Abstract
Background/objectives:
Patterns of postabsorptive adipose tissue fatty acid storage correlate with sex-specific body fat distribution. Some proteins and enzymes participating in this pathway include CD36 (facilitated transport), acyl-CoA synthetase (ACS; the first step in fat metabolism) and diacylglycerol acetyltransferase (DGAT; the final step of triglyceride synthesis). Our aim was to better define CD36, ACS and DGAT in relation to sex, subcutaneous fat depots and adipocyte size.
Subjects/methods:
Data were collected from studies conducted at Mayo Clinic between 2004 and 2012. Abdominal and femoral subcutaneous fat biopsy samples must have been collected in the postabsorptive state from healthy males and premenopausal females. Body composition was measured with dual-energy X-ray absorptiometry and abdominal computerized tomography scans. Adipocyte size (microscopy), CD36 protein content (enzyme-linked immunosorbent assay) and ACS and DGAT enzyme activities were measured. Data are presented as medians and 25th, 75th quartiles.
Results:
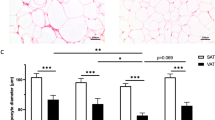
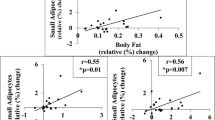
Males (n=60) and females (n=78) did not differ by age (37; 28, 46 years), body mass index (28.4; 24.6, 32.1 kg m−2) or abdominal (0.60; 0.45, 0.83 μg lipid per cell) and femoral adipocyte size (0.76; 0.60, 0.94 μg lipid per cell). Femoral ACS and DGAT were greater in females than males when expressed per mg lipid (ACS: 73 vs 55 pmol/mg lipid/min; DGAT: 5.5 vs 4.0 pmol/mg lipid/min; P<0.0001 for both) and per 1000 adipocytes (ACS: 59 vs 39 pmol per min per 1000 adipocytes; DGAT: 4.3 vs 3.1 pmol per min per 1000 adipocytes; P⩽0.0003 for both). There were no differences in abdominal fat storage factors between sexes. ACS and DGAT decreased as a function of adipocyte size (P<0.0001 for both). The decrease in ACS was greater in males and abdominal subcutaneous fat. There were no sex differences in CD36 in either fat depot, nor did it vary across adipocyte size.
Conclusions:
Facilitated transport of fatty acids by CD36 under postabsorptive conditions would not be different in those with large vs small adipocytes in either depot of both sexes. However, intracellular trafficking of fatty acids to triglyceride storage by ACS and DGAT may be less efficient in larger adipocytes.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Shadid S, Koutsari C, Jensen MD . Direct free fatty acid uptake into human adipocytes in vivo: relation to body fat distribution. Diabetes 2007; 56: 1369–1375.
Koutsari C, Ali AH, Mundi MS, Jensen MD . Storage of circulating FFA in adipose tissue of postabsorptive humans: quantitative measures and implications for body fat distribution. Diabetes 2011; 60: 2032–2040.
Sondergaard E, Gormsen LC, Nellemann B, Jensen MD, Nielsen S . Body composition determines direct FFA storage pattern in overweight women. Am J Physiol 2012; 302: E1599–E1604.
Bucci M, Borra R, Någren K, Maggio R, Tuunanen H, Oikonen V et al. Human obesity is characterized by defective fat storage and enhanced muscle fatty acid oxidation, and trimetazidine gradually counteracts these abnormalities. Am J Physiol 2011; 301: E105–E112.
Hannukainen JC, Kalliokoski KK, Borra RJ, Viljanen AP, Janatuinen T, Kujala UM et al. Higher free fatty acid uptake in visceral than in abdominal subcutaneous fat tissue in men. Obesity (Silver Spring, MD) 2010; 18: 261–265.
Bjorntorp P, Enzi G, Ohlson R, Persson B, Sponbergs P, Smith U . Lipoprotein lipase activity and uptake of exogenous triglycerides in fat cells of different size. Horm Metab Res 1975; 7: 230–237.
Koutsari C, Mundi MS, Ali AH, Jensen MD . Storage rates of circulating free fatty acid into adipose tissue during eating or walking in humans. Diabetes 2012; 61: 329–338.
Hames KC, Vella A, Kemp BJ, Jensen MD . Free fatty acid uptake in humans with CD36 deficiency. Diabetes 2014; 63: 3606–3614.
Tchoukalova Y, Votruba SB, Tchkonia T, Giorgadze N, Kirkland JL, Jensen MD . Regional differences in cellular mechanisms of adipose tissue gain with overfeeding. Proc Natl Acad Sci USA 2010; 107: 18226–18231.
Drolet R, Richard C, Sniderman AD, Mailloux J, Fortier M, Huot C et al. Hypertrophy and hyperplasia of abdominal adipose tissues in women. Int J Obes (Lond) 2008; 32: 283–291.
Jensen MD, Kanaley JA, Reed JE, Sheedy PF . Measurement of abdominal and visceral fat with computed tomography and dual-energy x-ray absorptiometry. Am J Clin Nutr 1995; 61: 274–278.
Tchoukalova YD, Harteneck DA, Karwoski RA, Tarara J, Jensen MD . A quick, reliable, and automated method for fat cell sizing. J Lipid Res 2003; 44: 1795–1801.
Allred CC, Krennmayr T, Koutsari C, Zhou L, Ali AH, Jensen MD . A novel ELISA for measuring CD36 protein in human adipose tissue. J Lipid Res 2011; 52: 408–415.
Hall AM, Smith AJ, Bernlohr DA . Characterization of the acyl CoA synthetase activity of purified murine fatty acid transport protein 1. J Biol Chem 2003; 278: 43008–43013.
Hou XG, Moser S, Sarr MG, Thompson GB, Que FG, Jensen MD . Visceral and subcutaneous adipose tissue diacylglycerol acyltransferase activity in humans. Obesity 2009; 17: 1129–1134.
Santosa S, Jensen MD . Effects of male hypogonadism on regional adipose tissue fatty acid storage and lipogenic proteins. PLoS One 2012; 7: e31473.
Santosa S, Jensen MD . Adipocyte fatty acid storage factors enhance subcutaneous fat storage in postmenopausal women. Diabetes 2013; 62: 775–782.
Febbraio M, Abumrad NA, Hajjar DP, Sharma K, Cheng W, Pearce SF et al. A null mutation in murine CD36 reveals an important role in fatty acid and lipoprotein metabolism. J Biol Chem 1999; 274: 19055–19062.
Bonen A, Tandon NN, Glatz JFC, JJFP Luiken, Heigenhauser GJF . The fatty acid transporter FAT/CD36 is upregulated in subcutaneous and visceral adipose tissues in human obesity and type 2 diabetes. Int J Obes 2006; 30: 877–883.
Roberts R, Hodson L, Dennis AL, Neville MJ, Humphreys SM, Harnden KE et al. Markers of de novo lipogenesis in adipose tissue: associations with small adipocytes and insulin sensitivity in humans. Diabetologia 2009; 52: 882–890.
Tchoukalova YD, Koutsari C, Karpyak MV, Votruba SB, Wendland E, Jensen MD . Subcutaneous adipocyte size and body fat distribution. Am J Clin Nutr 2008; 87: 56–63.
Sparks LM, Pasarica M, Sereda O, deJonge L, Thomas S, Loggins H et al. Effect of adipose tissue on the sexual dimorphism in metabolic flexibility. Metabolism 2009; 58: 1564–1571.
Vogel C, Marcotte EM . Insights into the regulation of protein abundance from proteomic and transcriptomic analyses. Nat Rev Genet 2012; 13: 227–232.
Blaak E . Gender differences in fat metabolism. Curr Opin Clin Nutr Metab Care 2001; 4: 499–502.
Abate N, Garg A . Heterogeneity in adipose tissue metabolism: causes, implications and management of regional adiposity. Progr Lipid Res 1995; 34: 53–70.
Acknowledgements
We are grateful to the participants for playing an integral role in making this research possible. In addition, thanks goes to the Mayo Clinic CRU staff, and especially Barbara Norby, Carley Vrieze, Christy Allred, Lendia Zhou, Debra Harteneck and Darlene Lucas (all from Mayo Clinic), for their technical assistance and help with data collection. This work was supported by Grant NCRR UL1 TR000135, and National Institutes of Health Grants DK-45343, DK-40484 and DK-50456.
Author Contributions
KCH and MDJ designed the research. KCH, CK, SS and NCB performed the research. KCH and MDJ analyzed the data and wrote the paper. Dr Michael Jensen is the guarantor of this work and, as such, had full access to all of the data in the study and takes responsibility for the integrity of the data and the accuracy of the data analysis.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Rights and permissions
About this article
Cite this article
Hames, K., Koutsari, C., Santosa, S. et al. Adipose tissue fatty acid storage factors: effects of depot, sex and fat cell size. Int J Obes 39, 884–887 (2015). https://doi.org/10.1038/ijo.2015.10
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ijo.2015.10
This article is cited by
-
Peripancreatic adipose tissue protects against high-fat-diet-induced hepatic steatosis and insulin resistance in mice
International Journal of Obesity (2020)
-
Association of calf circumference with insulin resistance and non-alcohol fatty liver disease: the REACTION study
BMC Endocrine Disorders (2017)
-
Adipose tissue plasticity: how fat depots respond differently to pathophysiological cues
Diabetologia (2016)