Abstract
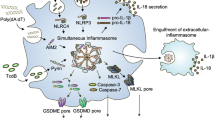
Inflammation is a protective attempt by the host to remove injurious stimuli and initiate the tissue healing process1. The inflammatory response must be actively terminated, however, because failure to do so can result in ‘bystander’ damage to tissues and diseases such as arthritis or type-2 diabetes. Yet the mechanisms controlling excessive inflammatory responses are still poorly understood. Here we show that mouse effector and memory CD4+ T cells abolish macrophage inflammasome-mediated caspase-1 activation and subsequent interleukin 1β release in a cognate manner. Inflammasome inhibition is observed for all tested NLRP1 (commonly called NALP1) and NLRP3 (NALP3 or cryopyrin) activators, whereas NLRC4 (IPAF) inflammasome function and release of other inflammatory mediators such as CXCL2, interleukin 6 and tumour necrosis factor are not affected. Suppression of the NLRP3 inflammasome requires cell-to-cell contact and can be mimicked by macrophage stimulation with selected ligands of the tumour necrosis factor family, such as CD40L (also known as CD40LG). In a NLRP3-dependent peritonitis model, effector CD4+ T cells are responsible for decreasing neutrophil recruitment in an antigen-dependent manner. Our findings reveal an unexpected mechanism of inflammasome inhibition, whereby effector and memory T cells suppress potentially damaging inflammation, yet leave the primary inflammatory response, crucial for the onset of immunity, intact.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Medzhitov, R. Origin and physiological roles of inflammation. Nature 454, 428–435 (2008)
Miyara, M. & Sakaguchi, S. Natural regulatory T cells: mechanisms of suppression. Trends Mol. Med. 13, 108–116 (2007)
Maloy, K. J., Antonelli, L. R., Lefevre, M. & Powrie, F. Cure of innate intestinal immune pathology by CD4+CD25+ regulatory T cells. Immunol. Lett. 97, 189–192 (2005)
Garrett, W. S. et al. Communicable ulcerative colitis induced by T-bet deficiency in the innate immune system. Cell 131, 33–45 (2007)
Kim, K. D. et al. Adaptive immune cells temper initial innate responses. Nature Med. 13, 1248–1252 (2007)
Palm, N. W. & Medzhitov, R. Not so fast: adaptive suppression of innate immunity. Nature Med. 13, 1142–1144 (2007)
Martinon, F., Burns, K. & Tschopp, J. The inflammasome: a molecular platform triggering activation of inflammatory caspases and processing of proIL-beta. Mol. Cell 10, 417–426 (2002)
Martinon, F., Gaide, O., Petrilli, V., Mayor, A. & Tschopp, J. NALP inflammasomes: a central role in innate immunity. Semin. Immunopathol. 29, 213–229 (2007)
Yu, H. B. & Finlay, B. B. The caspase-1 inflammasome: a pilot of innate immune responses. Cell Host Microbe 4, 198–208 (2008)
Sallusto, F., Lenig, D., Forster, R., Lipp, M. & Lanzavecchia, A. Two subsets of memory T lymphocytes with distinct homing potentials and effector functions. Nature 401, 708–712 (1999)
Bromley, S. K., Mempel, T. R. & Luster, A. D. Orchestrating the orchestrators: chemokines in control of T cell traffic. Nature Immunol. 9, 970–980 (2008)
Boyden, E. D. & Dietrich, W. F. NALP1/NLRP1b controls mouse macrophage susceptibility to anthrax lethal toxin. Nature Genet. 38, 240–244 (2006)
Mempel, T. R., Henrickson, S. E. & Von Andrian, U. H. T-cell priming by dendritic cells in lymph nodes occurs in three distinct phases. Nature 427, 154–159 (2004)
Snelgrove, R. J. et al. A critical function for CD200 in lung immune homeostasis and the severity of influenza infection. Nature Immunol. 9, 1074–1083 (2008)
Sun, K. & Metzger, D. W. Inhibition of pulmonary antibacterial defense by interferon-gamma during recovery from influenza infection. Nature Med. 14, 558–564 (2008)
Croft, M. Co-stimulatory members of the TNFR family: keys to effective T-cell immunity? Nature Rev. Immunol. 3, 609–620 (2003)
Watts, T. H. TNF/TNFR family members in costimulation of T cell responses. Annu. Rev. Immunol. 23, 23–68 (2005)
O’Sullivan, B. & Thomas, R. CD40 and dendritic cell function. Crit. Rev. Immunol. 23, 83–107 (2003)
Martin-Fontecha, A. et al. CD40L+ CD4+ memory T cells migrate in a CD62P-dependent fashion into reactive lymph nodes and license dendritic cells for T cell priming. J. Exp. Med. 205, 2561–2574 (2008)
Bossen, C. et al. Interactions of tumor necrosis factor (TNF) and TNF receptor family members in the mouse and human. J. Biol. Chem. 281, 13964–13971 (2006)
Holler, N. et al. Two adjacent trimeric Fas ligands are required for Fas signaling and formation of a death-inducing signaling complex. Mol. Cell. Biol. 23, 1428–1440 (2003)
Kool, M. et al. Cutting edge: alum adjuvant stimulates inflammatory dendritic cells through activation of the NALP3 inflammasome. J. Immunol. 181, 3755–3759 (2008)
Burger, D. & Dayer, J. M. The role of human T-lymphocyte-monocyte contact in inflammation and tissue destruction. Arthritis Res. 4 (Suppl 3). S169–S176 (2002)
McDermott, M. F. & Tschopp, J. From inflammasomes to fevers, crystals and hypertension: how basic research explains inflammatory diseases. Trends Mol. Med. 13, 381–388 (2007)
Martinon, F., Petrilli, V., Mayor, A., Tardivel, A. & Tschopp, J. Gout-associated uric acid crystals activate the NALP3 inflammasome. Nature 440, 237–241 (2006)
Wang, Y. et al. Th2 lymphoproliferative disorder of LatY136F mutant mice unfolds independently of TCR–MHC engagement and is insensitive to the action of Foxp3+ regulatory T cells. J. Immunol. 180, 1565–1575 (2008)
Kawabe, T. et al. The immune responses in CD40-deficient mice: impaired immunoglobulin class switching and germinal center formation. Immunity 1, 167–178 (1994)
Kuida, K. et al. Altered cytokine export and apoptosis in mice deficient in interleukin-1 beta converting enzyme. Science 267, 2000–2003 (1995)
Chen, J. et al. Immunoglobulin gene rearrangement in B cell deficient mice generated by targeted deletion of the JH locus. Int. Immunol. 5, 647–656 (1993)
Acknowledgements
We thank H. Acha-Orbea, P. Launois, E. Luescher, H. R. MacDonald, P. Romero, M. Kopf and R. Flavell for knockout and transgenic mice; H. Acha-Orbea and M. Eckert for critical reading and comments; A. Yazdi for discussions; and M. Braun and D. Labes for technical help. This work was in part supported by the Swiss National Science foundation and the NCCR Molecular Oncology. C.D. is supported by an EMBO long-term fellowship.
Author Contributions G.G. performed and designed the experiments and wrote the paper. C.D., F.S., K.C., R.C. and A.T. contributed to performing the experiments. P.S. provided the TNF ligand reagents and discussed the data throughout the experimental phase. J.T. designed and discussed the experiments and supervised the project.
Author information
Authors and Affiliations
Corresponding author
Supplementary information
Supplementary Figures
This file contains Supplementary Figures 1-12 with Legends. (PDF 2668 kb)
Rights and permissions
About this article
Cite this article
Guarda, G., Dostert, C., Staehli, F. et al. T cells dampen innate immune responses through inhibition of NLRP1 and NLRP3 inflammasomes. Nature 460, 269–273 (2009). https://doi.org/10.1038/nature08100
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature08100
This article is cited by
-
Soluble CD4 effectively prevents excessive TLR activation of resident macrophages in the onset of sepsis
Signal Transduction and Targeted Therapy (2023)
-
Understanding the Role of Inflammasomes in Rheumatoid Arthritis
Inflammation (2020)
-
NLRP3 inflammasome as a potential treatment in ischemic stroke concomitant with diabetes
Journal of Neuroinflammation (2019)
-
Propofol directly induces caspase-1-dependent macrophage pyroptosis through the NLRP3-ASC inflammasome
Cell Death & Disease (2019)
-
Not so crystal clear: observations from a case of crystalline arthritis with cytokine release syndrome (CRS) after chimeric antigen receptor (CAR)-T cell therapy
Bone Marrow Transplantation (2019)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.