Abstract
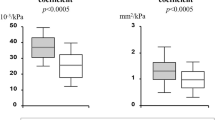
Hyperhomocysteinemia (HHcy) is an independent risk factor for coronary artery disease. Chronic HHcy induces coronary endothelial injury, and homocysteine (Hcy) inhibits insulin sensitivity by inducing endoplasmic reticulum stress in adipose tissue. However, the combined effects of chronic HHcy and hypertension on coronary artery endothelial function have still remained unclear. Fifty-one patients with essential hypertension (HT) were recruited, and were divided into nHT group (without HHcy) and H-type HT group (with HHcy). Forty healthy subjects were enrolled as controls. We assessed coronary artery endothelial function by coronary flow velocity reserve (CFVR). Hcy levels were significantly higher (15.60±6.58 vs 13.08±4.13 μmol l−1, P<0.05), whereas CFVR values were significantly lower in the HT patients than in the control group (2.88±0.70 vs 3.23±0.54, P<0.05). Hcy levels were negatively correlated with CFVR value (r=−0.578, P<0.01, 95% confidence interval −0.085 to −0.046). The homeostasis model assessment-insulin resistance values were significantly higher (3.92±2.16 vs 2.72±1.50, P<0.05) , whereas CFVR values were significantly lower in H-type HT group than in nHT group (2.31±0.31 vs 3.25±0.62, P<0.01). Coronary artery endothelial function may be impaired in essential hypertensive patients with HHcy, and insulin resistance induced by HHcy may contribute to this damage.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Clarke R, Daly L, Robinson K, Naughten E, Cahalane S, Fowler B et al. Hyperhomocysteinemia: an independent risk factor for vascular disease. New Engl J Med 1991; 324 (17): 1149–1155.
Veeranna V, Zalawadiya SK, Niraj A, Pradhan J, Ference B, Burack RC et al. Homocysteine and reclassification of cardiovascular disease risk. J Am Coll Cardiol 2011; 58 (10): 1025–1033.
Wang G, Dai J, Mao J, Zeng X, Yang X, Wang X . Folic acid reverses hyper-responsiveness of LPS-induced chemokine secretion from monocytes in patients with hyperhomocysteinemia. Atherosclerosis 2005; 179 (2): 395–402.
Wang G, Mao JM, Wang X, Zhang FC . Effect of homocysteine on plaque formation and oxidative stress in patients with acute coronary syndromes. Chin Med J (Engl) 2004; 117 (11): 1650–1654.
He L, Zeng H, Li F, Feng J, Liu S, Liu J et al. Homocysteine impairs coronary artery endothelial function by inhibiting tetrahydrobiopterin in patients with hyperhomocysteinemia. Am J Physiol Endocrinol Metab 2010; 299 (6): E1061–E1065.
Li Y, Zhang H, Jiang C, Xu M, Pang Y, Feng J et al. Hyperhomocysteinemia promotes insulin resistance by inducing endoplasmic reticulum stress in adipose tissue. J Biol Chem 2013; 288 (14): 9583–9592.
Shimbo D, Muntner P, Mann D, Viera AJ, Homma S, Polak JF et al. Endothelial dysfunction and the risk of hypertension: the multi-ethnic study of atherosclerosis. Hypertension 2010; 55 (5): 1210–1216.
Gupta V, Sachdeva S, Khan AS, Haque SF . Endothelial dysfunction and inflammation in different stages of essential hypertension. Saudi J Kidney Dis Transpl 2011; 22 (1): 97–103.
Schulz E, Gori T, Munzel T . Oxidative stress and endothelial dysfunction in hypertension. Hypertens Res 2011; 34 (6): 665–673.
Wang G, Woo CW, Sung FL, Siow YL, O K . Increased monocyte adhesion to aortic endothelium in rats with hyperhomocysteinemia: role of chemokine and adhesion molecules. Arterioscler Thromb Vasc Biol 2002; 22 (11): 1777–1783.
Poddar R, Sivasubramanian N, DiBello PM, Robinson K, Jacobsen DW . Homocysteine induces expression and secretion of monocyte chemoattractant protein-1 and interleukin-8 in human aortic endothelial cells: implications for vascular disease. Circulation 2001; 103 (22): 2717–2723.
Chobanian AV, Bakris GL, Black HR, Cushman WC, Green LA, Izzo JL et al. Seventh report of the Joint National Committee on Prevention, Detection, Evaluation, and Treatment of High Blood Pressure. Hypertension 2003; 42 (6): 1206–1252.
Matsumura Y, Hozumi T, Watanabe H, Fujimoto K, Sugioka K, Takemoto Y et al. Cut-off value of coronary flow velocity reserve by transthoracic Doppler echocardiography for diagnosis of significant left anterior descending artery stenosis in patients with coronary risk factors. Am J Cardiol 2003; 92 (12): 1389–1393.
Pizzuto F, Voci P, Mariano E, Puddu PE, Chiavari PA, Romeo F . Noninvasive coronary flow reserve assessed by transthoracic coronary Doppler ultrasound in patients with left anterior descending coronary artery stents. Am J Cardiol 2003; 91 (5): 522–526.
Yan TT, Li Q, Zhang XH, Wu WK, Sun J, Li L et al. Homocysteine impaired endothelial function through compromised vascular endothelial growth factor/Akt/endothelial nitric oxide synthase signalling. Clin Exp Pharmacol Physiol 2010; 37 (11): 1071–1077.
Dhillon B, Badiwala MV, Maitland A, Rao V, Li SH, Verma S . Tetrahydrobiopterin attenuates homocysteine induced endothelial dysfunction. Mol Cell Biochem 2003; 247 (1-2): 223–227.
Suematsu N, Ojaimi C, Kinugawa S, Wang Z, Xu X, Koller A et al. Hyperhomocysteinemia alters cardiac substrate metabolism by impairing nitric oxide bioavailability through oxidative stress. Circulation 2007; 115 (2): 255–262.
Wang D, Wang H, Luo P, Hwang A, Sun D, Wang Y et al. Effects of ghrelin on homocysteine-induced dysfunction and inflammatory response in rat cardiac microvascular endothelial cells. Cell Biol Int 2012; 36 (6): 511–517.
Edirimanne VE, Woo CW, Siow YL, Pierce GN, Xie JY, O K . Homocysteine stimulates NADPH oxidase-mediated superoxide production leading to endothelial dysfunction in rats. Can J Physiol Pharmacol 2007; 85 (12): 1236–1247.
Ji C, Kaplowitz N . Hyperhomocysteinemia, endoplasmic reticulum stress, and alcoholic liver injury. World J Gastroenterol 2004; 10 (12): 1699–1708.
Chiang JK, Sung ML, Yu HR, Chang HI, Kuo HC, Tsai TC et al. Homocysteine induces smooth muscle cell proliferation through differential regulation of cyclins A and D1 expression. J Cell Physiol 2011; 226 (4): 1017–1026.
Rajkumar V, Ragatzki P, Sima A, Levy J . Enhanced platelet aggregation, high homocysteine level, and microvascular disease in diabetic muscle infarctions: implications for therapy. Endocrine 1999; 11 (1): 57–60.
Vignini A, Nanetti L, Bacchetti T, Ferretti G, Curatola G, Mazzanti L . Modification induced by homocysteine and low-density lipoprotein on human aortic endothelial cells: an in vitro study. J Clin Endocrinol Metab 2004; 89 (9): 4558–4561.
Tousoulis D, Bouras G, Antoniades C, Marinou K, Papageorgiou N, Miliou A et al. Methionine-induced homocysteinemia impairs endothelial function in hypertensives: the role of asymmetrical dimethylarginine and antioxidant vitamins. Am J Hypertens 2011; 24 (8): 936–942.
Emoto M, Kanda H, Shoji T, Kawagishi T, Komatsu M, Mori K et al. Impact of insulin resistance and nephropathy on homocysteine in type 2 diabetes. Diabetes Care 2001; 24 (3): 533–538.
Golbahar J, Aminzadeh MA, Kassab SE, Omrani GR . Hyperhomocysteinemia induces insulin resistance in male Sprague-Dawley rats. Diabetes Res Clin Pract 2007; 76 (1): 1–5.
Buysschaert M, Dramais AS, Wallemacq PE, Hermans MP . Hyperhomocysteinemia in type 2 diabetes: relationship to macroangiopathy, nephropathy, and insulin resistance. Diabetes Care 2000; 23 (12): 1816–1822.
Li Y, Jiang C, Xu G, Wang N, Zhu Y, Tang C et al. Homocysteine upregulates resistin production from adipocytes in vivo and in vitro. Diabetes 2008; 57 (4): 817–827.
Najib S, Sanchez-Margalet V . Homocysteine thiolactone inhibits insulin signaling, and glutathione has a protective effect. J Mol Endocrinol 2001; 27 (1): 85–91.
Montagnani M, Chen H, Barr VA, Quon MJ . Insulin-stimulated activation of eNOS is independent of Ca2+ but requires phosphorylation by Akt at Ser(1179). J Biol Chem 2001; 276 (32): 30392–30398.
Taniguchi CM, Emanuelli B, Kahn CR . Critical nodes in signalling pathways: insights into insulin action. Nat Rev Mol Cell Biol 2006; 7 (2): 85–96.
Acknowledgements
This work was supported by grants from the Major National Basic Research Program of P.R. China (No. 2011CB503904) and the Chinese National Natural Science Foundation (No. 81270369, 81070244) to Guang Wang. We thank the NIH Fellows Editorial Board for modifying our manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Rights and permissions
About this article
Cite this article
Liu, J., Xu, Y., Zhang, H. et al. Coronary flow velocity reserve is impaired in hypertensive patients with hyperhomocysteinemia. J Hum Hypertens 28, 743–747 (2014). https://doi.org/10.1038/jhh.2014.22
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/jhh.2014.22
This article is cited by
-
H-type hypertension is a risk factor for chronic total coronary artery occlusion: a cross-sectional study from southwest China
BMC Cardiovascular Disorders (2023)
-
Homocysteine diminishes apolipoprotein A-I function and expression in patients with hypothyroidism: a cross-sectional study
Lipids in Health and Disease (2016)
-
Hyperhomocysteinemia is associated with decreased apolipoprotein AI levels in normal healthy people
BMC Cardiovascular Disorders (2016)
-
“Boomerang Neuropathology” of Late-Onset Alzheimer’s Disease is Shrouded in Harmful “BDDS”: Breathing, Diet, Drinking, and Sleep During Aging
Neurotoxicity Research (2015)