Abstract
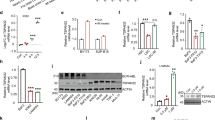
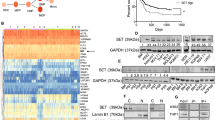
Protein phosphatase 2A (PP2A) is a human tumor suppressor that inhibits cellular transformation by regulating the activity of several signaling proteins critical for malignant cell behavior. PP2A has been described as a potential therapeutic target in chronic myeloid leukemia, Philadelphia chromosome-positive acute lymphoblastic leukemia and B-cell chronic lymphocytic leukemia. Here, we show that PP2A inactivation is a recurrent event in acute myeloid leukemia (AML), and that restoration of PP2A phosphatase activity by treatment with forskolin in AML cells blocks proliferation, induces caspase-dependent apoptosis and affects AKT and ERK1/2 activity. Moreover, treatment with forskolin had an additive effect with Idarubicin and Ara-c, drugs used in standard induction therapy in AML patients. Analysis at protein level of the PP2A activation status in a series of patients with AML at diagnosis showed PP2A hyperphosphorylation in 78% of cases (29/37). In addition, we found that either deregulated expression of the endogenous PP2A inhibitors SET or CIP2A, overexpression of SETBP1, or downregulation of some PP2A subunits, might be contributing to PP2A inhibition in AML. In conclusion, our results show that PP2A inhibition is a common event in AML cells and that PP2A activators, such as forskolin or FTY720, could represent potential novel therapeutic targets in AML.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Rowe JM . Optimal induction and post-remission therapy for AML in first remission. Hematology Am Soc Hematol Educ Program 2009; 1: 396–405.
Haferlach T . Molecular genetic pathways as therapeutic targets in acute myeloid leukemia. Hematology Am Soc Hematol Educ Program 2008; 1: 400–411.
Mumby M . PP2A: unveiling a reluctant tumor suppressor. Cell 2007; 130: 21–24.
Millward TA, Zolnierowicz S, Hemmings BA . Regulation of protein kinase cascades by protein phosphatase 2A. Trends Biochem Sci 1999; 24: 186–191.
Hemmings BA, Adams-Pearson C, Maurer F, Muller P, Goris J, Merlevede W et al. α- and β-Forms of the 65-kDa subunit of protein phosphatase 2A have a similar 39 amino acid repeating structure. Biochemistry 1990; 29: 3166–3173.
Zhou J, Pham HT, Ruediger R, Walter G . Characterization of the Aα and Aβ subunit isoforms of protein phosphatase 2A: differences in expression, subunit interaction, and evolution. Biochem J 2003; 369: 387–398.
Arino J, Woon CW, Brautigan DL, Miller Jr TB, Johnson GL . Human liver phosphatase 2A: cDNA and amino acid sequence of two catalytic subunit isotypes. Proc Natl Acad Sci USA 1988; 85: 4252–4256.
Cohen P . The structure and regulation of protein phosphatases. Annu Rev Biochem 1989; 58: 453–508.
Eichhorn PJ, Creyghton MP, Bernards R . Protein phosphatase 2A regulatory subunits and cancer. Biochim Biophys Acta 2009; 1795: 1–15.
Westermarck J, Hahn WC . Multiple pathways regulated by the tumor suppressor PP2A in transformation. Trends Mol Med 2008; 14: 152–160.
Ruediger R, Pham HT, Walter G . Disruption of protein phosphatase 2A subunit interaction in human cancers with mutations in the A alpha subunit gene. Oncogene 2001; 20: 10–15.
Ruediger R, Pham HT, Walter G . Alterations in protein phosphatase 2A subunit interaction in human carcinomas of the lung and colon with mutations in the A beta subunit gene. Oncogene 2001; 20: 1892–1899.
Wang SS, Esplin ED, Li JL, Huang L, Gazdar A, Minna J et al. Alterations of the PPP2R1B gene in human lung and colon cancer. Science 1998; 282: 284–287.
Calin GA, di Iasio MG, Caprini E, Vorechovsky I, Natali PG, Sozzi G et al. Low frequency of alterations of the alpha (PPP2R1A) and beta (PPP2R1B) isoforms of the subunit A of the serine-threonine phosphatase 2A in human neoplasms. Oncogene 2000; 19: 1191–1195.
Esplin ED, Ramos P, Martinez B, Tomlinson GE, Mumby MC, Evans GA . The glycine 90 to aspartate alteration in the Abeta subunit of PP2A (PPP2R1B) associates with breast cancer and causes a deficit in protein function. Genes Chromosomes Cancer 2006; 45: 182–190.
Takagi Y, Futamura M, Yamaguchi K, Aoki S, Takahashi T, Saji S . Alterations of the PPP2R1B gene located at 11q23 in human colorectal cancers. Gut 2000; 47: 268–271.
Tamaki M, Goi T, Hirono Y, Katayama K, Yamaguchi A . PPP2R1B gene alterations inhibit interaction of PP2A-Abeta and PP2A-C proteins in colorectal cancers. Oncol Rep 2004; 11: 655–659.
Kalla C, Scheuermann MO, Kube I, Schlotter M, Mertens D, Dohner H et al. Analysis of 11q22-q23 deletion target genes in B-cell chronic lymphocytic leukaemia: evidence for a pathogenic role of NPAT, CUL5, and PPP2R1B. Eur J Cancer 2007; 43: 1328–1335.
Hemmer S, Wasenius VM, Haglund C, Zhu Y, Knuutila S, Franssila K et al. Alterations in the suppressor gene PPP2R1B in parathyroid hyperplasias and adenomas. Cancer Genet Cytogenet 2002; 134: 13–17.
Chou HC, Chen CH, Lee HS, Lee CZ, Huang GT, Yang PM et al. Alterations of tumour suppressor gene PPP2R1B in hepatocellular carcinoma. Cancer Lett 2007; 253: 138–143.
Sablina AA, Chen W, Arroyo JD, Corral L, Hector M, Bulmer SE et al. The tumor suppressor PP2A Abeta regulates the RalA GTPase. Cell 2007; 129: 969–982.
Chen W, Arroyo JD, Timmons JC, Possemato R, Hahn WC . Cancer-associated PP2A Aalpha subunits induce functional haploinsufficiency and tumorigenicity. Cancer Res 2005; 65: 8183–8192.
Sablina AA, Hahn WC . The role of PP2A A subunits in tumor suppression. Cell Adh Migr 2007; 1: 140–141.
Arnold HK, Sears RC . Protein phosphatase 2A regulatory subunit B56alpha associates with c-myc and negatively regulates c-myc accumulation. Mol Cell Biol 2006; 26: 2832–2844.
Margolis SS, Perry JA, Forester CM, Nutt LK, Guo Y, Jardim MJ et al. Role for the PP2A/B56delta phosphatase in regulating 14-3-3 release from Cdc25 to control mitosis. Cell 2006; 127: 759–773.
Ma J, Arnold HK, Lilly MB, Sears RC, Kraft AS . Negative regulation of Pim-1 protein kinase levels by the B56beta subunit of PP2A. Oncogene 2007; 26: 5145–5153.
Letourneux C, Rocher G, Porteu F . B56-containing PP2A dephosphorylate ERK and their activity is controlled by the early gene IEX-1 and ERK. EMBO J 2006; 25: 727–738.
Chen W, Possemato R, Campbell KT, Plattner CA, Pallas DC, Hahn WC . Identification of specific PP2A complexes involved in human cell transformation. Cancer Cell 2004; 5: 127–136.
Shouse GP, Nobumori Y, Liu X . A B56gamma mutation in lung cancer disrupts the p53-dependent tumor-suppressor function of protein phosphatase 2A. Oncogene 2010; 29: 1669–1681.
Li HH, Cai X, Shouse GP, Piluso LG, Liu X . A specific PP2A regulatory subunit, B56gamma, mediates DNA damage-induced dephosphorylation of p53 at Thr55. EMBO J 2007; 26: 402–411.
Neviani P, Santhanam R, Trotta R, Notari M, Blaser BW, Liu S et al. The tumor suppressor PP2A is functionally inactivated in blast crisis CML through the inhibitory activity of the BCR/ABL-regulated SET protein. Cancer Cell 2005; 8: 355–368.
Neviani P, Santhanam R, Oaks JJ, Eiring AM, Notari M, Blaser BW et al. FTY720, a new alternative for treating blast crisis chronic myelogenous leukemia and Philadelphia chromosome-positive acute lymphocytic leukemia. J Clin Invest 2007; 117: 2408–2421.
Junttila MR, Puustinen P, Niemela M, Ahola R, Arnold H, Bottzauw T et al. CIP2A inhibits PP2A in human malignancies. Cell 2007; 130: 51–62.
Yokoyama N, Reich NC, Miller WT . Determinants for the interaction between Janus kinase 2 and protein phosphatase 2A. Arch Biochem Biophys 2003; 417: 87–95.
Gallay N, Dos Santos C, Cuzin L, Bousquet M, Simmonet Gouy V, Chaussade C et al. The level of AKT phosphorylation on threonine 308 but not on serine 473 is associated with high-risk cytogenetics and predicts poor overall survival in acute myeloid leukaemia. Leukemia 2009; 23: 1029–1038.
Roberts KG, Smith AM, McDougall F, Carpenter H, Horan M, Neviani P et al. Essential requirement for PP2A inhibition by the oncogenic receptor c-KIT suggests PP2A reactivation as a strategy to treat c-KIT+ cancers. Cancer Res 2010; 70: 5438–5447.
Cristobal I, Blanco FJ, Garcia-Orti L, Marcotegui N, Vicente C, Rifon J et al. SETBP1 overexpression is a novel leukemogenic mechanism that predicts adverse outcome in elderly patients with acute myeloid leukemia. Blood 2010; 115: 615–625.
Luo LY, Huang J, Gou BD, Zhang TL, Wang K . Induction of human promyelocytic leukemia HL-60 cell differentiation into monocytes by arsenic sulphide: involvement of serine/threonine protein phosphatases. Leuk Res 2006; 30: 1399–1405.
Martelli AM, Papa V, Tazzari PL, Ricci F, Evangelisti C, Chiarini F et al. Erucylphosphohomocholine, the first intravenously applicable alkylphosphocholine, is cytotoxic to acute myelogenous leukemia cells through JNK- and PP2A-dependent mechanisms. Leukemia 2010; 24: 687–698.
Livak KJ, Schmittgen TD . Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)) method. Methods 2001; 25: 402–408.
Irizarry RA, Bolstad BM, Collin F, Cope LM, Hobbs B, Speed TP . Summaries of affymetrix genechip probe level data. Nucleic Acids Res 2003; 31: e15.
Smyth GK . Linear models and empirical bayes methods for assessing differential expression in microarray experiments. Stat Appl Genet Mol Biol 2004; 3: Article 3.
Chen J, Martin BL, Brautigan DL . Regulation of protein serine-threonine phosphatase type-2A by tyrosine phosphorylation. Science 1992; 257: 1261–1264.
Saydam G, Aydin HH, Sahin F, Selvi N, Oktem G, Terzioglu E et al. Involvement of protein phosphatase 2A in interferon-alpha-2b-induced apoptosis in K562 human chronic myelogenous leukaemia cells. Leuk Res 2003; 27: 709–717.
Gilliland DG, Griffin JD . Role of FLT3 in leukemia. Curr Opin Hematol 2002; 9: 274–281.
Liu Q, Zhao X, Frissora F, Ma Y, Santhanam R, Jarjoura D et al. FTY720 demonstrates promising preclinical activity for chronic lymphocytic leukemia and lymphoblastic leukemia/lymphoma. Blood 2008; 111: 275–284.
Junttila MR, Westermarck J . Mechanisms of MYC stabilization in human malignancies. Cell Cycle 2008; 7: 592–596.
Kornblau SM, Singh N, Qiu Y, Chen W, Zhang N, Coombes KR . Highly phosphorylated FOXO3A is an adverse prognostic factor in acute myeloid leukemia. Clin Cancer Res 2010; 16: 1865–1874.
Samanta AK, Chakraborty SN, Wang Y, Kantarjian H, Sun X, Hood J et al. Jak2 inhibition deactivates Lyn kinase through the SET-PP2A-SHP1 pathway, causing apoptosis in drug-resistant cells from chronic myelogenous leukemia patients. Oncogene 2009; 28: 1669–1681.
Yokoyama N, Reich NC, Miller WT . Involvement of protein phosphatase 2A in the interleukin-3-stimulated Jak2-Stat5 signaling pathway. J Interferon Cytokine Res 2001; 21: 369–378.
Li M, Makkinje A, Damuni Z . The myeloid leukemia-associated protein SET is a potent inhibitor of protein phosphatase 2A. J Biol Chem 1996; 271: 11059–11062.
Cervoni N, Detich N, Seo SB, Chakravarti D, Szyf M . The oncoprotein Set/TAF-1beta, an inhibitor of histone acetyltransferase, inhibits active demethylation of DNA, integrating DNA methylation and transcriptional silencing. J Biol Chem 2002; 277: 25026–25031.
Rosati R, La Starza R, Barba G, Gorello P, Pierini V, Matteucci C et al. Cryptic chromosome 9q34 deletion generates TAF-Ialpha/CAN and TAF-Ibeta/CAN fusion transcripts in acute myeloid leukemia. Haematologica 2007; 92: 232–235.
Xu Y, Xing Y, Chen Y, Chao Y, Lin Z, Fan E et al. Structure of the protein phosphatase 2A holoenzyme. Cell 2006; 127: 1239–1251.
Cho US, Xu W . Crystal structure of a protein phosphatase 2A heterotrimeric holoenzyme. Nature 2007; 445: 53–57.
Acknowledgements
This work was supported by the Ministerio Educación y Ciencia (SAF2005/06425 and AP2006-03038), Ministerio Ciencia e Innovación (PI081687), Departamento Salud del Gobierno de Navarra (14/2008), ISCIII-RTICC (RD06/0020/0078) and Fundación para la Investigación Médica Aplicada y UTE (Spain). We thank Dr Anna Sablina for useful discussion, Elizabeth Guruceaga of the Unit of Proteomics, Genomics and Bioinformatics of CIMA for bioinformatics analysis, and Leyre Urquiza and Marisol Gonzalez-Huarriz for technical assistance.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies the paper on the Leukemia website
Supplementary information
Rights and permissions
About this article
Cite this article
Cristóbal, I., Garcia-Orti, L., Cirauqui, C. et al. PP2A impaired activity is a common event in acute myeloid leukemia and its activation by forskolin has a potent anti-leukemic effect. Leukemia 25, 606–614 (2011). https://doi.org/10.1038/leu.2010.294
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/leu.2010.294
Keywords
This article is cited by
-
Altering phosphorylation in cancer through PP2A modifiers
Cancer Cell International (2024)
-
Novel SETBP1 mutation in a chinese family with intellectual disability
BMC Medical Genomics (2023)
-
SET-PP2A complex as a new therapeutic target in KMT2A (MLL) rearranged AML
Oncogene (2023)
-
Prognostic value of CIP2A gene expression in adult Egyptian acute myeloid leukemia patients
Egyptian Journal of Medical Human Genetics (2020)
-
A new regulatory mechanism of protein phosphatase 2A activity via SET in acute myeloid leukemia
Blood Cancer Journal (2020)