Abstract
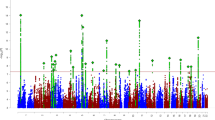
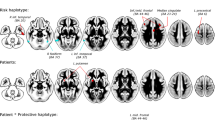
Attention-Deficit/Hyperactivity Disorder (ADHD) has a very high heritability (0.8), suggesting that about 80% of phenotypic variance is due to genetic factors. We used the integration of statistical and functional approaches to discover a novel gene that contributes to ADHD. For our statistical approach, we started with a linkage study based on large multigenerational families in a population isolate, followed by fine mapping of targeted regions using a family-based design. Family- and population-based association studies in five samples from disparate regions of the world were used for replication. Brain imaging studies were performed to evaluate gene function. The linkage study discovered a genome region harbored in the Latrophilin 3 gene (LPHN3). In the world-wide samples (total n=6360, with 2627 ADHD cases and 2531 controls) statistical association of LPHN3 and ADHD was confirmed. Functional studies revealed that LPHN3 variants are expressed in key brain regions related to attention and activity, affect metabolism in neural circuits implicated in ADHD, and are associated with response to stimulant medication. Linkage and replicated association of ADHD with a novel non-candidate gene (LPHN3) provide new insights into the genetics, neurobiology, and treatment of ADHD.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Biederman J, Faraone SV . Attention-deficit hyperactivity disorder. Lancet 2005; 366: 237–248.
Faraone SV, Biederman J, Mennin D, Gershon J, Tsuang MT . A prospective four-year follow-up study of children at risk for ADHD: psychiatric, neuropsychological, and psychosocial outcome. J Am Acad Child Adolesc Psychiatry 1996; 35: 1449–1459.
Pelham WE, Foster EM, Robb JA . The economic impact of attention-deficit/hyperactivity disorder in children and adolescents. J Pediatr Psychol 2007; 32: 711–727.
Palacio JD, Castellanos FX, Pineda DA, Lopera F, Arcos-Burgos M, Quiroz YT et al. Attention-deficit/hyperactivity disorder and comorbidities in 18 Paisa Colombian multigenerational families. J Am Acad Child Adolesc Psychiatry 2004; 43: 1506–1515.
Biederman J, Petty CR, Wilens TE, Fraire MG, Purcell CA, Mick E et al. Familial risk analyses of attention deficit hyperactivity disorder and substance use disorders. Am J Psychiatry 2008; 165: 107–115.
Cook Jr EH, Stein MA, Krasowski MD, Cox NJ, Olkon DM, Kieffer JE et al. Association of attention deficit disorder and the dopamine transporter gene. Am J Hum Genet 1995; 56: 993–998.
LaHoste GJ, Swanson JM, Wigal SB, Glabe C, King N, Kennedy JL et al. Dopamine D4 receptor gene polymorphism is associated with attention deficit hyperactivity disorder. Mol Psychiatry 1996; 1: 121–124.
Thapar A, Langley K, Owen MJ, O’Donovan MC . Advances in genetic findings on attention deficit hyperactivity disorder. Psychol Med 2007; 37: 1681–1692.
Li D, Sham PC, Owen MJ, He L . Meta-analysis shows significant association between dopamine system genes and attention deficit hyperactivity disorder (ADHD). Hum Mol Genet 2006; 15: 2276–2284.
Risch N, Merikangas K . The future of genetic studies of complex human diseases. Science 1996; 273: 1516–1517.
Neale BM, Lasky-Su J, Anney R, Franke B, Zhou K, Maller JB et al. Genome-wide association scan of attention deficit hyperactivity disorder. Am J Med Genet B Neuropsychiatr Genet 2008; 147B: 1337–1344.
Maher BS, Riley BP, Kendler KS . Psychiatric genetics gets a boost. Nat Genet 2008; 40: 1042–1044.
McCarthy MI, Hirschhorn JN . Genome-wide association studies: potential next steps on a genetic journey. Hum Mol Genet 2008; 17: R156–R165.
Arcos-Burgos M, Castellanos FX, Lopera F, Pineda D, Palacio JD, Garcia M et al. Attention-deficit/hyperactivity disorder (ADHD): feasibility of linkage analysis in a genetic isolate using extended and multigenerational pedigrees. Clin Genet 2002; 61: 335–343.
Arcos-Burgos M, Castellanos FX, Pineda D, Lopera F, Palacio JD, Palacio LG et al. Attention-deficit/hyperactivity disorder in a population isolate: linkage to loci at 4q13.2, 5q33.3, 11q22, and 17p11. Am J Hum Genet 2004; 75: 998–1014.
Jain M, Palacio LG, Castellanos FX, Palacio JD, Pineda D, Restrepo MI et al. Attention-deficit/hyperactivity disorder and comorbid disruptive behavior disorders: evidence of pleiotropy and new susceptibility loci. Biol Psychiatry 2007; 61: 1329–1339.
Acosta MT, Castellanos FX, Bolton KL, Balog JZ, Eagen P, Nee L et al. Latent class subtyping of attention-deficit/hyperactivity disorder and comorbid conditions1. J Am Acad Child Adolesc Psychiatry 2008; 47: 797–807.
Elia J, Ambrosini P, Berrettini W . ADHD Characteristics: I. Concurrent Co-morbidity Patterns in Children & Adolescents. Child Adolesc Psychiatry Ment Health 2008; 2: 15.
Walitza S, Renner TJ, Dempfle A, Konrad K, Wewetzer C, Halbach A et al. Transmission disequilibrium of polymorphic variants in the tryptophan hydroxylase-2 gene in attention-deficit/hyperactivity disorder. Mol Psychiatry 2005; 10: 1126–1132.
Johansson S, Halleland H, Halmoy A, Jacobsen KK, Landaas ET, Dramsdahl M et al. Genetic analyses of dopamine related genes in adult ADHD patients suggest an association with the DRD5-microsatellite repeat, but not with DRD4 or SLC6A3 VNTRs. Am J Med Genet B Neuropsychiatr Genet 2008; 147B: 1470–1475.
Ribases M, Ramos-Quiroga JA, Hervas A, Bosch R, Bielsa A, Gastaminza X et al. Exploration of 19 serotoninergic candidate genes in adults and children with attention-deficit/hyperactivity disorder identifies association for 5HT2A, DDC and MAOB. Mol Psychiatry 2009; 14: 71–85.
Service S, DeYoung J, Karayiorgou M, Roos JL, Pretorious H, Bedoya G et al. Magnitude and distribution of linkage disequilibrium in population isolates and implications for genome-wide association studies. Nat Genet 2006; 38: 556–560.
Purcell S, Neale B, Todd-Brown K, Thomas L, Ferreira MA, Bender D et al. PLINK: a tool set for whole-genome association and population-based linkage analyses. Am J Hum Genet 2007; 81: 559–575.
Wigginton JE, Abecasis GR . PEDSTATS: descriptive statistics graphics quality assessment for gene mapping data. Bioinformatics 2005; 21: 3445–3447.
Zollner S, Wen X, Hanchard NA, Herbert MA, Ober C, Pritchard JK . Evidence for extensive transmission distortion in the human genome. Am J Hum Genet 2004; 74: 62–72.
Durrant C, Zondervan KT, Cardon LR, Hunt S, Deloukas P, Morris AP . Linkage disequilibrium mapping via cladistic analysis of single-nucleotide polymorphism haplotypes. Am J Hum Genet 2004; 75: 35–43.
Marchini J, Howie B, Myers S, McVean G, Donnelly P . A new multipoint method for genome-wide association studies by imputation of genotypes. Nat Genet 2007; 39: 906–913.
Spielman RS, McGinnis RE, Ewens WJ . Transmission test for linkage disequilibrium: the insulin gene region and insulin-dependent diabetes mellitus (IDDM). Am J Hum Genet 1993; 52: 506–516.
Kazeem GR, Farrall M . Integrating case-control and TDT studies. Ann Hum Genet 2005; 69: 329–335.
DerSimonian R, Laird N . Meta-analysis in clinical trials. Control Clin Trials 1986; 7: 177–188.
Mantel N, Haenszel W . Statistical aspects of the analysis of data from retrospective studies of disease. J Natl Cancer Inst 1959; 22: 719–748.
Carrey N, MacMaster FP, Fogel J, Sparkes S, Waschbusch D, Sullivan S et al. Metabolite changes resulting from treatment in children with ADHD: a 1H-MRS study. Clin Neuropharmacol 2003; 26: 218–221.
Moore CM, Biederman J, Wozniak J, Mick E, Aleardi M, Wardrop M et al. Differences in brain chemistry in children and adolescents with attention deficit hyperactivity disorder with and without comorbid bipolar disorder: a proton magnetic resonance spectroscopy study. Am J Psychiatry 2006; 163: 316–318.
Sun L, Jin Z, Zang YF, Zeng YW, Liu G, Li Y et al. Differences between attention-deficit disorder with and without hyperactivity: a 1H-magnetic resonance spectroscopy study. Brain Dev 2005; 27: 340–344.
Yeo RA, Hill D, Campbell R, Vigil J, Brooks WM . Developmental instability and working memory ability in children: a magnetic resonance spectroscopy investigation. Dev Neuropsychol 2000; 17: 143–159.
Jin Z, Zang YF, Zeng YW, Zhang L, Wang YF . Striatal neuronal loss or dysfunction and choline rise in children with attention-deficit hyperactivity disorder: a 1H-magnetic resonance spectroscopy study. Neurosci Lett 2001; 315: 45–48.
Provencher SW . Automatic quantitation of localized in vivo 1H spectra with LCModel. NMR Biomed 2001; 14: 260–264.
Castillo M, Smith JK, Kwock L . Correlation of myo-inositol levels and grading of cerebral astrocytomas. AJNR Am J Neuroradiol 2000; 21: 1645–1649.
Swanson JM, Elliott GR, Greenhill LL, Wigal T, Arnold LE, Vitiello B et al. Effects of stimulant medication on growth rates across (3 years in the MTA follow-up. J Am Acad Child Adolesc Psychiatry 2007; 46: 1015–1027.
Vermunt JK, Magidson J . LatentGOLD. [3 01]2003. Statistical Innovations, Inc. 2-10-2003.
Magidson J, Vermunt JK . Latent class analysis. In: Handbook of quantitative methodology for the social sciences, Kaplan D (ed). Sage Publications: Newbury Park, CA, USA, 2003.
Vermunt JK, Magidson J . Latent class cluster analysis. In: Applied latent class analysis, Hagenaars JA & McCutcheon AL (eds) Cambridge University Press, Cambridge, UK, 2002; 89–106.
Hagenaars JA . Latent structure models with direct effects between indicators: local dependence models. Sociol Meth Res 1988; 16: 379–405.
Skol AD, Scott LJ, Abecasis GR, Boehnke M . Joint analysis is more efficient than replication-based analysis for two-stage genome-wide association studies. Nat Genet 2006; 38: 209–213.
Martin ER, Bass MP, Gilbert JR, Pericak-Vance MA, Hauser ER . Genotype-based association test for general pedigrees: the genotype-PDT. Genet Epidemiol 2003; 25: 203–213.
Shapiro MB, Senapathy P . RNA splice junctions of different classes of eukaryotes: sequence statistics functional implications in gene expression. Nucleic Acids Res 1987; 15: 7155–7174.
Plessen KJ, Bansal R, Zhu H, Whiteman R, Amat J, Quackenbush GA et al. Hippocampus and amygdala morphology in attention-deficit/hyperactivity disorder. Arch Gen Psychiatry 2006; 63: 795–807.
Krain AL, Castellanos FX . Brain development and ADHD. Clin Psychol Rev 2006; 26: 433–444.
Anderson CM, Polcari AM, Lowen SB, Renshaw PF, Teicher MH . Effects of methylphenidate on functional magnetic resonance relaxometry of the cerebellar vermis in children with ADHD. Am J Psychiatry 2002; 159: 1322–1328.
Sugita S, Ichtchenko K, Khvotchev M, Sudhof TC . alpha-Latrotoxin receptor CIRL/latrophilin 1 (CL1) defines an unusual family of ubiquitous G-protein-linked receptors. G-protein coupling not required for triggering exocytosis. J Biol Chem 1998; 273: 32715–32724.
Linets'ka MV, Storchak LH, Himmelreich NH . Effect of synaptosomal cytosolic [3H]GABA pool depletion on secretory ability of alpha-latrotoxin]. Ukr Biokhim Zh 2002; 74: 65–72.
Ichtchenko K, Khvotchev M, Kiyatkin N, Simpson L, Sugita S, Sudhof TC . alpha-latrotoxin action probed with recombinant toxin: receptors recruit alpha-latrotoxin but do not transduce an exocytotic signal. EMBO J 1998; 17: 6188–6199.
Faraone SV, Perlis RH, Doyle AE, Smoller JW, Goralnick JJ, Holmgren MA et al. Molecular genetics of attention-deficit/hyperactivity disorder. Biol Psychiatry 2005; 57: 1313–1323.
Tobaben S, Sudhof TC, Stahl B . Genetic analysis of alpha-latrotoxin receptors reveals functional interdependence of CIRL/latrophilin 1 and neurexin 1 alpha. J Biol Chem 2002; 277: 6359–6365.
Benichou J . Methods of adjustment for estimating the attributable risk in case-control studies: a review. Stat Med 1991; 10: 1753–1773.
Benichou J, Gail MH . Methods of inference for estimates of absolute risk derived from population-based case-control studies. Biometrics 1995; 51: 182–194.
Spiegelman D, Hertzmark E, Wand HC . Point and interval estimates of partial population attributable risks in cohort studies: examples and software. Cancer Causes Control 2007; 18: 571–579.
Hildebrandt M, Bender R, Gehrmann U, Blettner M . Calculating confidence intervals for impact numbers. BMC Med Res Methodol 2006; 6: 32.
Ichtchenko K, Bittner MA, Krasnoperov V, Little AR, Chepurny O, Holz RW et al. A novel ubiquitously expressed alpha-latrotoxin receptor is a member of the CIRL family of G-protein-coupled receptors. J Biol Chem 1999; 274: 5491–5498.
Acknowledgements
We are grateful to the families who participated in this research. This research was supported by the Division of Intramural Research, NHGRI, NIH and in part by COLCIENCIAS, Grants 1115-04-12010, 11150418083, and by the Deutsche Forschungsgemeinschaft (KFO 125/1-1, SFB 581). This study used the high-performance computational capabilities of the SGI Origin 2000 system at the Center for Information Technology, National Institutes of Health, Bethesda, MD and the services of the NHGRI Genomics Core under the supervision of Marypat S Jones and Chandra Settara. Darryl Leja provided graphical assistance to the figures. N Steigerwald provided technical assistance in the DNA German sample processing. Mahim Jain, Sharon B Shively and Sabina Domené are doctoral students in the NIH Graduate Partnerships Program, at Oxford University, George Washington University and the University of Buenos Aires, respectively. Some of this work is to be presented to the above programs in partial fulfillment of the requirements for the PhD degree. Authors from Spain are grateful to Anna Bielsa, Xavier Gastaminza, Rosa Bosch, Monica Fernandez Anguiano and Sílvia Rodríguez-Ben for their participation in the clinical assessment and to Cristina Sánchez-Mora for laboratory assistance. MR is a recipient of a Miguel de Servet contract from the ‘Ministerio de Sanidad y Política Social’, Spain. Financial support was received from ‘Instituto de Salud Carlos III-FIS’ (PI041267, PI042010, PI040524, PI080519) and ‘Agència de Gestió d’Ajuts Universitaris i de Recerca-AGAUR’ (2009GR971). SNP genotyping of the Spanish samples was performed at the Barcelona node of the ‘Centro Nacional de Genotipado’ (CEGEN; www.cegen.org).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
None of the authors at the National Institutes of Health has any conflict of interest. Authors from other institutions contributed samples for replication studies only.
Additional information
Supplementary Information accompanies the paper on the Molecular Psychiatry website
Supplementary information
Rights and permissions
About this article
Cite this article
Arcos-Burgos, M., Jain, M., Acosta, M. et al. A common variant of the latrophilin 3 gene, LPHN3, confers susceptibility to ADHD and predicts effectiveness of stimulant medication. Mol Psychiatry 15, 1053–1066 (2010). https://doi.org/10.1038/mp.2010.6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/mp.2010.6
Keywords
This article is cited by
-
Brain exposure to SARS-CoV-2 virions perturbs synaptic homeostasis
Nature Microbiology (2024)
-
adgrl3.1-deficient zebrafish show noradrenaline-mediated externalizing behaviors, and altered expression of externalizing disorder-candidate genes, suggesting functional targets for treatment
Translational Psychiatry (2023)
-
Novel non-stimulants rescue hyperactive phenotype in an adgrl3.1 mutant zebrafish model of ADHD
Neuropsychopharmacology (2023)
-
Animal Models of Childhood Exposure to Lead or Manganese: Evidence for Impaired Attention, Impulse Control, and Affect Regulation and Assessment of Potential Therapies
Neurotherapeutics (2023)
-
Convergent selective signaling impairment exposes the pathogenicity of latrophilin-3 missense variants linked to inheritable ADHD susceptibility
Molecular Psychiatry (2022)