Abstract
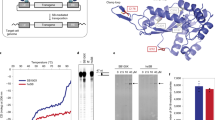
Sleeping Beauty (SB3) transposon and transposase constitute a DNA plasmid system used for therapeutic human cell genetic engineering. Here we report a comparison of SB100X, a newly developed hyperactive SB transposase, to a previous generation SB11 transposase to achieve stable expression of a CD19-specific chimeric antigen receptor (CAR3) in primary human T cells. The electro-transfer of SB100X expressed from a DNA plasmid or as an introduced mRNA species had superior transposase activity in T cells based on the measurement of excision circles released after transposition and emergence of CAR expression on T cells selectively propagated upon CD19+ artificial antigen-presenting cells. Given that T cells modified with SB100X and SB11 integrate on average one copy of the CAR transposon in each T-cell genome, the improved transposition mediated by SB100X apparently leads to an augmented founder effect of electroporated T cells with durable integration of CAR. In aggregate, SB100X improves SB transposition in primary human T cells and can be titrated with an SB transposon plasmid to improve the generation of CD19-specific CAR+ T cells.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Abbreviations
- SB:
-
Sleeping Beauty
- CAR:
-
chimeric antigen receptor
- aAPC:
-
artificial antigen presenting cell
- PBMC:
-
peripheral blood mononuclear cells
References
Jena B, Dotti G, Cooper LJ . Redirecting T-cell specificity by introducing a tumor-specific chimeric antigen receptor. Blood 2010; 116: 1035–1044.
Tan PH, Tan PL, George AJ, Chan CL . Gene therapy for transplantation with viral vectors—how much of the promise has been realized? Expert Opin Biol Ther 2006; 6: 759–772.
Ciuffi A . Mechanisms governing lentivirus integration site selection. Curr Gene Ther 2008; 8: 419–429.
Hacein-Bey-Abina S, Von Kalle C, Schmidt M, McCormack MP, Wulffraat N, Leboulch P et al. LMO2-associated clonal T cell proliferation in two patients after gene therapy for SCID-X1. Science 2003; 302: 415–419.
Liang Q, Kong J, Stalker J, Bradley A . Chromosomal mobilization and reintegration of Sleeping Beauty and PiggyBac transposons. Genesis 2009; 47: 404–408.
Grabundzija I, Irgang M, Mátés L, Belay E, Matrai J, Gogol-Döring A et al. Comparative analysis of transposable element vector systems in human cells. Mol Ther 2010; 18: 1200–1209.
Huang X, Wilber AC, Bao L, Tuong D, Tolar J, Orchard PJ et al. Stable gene transfer and expression in human primary T cells by the Sleeping Beauty transposon system. Blood 2006; 107: 483–491.
Singh H, Manuri PR, Olivares S, Dara N, Dawson MJ, Huls H et al. Redirecting specificity of T-cell populations for CD19 using the Sleeping Beauty system. Cancer Res 2008; 68: 2961–2971.
Peng PD, Cohen CJ, Yang S, Hsu C, Jones S, Zhao Y et al. Efficient nonviral Sleeping Beauty transposon-based TCR gene transfer to peripheral blood lymphocytes confers antigen-specific antitumor reactivity. Gene Therapy 2009; 16: 1042–1049.
Geurts AM, Yang Y, Clark KJ, Liu G, Cui Z, Dupuy AJ et al. Gene transfer into genomes of human cells by the Sleeping Beauty transposon system. Mol Ther 2003; 8: 108–117.
Williams DA . Sleeping Beauty vector system moves toward human trials in the United States. Mol Ther 2008; 16: 1515–1516.
Hackett PB, Largaespada DA, Cooper LJ . A transposon and transposase system for human application. Mol Ther 2010; 18: 674–683.
Mátés L, Chuah MK, Belay E, Jerchow B, Manoj N, Acosta-Sanchez A et al. Molecular evolution of a novel hyperactive Sleeping Beauty transposase enables robust stable gene transfer in vertebrates. Nat Genet 2009; 41: 753–761.
Xue X, Huang X, Nodland SE, Mátés L, Ma L, Izsvák Z et al. Stable gene transfer and expression in cord blood-derived CD34+ hematopoietic stem and progenitor cells by a hyperactive Sleeping Beauty transposon system. Blood 2009; 114: 1319–1330.
Kowolik CM, Topp MS, Gonzalez S, Pfeiffer T, Olivares S, Gonzalez N et al. CD28 costimulation provided through a CD19-specific chimeric antigen receptor enhances in vivo persistence and antitumor efficacy of adoptively transferred T cells. Cancer Res 2006; 66: 10995–11004.
Geurts AM, Yang Y, Clark KJ, Liu G, Cui Z, Dupuy AJ et al. Gene transfer into genomes of human cells by the Sleeping Beauty transposon system. Mol Ther 2003; 8: 108–117.
Manuri PV, Wilson MH, Maiti SN, Mi T, Singh H, Olivares S et al. piggyBac transposon/transposase system to generate CD19-specific T cells for treatment of B-lineage malignancies. Hum Gene Ther 2010; 21: 427–437.
Izsvák Z, Chuah MK, Vandendriessche T, Ivics Z . Efficient stable gene transfer into human cells by the Sleeping Beauty transposon vectors. Methods 2009; 49: 287–297.
Liu G, Aronovich EL, Cui Z, Whitley CB, Hackett PB . Excision of Sleeping Beauty transposons: parameters and applications to gene therapy. J Gene Med 2004; 6: 574–583.
Szilagyi A, Blasko B, Szilassy D, Fust G, Sasvari-Szekely M, Ronai Z . Real-time PCR quantification of human complement C4A and C4B genes. BMC Genet 2006; 7: 1.
Nakazawa Y, Huye LE, Dotti G, Foster AE, Vera JF, Manuri PR et al. Optimization of the PiggyBac transposon system for the sustained genetic modification of human T lymphocytes. J Immunother 2009; 32: 826–836.
Huang X, Guo H, Kang J, Choi S, Zhou TC, Tammana S et al. Sleeping Beauty transposon-mediated engineering of human primary T cells for therapy of CD19+ lymphoid malignancies. Mol Ther 2008; 16: 580–589.
Wilber A, Frandsen JL, Geurts JL, Largaespada DA, Hackett PB, McIvor RS . RNA as a source of transposase for Sleeping Beauty-mediated gene insertion and expression in somatic cells and tissues. Mol Ther 2006; 13: 625–630.
Staunstrup NH, Moldt B, Mátés L, Villesen P, Jakobsen M, Ivics Z et al. Hybrid lentivirus-transposon vectors with a random integration profile in human cells. Mol Ther 2009; 17: 1205–1214.
Vink CA, Gaspar HB, Gabriel R, Schmidt M, McIvor RS, Thrasher AJ et al. Sleeping beauty transposition from nonintegrating lentivirus. Mol Ther 2009; 17: 1197–1204.
Acknowledgements
We thank Dr Perry Hackett University of Minnesota for Sleeping Beauty system and Dr Carl June from University of Pennsylvania for assistance generating the K562-aAPC. We are grateful to the Flow Cytometry Core Laboratory at MDACC. Cancer Prevention Research Institute of Texas, Department of Defense, PO1 (CA100265), CCSG (CA16672), RO1s (CA124782, CA120956), R33 (CA116127), Alex's Lemonade Stand Foundation, Alliance for Cancer Gene Therapy, Burroughs Wellcome Fund, Gillson Longenbaugh Foundation, Harry T Mangurian, Jr, Foundation, Institute of Personalized Cancer Therapy, Leukemia and Lymphoma Society, Lymphoma Research Foundation, Miller Foundation, National Foundation for Cancer Research, Pediatric Cancer Research Foundation, National Marrow Donor Program, William Lawrence and Blanche Hughes Foundation.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies the paper on Gene Therapy website
Supplementary information
Rights and permissions
About this article
Cite this article
Jin, Z., Maiti, S., Huls, H. et al. The hyperactive Sleeping Beauty transposase SB100X improves the genetic modification of T cells to express a chimeric antigen receptor. Gene Ther 18, 849–856 (2011). https://doi.org/10.1038/gt.2011.40
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/gt.2011.40
Keywords
This article is cited by
-
AAV-mediated delivery of a Sleeping Beauty transposon and an mRNA-encoded transposase for the engineering of therapeutic immune cells
Nature Biomedical Engineering (2023)
-
Construction and Quantitative Evaluation of a Tissue-Specific Sleeping Beauty by EDL2-Specific Transposase Expression in Esophageal Squamous Carcinoma Cell Line KYSE-30
Molecular Biotechnology (2023)
-
Transposon-mediated generation of CAR-T cells shows efficient anti B-cell leukemia response after ex vivo expansion
Gene Therapy (2020)
-
Chimeric antigen receptor T cell (CAR-T) immunotherapy for solid tumors: lessons learned and strategies for moving forward
Journal of Hematology & Oncology (2018)
-
Biomanufacturing for clinically advanced cell therapies
Nature Biomedical Engineering (2018)