Abstract
The renin–angiotensin–aldosterone system (RAAS) is a pivotal regulator of physiological homeostasis and diseases of the cardiovascular system. Recently, new factors have been discovered, such as angiotensin-converting enzyme 2 (ACE2), angiotensin-(1–7) and Mas. This newly defined ACE2–angiotensin-(1–7)–Mas axis was shown to have a critical role in the vasculature and in the heart, exerting mainly protective effects. One important mechanism of the classic and the new RAAS regulate vascular function is through the regulation of redox signaling. Angiotensin II is a classic prooxidant peptide that increases superoxide production through the activation of NAD(P)H oxidases. This review summarizes the current knowledge about the ACE2–angiotensin-(1–7)–Mas axis and redox signaling in the context of cardiovascular regulation and disease. By interacting with its receptor Mas, angiotensin-(1–7) induces the release of nitric oxide from endothelial cells and thereby counteracts the effects of angiotensin II. ACE2 converts angiotensin II to angiotensin-(1–7) and, thus, is a pivotal regulator of the local effects of the RAAS on the vessel wall. Taken together, the ACE2–angiotensin-(1–7)–Mas axis emerges as a novel therapeutic target in the context of cardiovascular and metabolic diseases.
Similar content being viewed by others
The classic pathway and the novel components of the renin–angiotensin–aldosterone system (RAAS)
In recent decades, cardiovascular disease has been considered the main cause of morbidity and mortality worldwide. Hypertension is a critical risk factor for these diseases, which include coronary and peripheral arterial disease, stroke and heart failure.1 One of the major regulatory mechanisms of cardiovascular homeostasis is the RAAS.2, 3 The classic pathway involves a two-step enzymatic pathway (Figure 1). First, the aspartyl protease renin, which is primarily released by the kidneys, cleaves a hepatic protein, angiotensinogen, to angiotensin I (Ang I).2, 3 The second step involves hydrolysis of Ang I by angiotensin-converting enzyme (ACE), resulting in the production of the bioactive octapeptide angiotensin II (Ang II), which is a potent vasoconstrictor and stimulates the release of aldosterone from the adrenal cortex.2, 3, 4, 5 Moreover, ACE inactivates the vasodilator bradykinin by degradation of the peptide.2
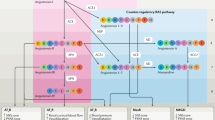
Classic pathway and new components of the RAAS. Three main enzymes are involved in the generation of active angiotensin peptides: First, renin cleaves angiotensinogen into angiotensin I (Ang I). The second step involves hydrolysis of Ang I by angiotensin-converting enzyme (ACE), resulting in the production of the bioactive octapeptide angiotensin II (Ang II), which interacts with angiotensin type I (AT1-R) and angiotensin type II (AT2-R) receptors. Third, ACE2 catalyzes the conversion of Ang II to Ang-(1–7), which mediates its effects by interaction with the G-protein-coupled receptor Mas.
The discovery of angiotensin-(1–7) (Ang-(1–7)) by Santos et al.6 and the subsequent cloning of angiotensin-converting enzyme 2 (ACE2)7, 8 shed new light on angiotensin metabolism and the regulation of the RAAS. ACE2, a zinc metalloprotease with carboxypeptidase activity, catalyzes the conversion of Ang I to the non-apeptide Ang-(1–9) or the conversion of Ang II to Ang-(1–7) by the removal of a single carboxy-terminal amino acid (phenylalanine; Figure 1).8 This heptapeptide can also be formed from Ang I by the action of neprilysin (also known as neutral endopeptidase 24.11).9 It has been suggested that Ang-(1–7) mediates its effects by interacting with the G-protein-coupled receptor Mas,10 a prototypic seven-transmembrane domain receptor (Figure 1), which is predominantly expressed in the brain and testis11 but is also found in the kidney, heart and vessels.11, 12, 13 Moreover, several studies have shown that the interaction of Ang-(1–7) with Mas evokes numerous protective cardiovascular actions, such as nitric oxide (•NO)14, 15 release, Akt phosphorylation16 and vasodilation (Figure 2).17 Nevertheless, other studies indicate that Ang-(1–7) may function through angiotensin type 2 receptor18 and that Mas can antagonize the actions of the angiotensin type 1 receptor.19, 20
Role of ACE2–Ang-(1–7)–Mas axis in vascular function and redox signaling in vessels. Under normal physiological conditions (a), •NO exerts pleiotropic effects in the regulation of vascular function. The balance between levels of •O2− and released •NO has a critical role in the maintenance of normal endothelial function. However, in pathological conditions (b), excessive •O2−, mainly produced by NAD(P)H oxidase, stimulates vasoconstriction and inflammation, resulting in endothelial dysfunction, mainly through the reduction of •NO bioavailability and the imbalance between ROS and antioxidant capacity. Thus, the classic RAAS is a potent prooxidant system in vessels, causing endothelial dysfunction. The ACE2–Ang-(1–7)–Mas axis counteracts these effects. For more details, see text. AT1-R, angiotensin type I receptor; Cat, catalase; COX, cyclooxygenase; GPX, glutathione peroxidase; Ec-SOD; extracellular-superoxide dismutase; LPO, lipid peroxidation; MPO, myeloperoxidase; XO, xanthine oxidase.
The local activity of ACE2 determines the relative levels of the vasoconstrictor and pro-oxidative peptide Ang II and its vasodilatory and antioxidative metabolite Ang-(1–7) at the corresponding receptors (Figure 2).21, 22 There is now a very large body of evidence showing that the newly discovered angiotensin system, ACE2–Ang-(1–7)–Mas, is pivotal for physiological homeostasis.23
Endothelial dysfunction and oxidative stress in the etiology of cardiovascular disease
Strategically located between the circulating blood and the other vascular layers, the endothelium is a sensor of hemodynamic changes and is known to have a central role in vascular homeostasis. However, for a long time, this layer was seen ‘only’ as a cluster of cells that separates the circulating blood from the other layers. Exactly 30 years ago, a seminal paper revised this foundational idea. The seminal experiments of Furchgott and Zawadzki24 first demonstrated the existence of endothelium-derived relaxing factor, which was subsequently identified as nitric oxide (•NO; nitrogen monoxide).25 In the endothelium, this free radical is produced from L-arginine by endothelial nitric oxide synthase (eNOS) in the presence of cofactors, mainly tetrahydrobiopterin (BH4) (Figure 2).26, 27, 28, 29 Interestingly, Landmesser et al.,30 in an elegant study, showed that oxidation of this biopterin induces eNOS uncoupling. In this structural state, the enzyme is an important source of reactive oxygen species (ROS).31, 32 Thus, in the absence of sufficient levels of cofactors, such as BH4 for enzymatic catalysis, eNOS may reduce molecular oxygen rather than transfer electrons to the substrate L-arginine, which results in the generation of superoxide anion (•O2−).30, 31, 32 Interestingly, BH4 is believed to be deficient in conditions associated with altered endothelial function,30 such as hypercholesterolemia,33 diabetes,34 high blood pressure35 and cigarette smoking.36, 37
Both •NO and •O2− are radicals. These molecules react rapidly with each other to generate peroxynitrite, •ONOO−38 (Figure 2), which is an important lipid peroxidation mediator. Peroxynitrite oxidizes low-density lipoprotein, a pivotal event for atherogenesis.39 However, it is well established that •NO exerts pleiotropic effects in the regulation of vascular function. Under normal physiological conditions, this molecule serves as a vasodilator and platelet inhibitor, exhibiting antiproliferative,40 antithrombotic,41 antiatherogenic42 and antioxidant capacities.43 Thus, the balance between levels of •O2− and released •NO has a critical role in the maintenance of normal endothelial function.39, 43, 44
In physiological conditions, a certain amount of intracellular •O2− is required for normal redox homeostasis in the vessel wall.45, 46, 47 Therefore, this reactive species is an important scavenger of the free radical signaling performed by •NO.48 However, in pathological conditions, the extracellular increase in •O2− decreases the bioavailability of •NO, reducing its diffusion into the vascular smooth muscle.49 Indeed, in the endothelium, excessive •O2− stimulates vasoconstriction and inflammation,46 which mutually reinforce each other, resulting in endothelial dysfunction, mainly through the reduction of •NO bioavailability and the imbalance between ROS and antioxidant capacity.48 Therefore, a vicious cycle pivotal in numerous disease processes is established.
According to Dröge,45 the term redox signaling is used to describe a regulatory process in which the signal is delivered through reduction–oxidation (redox) chemistry. The cellular concentration of ROS is determined by the balance between producing sources and the rate of clearance by antioxidant compounds and enzymes. Direct ROS scavenging antioxidant enzymes include superoxide dismutase, glutathione peroxidase and catalase.44, 45, 46, 47 Superoxide dismutase represents the first defense against •O2− and converts superoxide radicals to hydrogen peroxide (H2O2); catalase and glutathione peroxidase, are two different but kinetically complementary enzymes that can eliminate H2O2 by breaking it down directly to H2O and O2 (Figure 2).45, 46 On the other hand, oxidative stress emerges when the production of ROS, most notably •O2−, exceeds the quenching antioxidant capacity of the protective systems of the cell.44, 45, 46, 47
Recent evidence suggests that oxidative stress, which is elevated in cardiovascular disease, contributes to endothelial dysfunction. This disorder is a common feature of hypertension and results from the imbalance in the release of endothelium-derived relaxing factors, mainly •NO32, 44, 45, 46 and endothelium-derived contracting factors, such as cyclooxygenase-derived constrictors, endothelins and •O2−48, 49, 50 (Figure 2). Indeed, clinical studies show that ROS has a significant role in several cardiovascular and metabolic diseases.27, 51, 52, 53 On the other hand, impaired endothelium-mediated vasodilation in hypertension has been linked to decreased •NO bioavailability. This may be secondary to decreased synthesis or to increased •NO degradation because of its interaction of •NO with •O2− to form •ONOO−.38 Thus, the status of the redox system and •NO bioavailability are key factors controlling the influence of oxidative stress on cardiovascular function.
Accumulating evidence suggests that NAD(P)H oxidase is a major source of ROS, most notably •O2−, in both endothelial and vascular smooth muscle cells.54, 55, 56, 57, 58, 59, 60 Ang II promotes ROS production by the activation of membrane-bound NAD(P)H oxidase.54, 57, 59 This activation was first demonstrated by Griendling et al.59 in vascular smooth muscle cells. The H2O2 generated from the •O2− produced by NAD(P)H oxidase is involved in vasoconstriction and vascular hypertrophy57 (Figure 2). Thus, the classic RAAS is a potent prooxidant system in vessels causing endothelial dysfunction and consequently cardiovascular disease.
Redox-sensitive signaling by the ACE2–Ang-(1–7)–Mas axis
In 1992, Santos et al.61 demonstrated for the first time that Ang-(1–7) is present in vessels. After this first report, vasodilatory actions of the heptapeptide have been demonstrated in animals in several vascular beds.21, 22, 62 In addition, numerous studies showed that Ang-(1–7) functions mostly as an antithrombotic,63, 64 antiproliferative65, 66 and antioxidative agent.67 The mechanisms of action in the vasculature has not yet been well established. However, several studies have focused on control of the •NO and •O2− balance, which regulates cardiovascular function.
We have recently demonstrated a reduction in superoxide dismutase and catalase activity in Mas-deficient mice from two different genetic backgrounds, demonstrating impaired antioxidant properties in these animals.67 In addition, TBARS levels, which are most widely used as lipid peroxidation markers, were increased in aorta of Mas-null mice on the FVB/N genetic background. Accordingly, isoprostanes, other sensitive and stable oxidative stress biomarkers, are significantly upregulated in the urine of this strain. Moreover, ROS levels are increased in both FVB/N- and C57Bl/6 Mas-deficient animals,67, 68 but this difference is more pronounced in mice on the FVB/N background. Consequently, these Mas-knockout animals exhibit impaired in vivo endothelial function and increased blood pressure. One potential mechanism accounting for these observations is a decrease in •NO bioavailability. To find out if more •O2− is produced by NAD(P)H oxidases, the expression level of the main catalytic subunit of this enzyme, gp91phox (NOX2), was quantified and found to be increased in Mas-null mice. Furthermore, tempol, a stable superoxide dismutase mimetic, reduced blood pressure in Mas−/− mice to levels comparable with those observed in wild-type animals.67 Taken together, these data suggested a causative link between ROS/•NO imbalance and endothelial dysfunction in Mas-deficient mice.
To confirm these findings, our group developed a transgenic model overexpressing human ACE2 in vascular smooth muscle cells, using spontaneously hypertensive stroke-prone rats as a background strain.69 These transgenic rats showed improved endothelial function and decreased vasoconstriction response to Ang II. One potential mechanism underlying these actions involves a reduction in oxidative stress by a decrease in Ang II and/or an increase in Ang-(1–7). Nevertheless, additional studies are needed to clarify this issue.
Benter et al.70 showed that streptozotocin-treated spontaneously hypertensive rats (diabetic spontaneously hypertensive rats) chronically treated with Ang-(1–7) showed improved endothelial dysfunction of the renal artery, mediated primarily by the reduction of NAD(P)H-mediated oxidative stress, which resulted in improved renal function. In contrast, another study suggests that Ang-(1–7) stimulates oxidative stress in rat kidney.71 Thus, the role of the ACE2–Ang-(1–7)–Mas axis and oxidative stress in kidney pathophysiology is still controversial.
It has been demonstrated that the vascular actions of the ACE2–Ang-(1–7)–Mas axis could be mediated by different pathways, such as antagonism of angiotensin type 1 receptor,19, 20 by vasoactive peptides and increases in the release and/or bioavailability of •NO. Indeed, a bradykinin-potentiating effect of Ang-(1–7) was reported in certain rodent models.72, 73 This peptide evokes endothelium-dependent relaxation in several vascular beds.74, 75, 76 In addition, Ang-(1–7) improves endothelial function in vivo,77 induces coronary vasodilation by •NO release72, 78 and activates eNOS in Chinese hamster ovary cells.16 Carvalho et al.79 showed that short-term infusions of AVE 0991, a Mas agonist, increase the hypotensive effect of bradykinin in normotensive rats; one plausible mechanism for this effect involves an increase in •NO levels.
In atherosclerosis, ACE2 protects endothelial cells from Ang II-mediated macrophage infiltration and oxidative stress in an Ang-(1–7)-dependent manner.80, 81 Corroborating these findings, Tesanovic et al.82 recently reported that long-term Ang-(1–7) treatment induces atheroprotective effects in ApoE−/− mice, the most widely used animal model for atherosclerosis. According to the authors, the improvement in endothelial function resulted from an increase in •NO bioavailability, as the heptapeptide increased NOS expression and decreased •O2− production. Concurrently, it was recently shown that genetic deletion of ACE2 significantly increases83 plaque formation in atherosclerotic animals, which is decreased by targeted vascular ACE2 overexpression.80, 84 Consequently, these actions of the ACE2–Ang-(1–7)–Mas axis may be exploited in the treatment of atherosclerosis and other vascular diseases.
As mentioned above, the activation of NAD(P)H oxidase has a key role in the intracellular signaling that leads to hypertension-induced oxidative stress and endothelial dysfunction.85, 86, 87, 88 Sampaio et al.89 showed that the activation of this enzymatic complex by Ang II is attenuated by Ang-(1–7) in human endothelial cells. The authors suggest that the principal mechanism involves the interaction of this peptide with Mas, leading to the activation of AKT. This kinase mediates the dual phosphorylation of the Ser1177/Thr495 sites in eNOS and thereby stimulates eNOS activity and increases •NO release.
Current findings also suggest that the ACE2–Ang-(1–7)–Mas axis may participate in pathomechanisms of cerebrovascular disorders. Increased levels of ACE2 and Ang-(1–7) were found in the cerebrospinal fluid of multiple sclerosis patients.90 Moreover, Lu et al.91 showed that chronic central administration of Ang-(1–7) reduces the infarct area and provides cerebroprotection following focal cerebral ischemia/reperfusion in rats. The authors suggest that the heptapeptide modulates in vivo •NO release and eNOS expression in this stroke model. Corroborating these findings, another group showed in transgenic mice that the overexpression of ACE2 was associated with significant upregulation of neuronal NOS and eNOS in the brainstem.92 In addition, ACE2 overexpression in the brain prevented the Ang II-mediated decrease in NOS expression in regions involved in blood pressure regulation.93 Zhang et al.94 reported that the central administration of Ang-(1–7) stimulates •NO release and upregulates eNOS in a rat model of cerebral ischemia/reperfusion, further corroborating the antioxidant actions of ACE2 in the brain.
On the basis of above findings, there are at least three potential mechanisms by which ACE2 may exert protective cardiovascular effects via the reduction and/or prevention of oxidative stress: first, Ang II is cleaved to Ang-(1–7) by ACE2, thereby attenuating Ang II-induced actions; second, Ang-(1–7) may reduce the effects of Ang II by downregulating the signaling of angiotensin type 1 receptor; and third, the production of •NO is potently induced by Ang-(1–7), which might protect against endothelial dysfunction and oxidative stress (Figure 2).3, 84, 85, 86, 87, 88, 89
Conclusions
In summary, Ang-(1–7) is generated in the vascular wall and other tissues from Ang II by ACE2, resulting in a decrease in Ang II levels and an increase in Ang-(1–7) concentrations in the vessels. Thus, the ACE2–Ang-(1–7)–Mas axis counterregulates the classic RAAS in tissues responsible for blood pressure regulation and cardiovascular homeostasis. It decreases the production of ROS and increases endothelial •NO synthesis, improving the antioxidant capacity of cardiovascular tissues. This leads to an improvement of endothelial function and, consequently, to a decrease in arterial stiffness and, therefore, beneficially alters overall arterial function.
The net cardiovascular effect of angiotensin peptides in the vascular wall depends on the relative expression of classic and novel components of the RAAS in vascular layers, as well as their effects on the redox balance. As the ACE2–Ang-(1–7)–Mas axis exerts primarily beneficial effects in the vasculature, it may become an important therapeutic target for cardiovascular protection.
References
Yusuf S, Hawken S, Ounpuu S, Dans T, Avezum A, Lanas F, McQueen M, Budaj A, Pais P, Varigos J, Lisheng L, INTERHEART Study Investigators. Effect of potentially modifiable risk factors associated with myocardial infarction in 52 countries (the INTERHEART study): case-control study. Lancet 2004; 364: 937–952.
Bader M, Ganten D . Update on the tissue renin-angiotensin system. J Mol Med 2008; 86: 615–621.
Bader M . Tissue renin-angiotensin-aldosterone systems: targets for pharmacological therapy. Annu Rev Pharmacol Toxicol 2010; 50: 439–465.
Carey RM . Aldosterone and cardiovascular disease. Curr Opin Endocrinol Diabetes Obes 2010; 17: 194–198.
Briet M, Schiffrin EL . Aldosterone: effects on the kidney and cardiovascular system. Nat Rev Nephrol 2010; 6: 261–273.
Santos RA, Brosnihan KB, Chappell MC, Pesquero J, Chernicky CL, Greene LJ, Ferrario CM . Converting enzyme activity and angiotensin metabolism in the dog brainstem. Hypertension 1988; 11 (2 Part 2): I153–I157.
Tipnis SR, Hooper NM, Hyde R, Karran E, Christie G, Turner AJ . A human homolog of angiotensin-converting enzyme. Cloning and functional expression as a captopril-insensitive carboxypeptidase. J Biol Chem 2000; 275: 33238–33243.
Donoghue M, Hsieh F, Baronas E, Godbout K, Gosselin M, Stagliano N, Donovan M, Woolf B, Robison K, Jeyaseelan R, Breitbart RE, Acton S . A novel angiotensin-converting enzyme-related carboxypeptidase (ACE2) converts angiotensin I to angiotensin 1–9. Circ Res 2000; 87: E1–E9.
Welches WR, Brosnihan KB, Ferrario CM . A comparison of the properties and enzymatic activities of three angiotensin processing enzymes: angiotensin converting enzyme, prolyl endopeptidase and neutral endopeptidase 24.11. Life Sci 1993; 52: 1461–1480.
Santos RA, Simoes e Silva AC, Maric C, Silva DMR, Machado RP, de Buhr I, Heringer-Walther S, Pinheiro SV, Lopes MT, Bader M, Mendes EP, Lemos VS, Campagnole-Santos MJ, Schultheiss HP, Speth R, Walther T . Angiotensin-(1-7) is an endogenous ligand for the G protein-coupled receptor Mas. Proc Natl Acad Sci USA 2003; 100: 8258–8263.
Metzger R, Bader M, Ludwig T, Berberich C, Bunnemann B, Ganten D . Expression of the mouse and rat mas proto-oncogene in the brain and peripheral tissues. FEBS Lett 1995; 357: 27–32.
Pinheiro SV, Ferreira AJ, Kitten GT, da Silveira KD, da Silva DA, Santos SH, Gava E, Castro CH, Magalhaes JA, da Mota RK, Botelho-Santos GA, Bader M, Alenina N, Santos RA, Simoes e Silva AC . Genetic deletion of the angiotensin-(1-7) receptor Mas leads to glomerular hyperfiltration and microalbuminuria. Kidney Int 2009; 75: 1184–1193.
Alenina N, Xu P, Rentzsch B, Patkin EL, Bader M . Genetically altered animal models for Mas and angiotensin-(1-7). Exp Physiol 2008; 93: 528–537.
Nakamoto H, Ferrario CM, Fuller SB, Robaczewski DL, Winicov E, Dean RH . Angiotensin-(1-7) and nitric oxide interaction in renovascular hypertension. Hypertension 1995; 25 (4 Part 2): 796–802.
da Costa Gonçalves AC, Leite R, Fraga-Silva RA, Pinheiro SV, Reis AB, Reis FM, Touyz RM, Webb RC, Alenina N, Bader M, Santos RA . Evidence that the vasodilator angiotensin-(1-7)-Mas axis plays an important role in erectile function. Am J Physiol Heart Circ Physiol 2007; 293: H2588–H2596.
Sampaio WO, Souza dos Santos RA, Faria-Silva R, da Mata Machado LT, Schiffrin EL, Touyz RM . Angiotensin-(1-7) through receptor Mas mediates endothelial nitric oxide synthase activation via Akt-dependent pathways. Hypertension 2007; 49: 185–192.
Silva DM, Vianna HR, Cortes SF, Campagnole-Santos MJ, Santos RA, Lemos VS . Evidence for a new angiotensin-(1-7) receptor subtype in the aorta of Sprague-Dawley rats. Peptides 2007; 28: 702–707.
Lara LS, Cavalcante F, Axelband F, De Souza AM, Lopes AG, Caruso-Neves C . Involvement of the Gi/o/cGMP/PKG pathway in the AT2-mediated inhibition of outer cortex proximal tubule Na+-ATPase by Ang-(1-7). Biochem J 2006; 395: 183–190.
Santos EL, Reis RI, Silva RG, Shimuta SI, Pecher C, Bascands JL, Schanstra JP, Oliveira L, Bader M, Paiva AC, Costa-Neto CM, Pesquero JB . Functional rescue of a defective angiotensin II AT1 receptor mutant by the Mas protooncogene. Regul Pept 2007; 141: 159–167.
Kostenis E, Milligan G, Christopoulos A, Sanchez-Ferrer CF, Heringer-Walther S, Sexton PM, Gembardt F, Kellett E, Martini L, Vanderheyden P, Schultheiss HP, Walther T . G protein-coupled receptor Mas is a physiological antagonist of the angiotensin II type 1 receptor. Circulation 2005; 111: 1806–1813.
Crackower MA, Sarao R, Oudit GY, Yagil C, Kozieradzki I, Scanga SE, Oliveira-dos-Santos AJ, da Costa J, Zhang L, Pei Y, Scholey J, Ferrario CM, Manoukian AS, Chappell MC, Backx PH, Yagil Y, Penninger JM . Angiotensin-converting enzyme 2 is an essential regulator of heart function. Nature 2002; 417: 822–828.
Lavrentyev EN, Malik KU . High glucose-induced Nox1-derived superoxides downregulate PKC-betaII, which subsequently decreases ACE2 expression and ANG-(1-7) formation in rat VSMCs. Am J Physiol Heart Circ Physiol 2009; 296: H106–H118.
Ferrario CM . New physiological concepts of the renin-angiotensin system from the investigation of precursors and products of angiotensin I metabolism. Hypertension 2010; 55: 445–452.
Furchgott RF, Zawadzki JV . The obligatory role of endothelial cells in the relaxation of arterial smooth muscle by acetylcholine. Nature 1980; 288: 373–376.
Palmer RMJ, Ferridge AG, Moncada S . Nitric oxide release accounts for the biological activity of endotheliumderived relaxing factor. Nature 1987; 327: 524–526.
Landmesser U, Hornig B, Drexler H . Endothelial function: a critical determinant in atherosclerosis? Circulation 2004; 109 (21 Suppl 1): II27–II33.
Lavi S, Yang EH, Prasad A, Mathew V, Barsness GW, Rihal CS, Lerman LO, Lerman A . The interaction between coronary endothelial dysfunction, local oxidative stress, and endogenous nitric oxide in humans. Hypertension 2007; 51: 127–133.
Miller AA, Budzyn K, Sobey CG . Vascular dysfunction in cerebrovascular disease: mechanisms and therapeutic intervention. Clin Sci 2010; 119: 1–17.
Hambrecht R, Hilbrich L, Erbs S, Gielen S, Fiehn E, Schoene N, Schuler G . Correction of endothelial dysfunction in chronic heart failure: additional effects of exercise training and oral L-arginine supplementation. J Am Coll Cardiol 2000; 35: 706–713.
Landmesser U, Dikalov S, Price SR, McCann L, Fukai T, Holland SM, Mitch WE, Harrison DG . Oxidation of tetrahydrobiopterin leads to uncoupling of endothelial cell nitric oxide synthase in hypertension. J Clin Invest 2003; 111: 1201–1209.
Vasquez-Vivar J, Kalyanaraman B, Martasek P, Hogg N, Masters BS, Karoui H, Tordo P, Pritchard Jr KA . Superoxide generation by endothelial nitric oxide synthase: the influence of cofactors. Proc Natl Acad Sci USA 1998; 95: 9220–9225.
Gryglewski RJ, Palmer RM, Moncada S . Superoxide anion is involved in the breakdown of endothelium-derived vascular relaxing factor. Nature 1986; 320: 454–456.
Stroes E, Kastelein J, Cosentino F, Erkelens W, Wever R, Koomans H, Lüscher T, Rabelink T . Tetrahydrobiopterin restores endothelial function in hypercholesterolemia. J Clin Invest 1997; 99: 41–46.
Settergren M, Böhm F, Malmström RE, Channon KM, Pernow J . L-arginine and tetrahydrobiopterin protects against ischemia/reperfusion-induced endothelial dysfunction in patients with type 2 diabetes mellitus and coronary artery disease. Atherosclerosis 2009; 204: 73–78.
Katusic ZS, d’Uscio LV, Nath KA . Vascular protection by tetrahydrobiopterin: progress and therapeutic prospects. Trends Pharmacol Sci 2009; 30: 48–54.
Lowe ER, Everett AC, Lee AJ, Lau M, Dunbar AY, Berka V, Tsai AL, Osawa Y . Time-dependent inhibition and tetrahydrobiopterin depletion of endothelial nitric-oxide synthase caused by cigarettes. Drug Metab Dispos 2005; 33: 131–138.
Barua RS, Ambrose JÁ, Srivastava S, DeVoe MC, Eales-Reynolds LJ . Reactive oxygen species are involved in smoking-induced dysfunction of nitric oxide biosynthesis and upregulation of endothelial nitric oxide synthase: an in vitro demonstration in human coronary artery endothelial cells. Circulation 2003; 107: 2342–2347.
Pacher P, Beckman JS, Liaudet L . Nitric oxide and peroxynitrite in health and disease. Physiol Rev 2007; 87: 315–424.
Lusis AJ . Atherosclerosis. Nature 2000; 407: 233–241.
Heller R, Polack T, Till GU . Nitric oxide inhibits proliferation of human endothelial cells via a mechanism independent of cGMP. Atherosclerosis 1999; 144: 49–57.
Moore C, Tymvios C, Emerson M . Functional regulation of vascular and platelet activity during thrombosis by nitric oxide and endothelial nitric oxide synthase. Thromb Haemost 2010; 104: 342–349.
Kauser K, Da Cunha V, Fitch R, Mallari C, Rubanyi GM . Role of endogenous nitric oxide in progression of atherosclerosis in apolipoprotein E-deficient mice. Am J Physiol 2000; 278: H1679–H1685.
Cangemi R, Angelico F, Loffredo L, Ben MD, Pignatelli P, Martini A, Violi F . Oxidative stress-mediated arterial dysfunction in patients with metabolic syndrome: effect of ascorbic acid. Free Radic Biol Med 2007; 43: 853–859.
Katusic ZS . Superoxide anion and endothelial regulation of arterial tone. Free Radic Biol Med 1996; 20: 443–448.
Dröge W . Free radicals in the physiological control of cell function. Physiol Rev 2002; 82: 47–95.
Kojda G, Harrison D . Interaction between NO and reactive oxygen species: pathophysiological importance in atherosclerosis, hypertension, diabetes and heart failure. Cardiovasc Res 1999; 43: 562–571.
Wassmann S, Wassman K, Nickenig G . Modulation of oxidant and antioxidant enzyme expression and function in vascular cells. Hypertension 2004; 44: 381–386.
Förstermann U . Nitric oxide and oxidative stress in vascular disease. Pflugers Arch 2010; 459: 923–939.
Lassègue B, Sorescu D, Szöes K, Yin Q, Akers M, Zhang Y, Grant SL, Lambeth JD, Griendling KK . Novel gp91phox homologues in vascular smooth muscle cells; nox 1 mediates angiotensin-II-induced superoxide formation and redox-sensitive signaling pathways. Circ Res 2001; 88: 888–894.
Flammer AJ, Lüscher TF . Human endothelial dysfunction: EDRFs. Pflugers Arch 2010; 459: 1005–1013.
Delles C, Miller WH, Dominiczak AF . Targeting reactive oxygen species in hypertension. Antioxid Redox Signal 2008; 10: 1061–1077.
Furukawa S, Fujita T, Shimabukuro M, Iwaki M, Yamada Y, Nakajima Y, Nakayama O, Makishima M, Matsuda M, Shimomura I . Increased oxidative stress in obesity and its impact on metabolic syndrome. J Clin Invest 2004; 114: 1752–1761.
Fujita K, Nishizawa H, Funahashi T, Shimomura I, Shimabukuro M . Systemic oxidative stress is associated with visceral fat accumulation and the metabolic syndrome. Circ J 2006; 70: 1437–1442.
Petry A, Djordjevic T, Weitnauer M, Kietzmann T, Hess J, Gorlach A . Nox2 and Nox4 mediate proliferative response in endothelial cells. Antioxid Redox Signal 2006; 8: 1473–1484.
Görlach A, Brandes RP, Nguyen K, Amidi M, Dehghani F, Busse R . A gp91phox containing NADPH oxidase selectively expressed in endothelial cells is a major source of oxygen radical generation in the arterial wall. Circ Res 2000; 87: 26–32.
Janiszewiski M, Lopes LR, Carmo AO, Pedro MA, Brandes RP, Santos CX, Laurindo FR . Regulation of NAD(P)H oxidase by associated protein disulfide isomerase in vascular smooth muscle cells. J Biol Chem 2005; 280: 40813–40819.
Zafari AM, Ushio-Fukai M, Minieri CA, Akers M, Lassegue B, Griendling KK . Arachidonic acid metabolites mediate angiotensin II-induced NADH/NADPH oxidase activity and hypertrophy in vascular smooth muscle cells. Antioxid Redox Signal 1999; 1: 167–179.
Touyz RM, Chen X, Tabet F, Yao G, He G, Quinn MT, Pagano PJ, Schiffrin EL . Expression of a functionally active gp91phox-containing neutrophil-type NAD(P)H oxidase in smooth muscle cells from human resistance arteries: regulation by angiotensin II. Circ Res 2002; 90: 1205–1213.
Griendling KK, Minieri CA, Ollerenshaw JD, Alexander RW . Angiotensin II stimulates NADH and NADPH oxidase activity in cultured vascular smooth muscle cells. Circ Res 1994; 74: 1141–1148.
Chen K, Kirber MT, Xiao H, Yang Y, Keaney Jr JF . Regulation of ROS signal transduction by NADPH oxidase 4 localization. J Cell Biol 2008; 181: 1129–1139.
Santos RA, Brosnihan KB, Jacobsen DW, DiCorleto PE, Ferrario CM . Production of angiotensin-(1–7) by human vascular endothelium. Hypertension 1992; 19: II56–II61.
Benter IF, Ferrario CM, Morris M, Diz DI . Antihypertensive actions of angiotensin-(1-7) in spontaneously hypertensive rats. Am J Physiol 1995; 269: H313–H319.
Fraga-Silva RA, Pinheiro SV, Goncalves AC, Alenina N, Bader M, Santos RA . The antithrombotic effect of angiotensin-(1-7) involves MAS mediated NO release from platelets. Mol Med 2008; 14: 28–35.
Kucharewicz I, Pawlak R, Matys T, Pawlak D, Buczko W . Antithrombotic effect of captopril and losartan is mediated by angiotensin-(1-7). Hypertension 2002; 40: 774–779.
Tallant EA, Clark MA . Molecular mechanisms of inhibition of vascular growth by angiotensin-(1-7). Hypertension 2003; 42: 574–579.
Langeveld B, van Gilst WH, Tio RA, Zijlstra F, Roks AJ . Angiotensin-(1-7) attenuates neointimal formation after stent implantation in the rat. Hypertension 2005; 45: 138–141.
Xu P, Costa-Goncalves AC, Todiras M, Rabelo LA, Sampaio WO, Moura MM, Santos SS, Luft FC, Bader M, Gross V, Alenina N, Santos RA . Endothelial dysfunction and elevated blood pressure in MAS gene-deleted mice. Hypertension 2008; 51: 574–580.
Rabelo LA, Xu P, Todiras M, Sampaio WO, Buttgereit J, Bader M, Santos RA, Alenina N . Ablation of angiotensin (1-7) receptor Mas in C57Bl/6 mice causes endothelial dysfunction. J Am Soc Hypertens 2008; 2: 418–424.
Rentzsch B, Todiras M, Iliescu R, Popova E, Campos LA, Oliveira ML, Baltatu OC, Santos RA, Bader M . Transgenic angiotensin-converting enzyme 2 overexpression in vessels of SHRSP rats reduces blood pressure and improves endothelial function. Hypertension 2008; 52: 967–973.
Benter IF, Yousif MH, Dhaunsi GS, Kaur J, Chappell MC, Diz DI . Angiotensin-(1-7) prevents activation of NADPH oxidase and renal vascular dysfunction in diabetic hypertensive rats. Am J Nephrol 2008; 28: 25–33.
Gonzales S, Noriega GO, Tomaro ML, Peña C . Angiotensin-(1-7) stimulates oxidative stress in rat kidney. Regul Pept 2002; 106: 67–70.
Li P, Chappell MC, Ferrario CM, Brosnihan KB . Angiotensin-(1-7) augments bradykinin-induced vasodilation by competing with ACE and releasing nitric oxide. Hypertension 1997; 29: 394–400.
Paula RD, Lima CV, Britto RR, Campagnole-Santos MJ, Khosla MC, Santos RA . Potentiation of the hypotensive effect of bradykinin by angiotensin-(1-7)-related peptides. Peptides 1999; 20: 493–500.
Feterik K, Smith L, Katusic ZS . Angiotensin-(1-7) causes endothelium-dependent relaxation in canine middle cerebral artery. Brain Res 2000; 873: 75–82.
Gorelik G, Carbini LA, Scicli AG . Angiotensin 1–7 induces bradykinin mediated relaxation in porcine coronary artery. J Pharmacol Exp Ther 1998; 286: 403–410.
Peiró C, Vallejo S, Gembardt F, Azcutia V, Heringer-Walther S, Rodríguez-Mañas L, Schultheiss HP, Sánchez-Ferrer CF, Walther T . Endothelial dysfunction through genetic deletion or inhibition of the G protein-coupled receptor Mas: a new target to improve endothelial function. J Hypertens 2007; 25: 2421–2425.
Faria-Silva R, Duarte FV, Santos RAS . Short-term angiotensin(1-7) receptor MAS stimulation improves endothelial function in normotensive rats. Hypertension 2005; 46: 948–952.
Pörsti I, Bara AT, Busse R, Hecker M . Release of nitric oxide by angiotensin-(1-7) from porcine coronary endothelium: implications for a novel angiotensin receptor. Br J Pharmacol 1994; 111: 652–654. Erratum in: Br J Pharmacol 1996; 117(1): 231.
Carvalho MB, Duarte FV, Faria-Silva R, Fauler B, da Mata Machado LT, de Paula RD, Campagnole-Santos MJ, Santos RA . Evidence for Mas-mediated bradykinin potentiation by the angiotensin-(1-7) nonpeptide mimic AVE 0991 in normotensive rats. Hypertension 2007; 50: 762–767.
Lovren F, Pan Y, Quan A, Teoh H, Wang G, Shukla PC, Levitt KS, Oudit GY, Al-Omran M, Stewart DJ, Slutsky AS, Peterson MD, Backx PH, Penninger JM, Verma S . Angiotensin converting enzyme-2 confers endothelial protection and attenuates atherosclerosis. Am J Physiol Heart Circ Physiol 2008; 295: H1377–H1384.
Guo YJ, Li WH, Wu R, Xie Q, Cui LQ . ACE2 overexpression inhibits angiotensin II-induced monocyte chemoattractant protein-1 expression in macrophages. Arch Med Res 2008; 39: 149–154.
Tesanovic S, Vinh A, Gaspari TA, Casley D, Widdop RE . Vasoprotective and atheroprotective effects of angiotensin (1-7) in apolipoprotein E-deficient mice. Arterioscler Thromb Vasc Biol 2010; 30: 1606–1613.
Thomas MC, Pickering RJ, Tsorotes D, Koitka A, Sheehy K, Bernardi S, Toffoli B, Nguyen-Huu TP, Head G, Fu Y, Chin-Dusting J, Cooper ME, Tikellis C . Genetic Ace2 deficiency accentuates vascular inflammation and atherosclerosis in the ApoE knockout mouse. Circ Res 2010; 107: 888–897.
Zhang C, Zhao YX, Zhang YH, Zhu L, Deng BP, Zhou ZL, Li SY, Lu XT, Song LL, Lei XM, Tang WB, Wang N, Pan CM, Song HD, Liu CX, Dong B, Zhang Y, Cao Y . Angiotensin-converting enzyme 2 attenuates atherosclerotic lesions by targeting vascular cells. Proc Natl Acad Sci USA 2010; 107: 15886–15891.
Paravicini TM, Gulluyan LM, Dusting GJ, Drummond GR . Increased NADPH oxidase activity, gp91phox expression, and endothelium-dependent vasorelaxation during neointima formation in rabbits. Circ Res 2002; 91: 54–61.
Touyz RM . Reactive oxygen species, vascular oxidative stress, and redox signaling in hypertension: what is the clinical significance? Hypertension 2004; 44: 248–252.
Kamper M, Tsimpoukidi O, Chatzigeorgiou A, Lymberi M, Kamper EF . The antioxidant effect of angiotensin II receptor blocker, losartan, in streptozotocin-induced diabetic rats. Transl Res 2010; 156: 26–36.
Wong WT, Tian XY, Xu A, Ng CF, Lee HK, Chen ZY, Au CL, Yao X, Huang Y . Angiotensin II type 1 receptor-dependent oxidative stress mediates endothelial dysfunction in type 2 diabetic mice. Antioxid Redox Signal 2010; 13: 757–768.
Sampaio WO, Castro CH, Santos RAS, Schiffrin EL, Touyz RM . Angiotensin-(1-7) counterregulates angiotensin II signaling in human endothelial cells. Hypertension 2007; 50: 1093–1098.
Kawajiri M, Mogi M, Higaki N, Matsuoka T, Ohyagi Y, Tsukuda K, Kohara K, Horiuchi M, Miki T, Kira JI . Angiotensin-converting enzyme (ACE) and ACE2 levels in the cerebrospinal fluid of patients with multiple sclerosis. Mult Scler 2009; 15: 262–265.
Lu J, Zhang Y, Shi J . Effects of intracerebroventricular infusion of angiotensin-(1-7) on bradykinin formation and the kinin receptor expression after focal cerebral ischemia-reperfusion in rats. Brain Res 2008; 1219: 127–135.
Zheng H, Liu X, Moser JS, Patel K . Gene transfer of angiotensin converting enzyme 2 to the paraventricular nucleus improves attenuated nitric oxide mechanism in rats with chronic heart failure. FASEB J 2009; 23: 956.2.
Feng Y, Xia H, Cai Y, Halabi CM, Becker LK, Santos RA, Speth RC, Sigmund CD, Lazartigues E . Brain-selective overexpression of human angiotensin-converting enzyme type 2 attenuates neurogenic hypertension. Circ Res 2010; 106: 373–382.
Zhang Y, Lu J, Shi J, Lin X, Dong J, Zhang S, Liu Y, Tong Q . Central administration of angiotensin-(1-7) stimulates nitric oxide release and upregulates the endothelial nitric oxide synthase expression following focal cerebral ischemia/reperfusion in rats. Neuropeptides 2008; 42: 593–600.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Rights and permissions
About this article
Cite this article
Rabelo, L., Alenina, N. & Bader, M. ACE2–angiotensin-(1–7)–Mas axis and oxidative stress in cardiovascular disease. Hypertens Res 34, 154–160 (2011). https://doi.org/10.1038/hr.2010.235
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/hr.2010.235
Keywords
This article is cited by
-
The antihypertensive action of C-phycocyanin is related to the prevention of angiotensin II-caused vascular dysfunction in chronic kidney disease
Hypertension Research (2024)
-
The renin-angiotensin-aldosterone system (RAAS) signaling pathways and cancer: foes versus allies
Cancer Cell International (2023)
-
Immune landscape and redox imbalance during neurological disorders in COVID-19
Cell Death & Disease (2023)
-
Effects of olmesartan and amlodipine on blood pressure, endothelial function, and vascular inflammation
Journal of Nuclear Cardiology (2023)
-
From Molecular Pathology of COVID 19 to Nigella Sativum as a Treatment Option: Scientific Based Evidence of Its Myth or Reality
Chinese Journal of Integrative Medicine (2022)