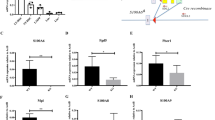
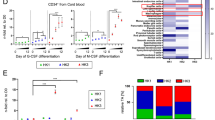
Abstract
Little is known about metabolic regulation in stem cells and how this modulates tissue regeneration or tumour suppression. We studied the Lkb1 tumour suppressor and its substrate AMP-activated protein kinase (AMPK), kinases that coordinate metabolism with cell growth. Deletion of the Lkb1 (also called Stk11) gene in mice caused increased haematopoietic stem cell (HSC) division, rapid HSC depletion and pancytopenia. HSCs depended more acutely on Lkb1 for cell-cycle regulation and survival than many other haematopoietic cells. HSC depletion did not depend on mTOR activation or oxidative stress. Lkb1-deficient HSCs, but not myeloid progenitors, had reduced mitochondrial membrane potential and ATP levels. HSCs deficient for two catalytic α-subunits of AMPK (AMPK-deficient HSCs) showed similar changes in mitochondrial function but remained able to reconstitute irradiated mice. Lkb1-deficient HSCs, but not AMPK-deficient HSCs, exhibited defects in centrosomes and mitotic spindles in culture, and became aneuploid. Lkb1 is therefore required for HSC maintenance through AMPK-dependent and AMPK-independent mechanisms, revealing differences in metabolic and cell-cycle regulation between HSCs and some other haematopoietic progenitors.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Shackelford, D. B. & Shaw, R. J. The LKB1-AMPK pathway: metabolism and growth control in tumour suppression. Nature Rev. Cancer 9, 563–575 (2009)
Alessi, D. R., Sakamoto, K. & Bayascas, J. R. LKB1-dependent signaling pathways. Annu. Rev. Biochem. 75, 137–163 (2006)
Inoki, K., Zhu, T. & Guan, K. L. TSC2 mediates cellular energy response to control cell growth and survival. Cell 115, 577–590 (2003)
Corradetti, M. N., Inoki, K., Bardeesy, N., DePinho, R. A. & Guan, K. L. Regulation of the TSC pathway by LKB1: evidence of a molecular link between tuberous sclerosis complex and Peutz-Jeghers syndrome. Genes Dev. 18, 1533–1538 (2004)
Gwinn, D. M. et al. AMPK phosphorylation of raptor mediates a metabolic checkpoint. Mol. Cell 30, 214–226 (2008)
Greer, E. L. et al. The energy sensor AMP-activated protein kinase directly regulates the mammalian FOXO3 transcription factor. J. Biol. Chem. 282, 30107–30119 (2007)
Cantó, C. et al. AMPK regulates energy expenditure by modulating NAD+ metabolism and SIRT1 activity. Nature 458, 1056–1060 (2009)
Salih, D. A. & Brunet, A. FoxO transcription factors in the maintenance of cellular homeostasis during aging. Curr. Opin. Cell Biol. 20, 126–136 (2008)
Watts, J. L., Morton, D. G., Bestman, J. & Kemphues, K. J. The C. elegans par-4 gene encodes a putative serine-threonine kinase required for establishing embryonic asymmetry. Development 127, 1467–1475 (2000)
Lee, J. H. et al. Energy-dependent regulation of cell structure by AMP-activated protein kinase. Nature 447, 1017–1020 (2007)
Martin, S. G. & St Johnston, D. A role for Drosophila LKB1 in anterior-posterior axis formation and epithelial polarity. Nature 421, 379–384 (2003)
Bonaccorsi, S. et al. The Drosophila Lkb1 kinase is required for spindle formation and asymmetric neuroblast division. Development 134, 2183–2193 (2007)
Ylikorkala, A. et al. Vascular abnormalities and deregulation of VEGF in Lkb1-deficient mice. Science 293, 1323–1326 (2001)
Jishage, K. et al. Role of Lkb1, the causative gene of Peutz-Jegher’s syndrome, in embryogenesis and polyposis. Proc. Natl Acad. Sci. USA 99, 8903–8908 (2002)
Sakamoto, K. et al. Deficiency of LKB1 in skeletal muscle prevents AMPK activation and glucose uptake during contraction. EMBO J. 24, 1810–1820 (2005)
Sakamoto, K. et al. Deficiency of LKB1 in heart prevents ischemia-mediated activation of AMPKα2 but not AMPKα1. Am. J. Physiol. Endocrinol. Metab. 290, E780–E788 (2006)
Shaw, R. J. et al. The kinase LKB1 mediates glucose homeostasis in liver and therapeutic effects of metformin. Science 310, 1642–1646 (2005)
Fu, A. et al. Loss of Lkb1 in adult β cells increases β cell mass and enhances glucose tolerance in mice. Cell Metab. 10, 285–295 (2009)
Granot, Z. et al. LKB1 regulates pancreatic beta cell size, polarity, and function. Cell Metab. 10, 296–308 (2009)
Hezel, A. F. et al. Pancreatic LKB1 deletion leads to acinar polarity defects and cystic neoplasms. Mol. Cell. Biol. 28, 2414–2425 (2008)
Tamás, P. et al. LKB1 is essential for the proliferation of T cell progenitors and mature peripheral T cells. Eur. J. Immunol. 40, 242–253 (2010)
Cao, Y. et al. The serine/threonine kinase LKB1 controls thymocyte survival through regulation of AMPK activation and Bcl-XL expression. Cell Res. 20, 99–108 (2010)
Barnes, A. P. et al. LKB1 and SAD kinases define a pathway required for the polarization of cortical neurons. Cell 129, 549–563 (2007)
Shorning, B. Y. et al. Lkb1 deficiency alters goblet and paneth cell differentiation in the small intestine. PLoS ONE 4, e4264 (2009)
Contreras, C. M. et al. Loss of Lkb1 provokes highly invasive endometrial adenocarcinomas. Cancer Res. 68, 759–766 (2008)
Pearson, H. B., McCarthy, A., Collins, C. M., Ashworth, A. & Clarke, A. R. Lkb1 deficiency causes prostate neoplasia in the mouse. Cancer Res. 68, 2223–2232 (2008)
Gurumurthy, S. et al. LKB1 deficiency sensitizes mice to carcinogen-induced tumorigenesis. Cancer Res. 68, 55–63 (2008)
Bardeesy, N. et al. Loss of the Lkb1 tumour suppressor provokes intestinal polyposis but resistance to transformation. Nature 419, 162–167 (2002)
Jenne, D. E. et al. Peutz-Jeghers syndrome is caused by mutations in a novel serine threonine kinase. Nature Genet. 18, 38–43 (1998)
Hemminki, A. et al. A serine/threonine kinase gene defective in Peutz-Jeghers syndrome. Nature 391, 184–187 (1998)
Kiel, M. J., Yilmaz, O. H., Iwashita, T., Terhorst, C. & Morrison, S. J. SLAM family receptors distinguish hematopoietic stem and progenitor cells and reveal endothelial niches for stem cells. Cell 121, 1109–1121 (2005)
Kiel, M. J., Yilmaz, O. H. & Morrison, S. J. CD150− cells are transiently reconstituting multipotent progenitors with little or no stem cell activity. Blood 111, 4413–4414 (2008)
Akashi, K., Traver, D., Miyamoto, T. & Weissman, I. L. A clonogenic common myeloid progenitor that gives rise to all myeloid lineages. Nature 404, 193–197 (2000)
Köntgen, F., Suss, G., Stewart, C., Steinmetz, M. & Bluethmann, H. Targeted disruption of the MHC Class-II AA gene in C57BL/6 mice. Int. Immunol. 5, 957–964 (1993)
Kuhn, R., Schwenk, F., Aguet, M. & Rajewsky, K. Inducible gene targeting in mice. Science 269, 1427–1429 (1995)
Yilmaz, O. H. et al. Pten dependence distinguishes haematopoietic stem cells from leukaemia-initiating cells. Nature 441, 475–482 (2006)
Hardie, D. G. AMP-activated/SNF1 protein kinases: conserved guardians of cellular energy. Nature Rev. Mol. Cell Biol. 8, 774–785 (2007)
Schieke, S. M. et al. The mammalian target of rapamycin (mTOR) pathway regulates mitochondrial oxygen consumption and oxidative capacity. J. Biol. Chem. 281, 27643–27652 (2006)
Ruzankina, Y. et al. Deletion of the developmentally essential gene ATR in adult mice leads to age-related phenotypes and stem cell loss. Cell Stem Cell 1, 113–126 (2007)
Miyamoto, K. et al. Foxo3a is essential for maintenance of the hematopoietic stem cell pool. Cell Stem Cell 1, 101–112 (2007)
Tothova, Z. et al. FoxOs are critical mediators of hematopoietic stem cell resistance to physiologic oxidative stress. Cell 128, 325–339 (2007)
Larsson, N. G. et al. Mitochondrial transcription factor A is necessary for mtDNA maintenance and embryogenesis in mice. Nature Genet. 18, 231–236 (1998)
Gan, B. et al. Lkb1 regulates quiescence and metabolic homeostasis of haematopoietic stem cells. Nature 10.1038/nature09595 (this issue)
Gurumurthy, S. et al. The Lkb1 metabolic sensor maintains haematopoietic stem cell survival. Nature 10.1038/nature09572 (this issue)
Holland, A. J. & Cleveland, D. W. Boveri revisited: chromosomal instability, aneuploidy and tumorigenesis. Nature Rev. Mol. Cell Biol. 10, 478–487 (2009)
Liu, P., Jenkins, N. A. & Copeland, N. G. A highly efficient recombineering-based method for generating conditional knockout mutations. Genome Res. 13, 476–484 (2003)
Rodriguez, C. I. et al. High-efficiency deleter mice show that FLPe is an alternative to Cre-loxP. Nature Genet. 25, 139–140 (2000)
Acknowledgements
This work was supported by the Howard Hughes Medical Institute. Flow cytometry was partially supported by the University of Michigan (UM) Comprehensive Cancer National Institutes of Health (NIH) CA46592. D.N. was supported by a postdoctoral fellowship from the Japan Society for the Promotion of Science. We thank A. Prendergast and C. Sifuentes for technical assistance; E. Hughes and the UM Transgenic Animal Model Core for help generating Lkb1fl mice; D. Adams and M. White for flow cytometry; E. Smith for antibody production; C. Mountford, S. Grove and R. Coolon for mouse colony management; and L. Cantley and C. Thompson for discussions.
Author information
Authors and Affiliations
Contributions
D.N. performed all experiments. T.L.S. helped to design and generate the Lkb1fl mice. D.N. and S.J.M. designed and interpreted all experiments and wrote the paper.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Figures
The file contains Supplementary Figures 1-11 with legends. (PDF 882 kb)
Rights and permissions
About this article
Cite this article
Nakada, D., Saunders, T. & Morrison, S. Lkb1 regulates cell cycle and energy metabolism in haematopoietic stem cells. Nature 468, 653–658 (2010). https://doi.org/10.1038/nature09571
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature09571
This article is cited by
-
MITOL deficiency triggers hematopoietic stem cell apoptosis via ER stress response
The EMBO Journal (2024)
-
Induction of mitochondrial recycling reverts age-associated decline of the hematopoietic and immune systems
Nature Aging (2023)
-
Myeloid liver kinase B1 contributes to lung inflammation induced by lipoteichoic acid but not by viable Streptococcus pneumoniae
Respiratory Research (2022)
-
Endothelial cell-specific expression of serine/threonine kinase 11 modulates dendritic cell differentiation
Nature Communications (2022)
-
Chromatin remodeling subunit BRM and valine regulate hematopoietic stem/progenitor cell function and self-renewal via intrinsic and extrinsic effects
Leukemia (2022)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.