Invest. 115, 2625–2632 (2005)
Pao, W. et al. Acquired resistance of lung adenocarcinomas to gefitinib or erlotinib is associated with a second mutation in the EGFR kinase domain. PLoS Med. 2, e73 (2005)
Bean, J. et al. MET amplification occurs with or without T790M mutations in EGFR mutant lung tumors with acquired resistance to gefitinib or erlotinib. Proc. Natl Acad. Sci. USA 104, 20932–20937 (2007)
Engelman, J. A. et al. Allelic dilution obscures detection of a biologically significant resistance mutation in EGFR-amplified lung cancer. J. Clin. Invest. 116, 2695–2706 (2006)
Turke, A. B. et al. Preexistence and clonal selection of MET amplification in EGFR mutant NSCLC. Cancer Cell 17, 77–88 (2010)
Johannessen, C. M. et al. COT drives resistance to RAF inhibition through MAP kinase pathway reactivation. Nature 468, 968–972 (2010)
Nazarian, R. et al. Melanomas acquire resistance to B-RAF(V600E) inhibition by RTK or N-RAS upregulation. Nature 468, 973–977 (2010)
Yang, J., Amiri, K. I., Burke, J. R., Schmid, J. A. & Richmond, A. BMS-345541 targets inhibitor of κB kinase and induces apoptosis in melanoma: involvement of nuclear factor κB and mitochondria pathways. Clin. Cancer Res. 12, 950–960 (2006)
Rosell, R. et al. Screening for epidermal growth factor receptor mutations in lung cancer. N. Engl. J. Med. 361, 958–967 (2009)
Chitale, D. et al. An integrated genomic analysis of lung cancer reveals loss of DUSP4 in EGFR-mutant tumors. Oncogene 28, 2773–2783 (2009)
Acknowledgements
We thank W. Pao, H. Varmus and members of the MSKCC Lung Cancer Oncogenome group for intellectual support. We thank J. Javier Sanchez for analysing human lung cancer gene expression data. We thank J. Wongvipat and E. Philips for technical assistance and W. Polkinghorn and members of the Sawyers Laboratory for critique of the manuscript. T.G.B. is supported by the MSKCC Clinical Scholars Fellowship funded by the Charles A. Dana Foundation, an ASCO YIA, and the Caine Halter Lung Cancer Research Fund/Uniting Against Lung Cancer Research Grant. C.L.S. and G.H. are Investigators of the Howard Hughes Medical Institute.
Author information
Authors and Affiliations
Contributions
T.G.B. and H.H. designed research, performed experiments, analysed data and co-wrote the paper. K.C., P.M., K.D., M.T. and C.C. provided reagents, performed experiments and analysed data. J.P. and V.A.M. analysed data. R.R., G.H. and C.L.S. designed research, analysed experiments and co-wrote the paper.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Information
The file contains Supplementary Tables 1-5, Supplementary Figures 1-16 with legends, Supplementary Methods and additional references. (PDF 4764 kb)
Rights and permissions
About this article
Cite this article
Bivona, T., Hieronymus, H., Parker, J. et al. FAS and NF-κB signalling modulate dependence of lung cancers on mutant EGFR. Nature 471, 523–526 (2011). https://doi.org/10.1038/nature09870
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature09870
This article is cited by
-
TP53 gain-of-function mutations promote osimertinib resistance via TNF-α–NF-κB signaling in EGFR-mutated lung cancer
npj Precision Oncology (2024)
-
Identification of acquired Notch3 dependency in metastatic Head and Neck Cancer
Communications Biology (2023)
-
Treatment of advanced non-small cell lung cancer with driver mutations: current applications and future directions
Frontiers of Medicine (2023)
-
Therapy-induced APOBEC3A drives evolution of persistent cancer cells
Nature (2023)
-
Osteosclerotic change as a therapeutic response to gefitinib in symptomatic non-small cell lung cancer bone metastasis
BMC Pulmonary Medicine (2022)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.