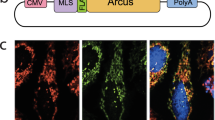
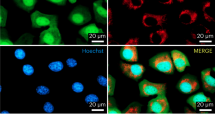
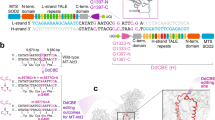
Abstract
Most pathogenic mtDNA mutations are heteroplasmic and there is a clear correlation between high levels of mutated mtDNA in a tissue and pathology. We have found that in vivo double-strand breaks (DSBs) in mtDNA lead to digestion of cleaved mtDNA and replication of residual mtDNA. Therefore, if DSB could be targeted to mutations in mtDNA, mutant genomes could be eliminated and the wild-type mtDNA would repopulate the cells. This can be achieved by using mitochondria-targeted restriction endonucleases as a means to degrade specific mtDNA haplotypes in heteroplasmic cells or tissues. In this work, we investigated the potential of systemic delivery of mitochondria-targeted restriction endonucleases to reduce the proportion of mutant mtDNA in specific tissues. Using the asymptomatic NZB/BALB mtDNA heteroplasmic mouse as a model, we found that a mitochondria-targeted ApaLI (that cleaves BALB mtDNA at a single site and does not cleave NZB mtDNA) increased the proportion of NZB mtDNA in target tissues. This was observed in heart, using a cardiotropic adeno-associated virus type-6 (AAV6) and in liver, using the hepatotropic adenovirus type-5 (Ad5). No mtDNA depletion or loss of cytochrome c oxidase activity was observed in any of these tissues. These results show the potential of systemic delivery of viral vectors to specific organs for the therapeutic application of mitochondria-targeted restriction enzymes in mtDNA disorders.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Schaefer AM, McFarland R, Blakely EL, He L, Whittaker RG, Taylor RW et al. Prevalence of mitochondrial DNA disease in adults. Ann Neurol 2008; 63: 35–39.
Haas RH, Parikh S, Falk MJ, Saneto RP, Wolf NI, Darin N et al. Mitochondrial disease: a practical approach for primary care physicians. Pediatrics 2007; 120: 1326–1333.
Horvath R, Gorman G, Chinnery PF . How can we treat mitochondrial encephalomyopathies? Approaches to therapy. Neurotherapeutics 2008; 5: 558–568.
Zeviani M, Carelli V . Mitochondrial disorders. Curr Opin Neurol 2007; 20: 564–571.
Holmgren D, Wahlander H, Eriksson BO, Oldfors A, Holme E, Tulinius M . Cardiomyopathy in children with mitochondrial disease; clinical course and cardiological findings. Eur Heart J 2003; 24: 280–288.
Towbin JA . Mitochondrial cardiology. In: Di Mauro S, Hirano M, Schon EA (eds). Mitochondrial Medicine. Informa Healthcare: Oxon, 2006, pp 75–103.
Scaglia F, Towbin JA, Craigen WJ, Belmont JW, Smith EO, Neish SR et al. Clinical spectrum, morbidity, and mortality in 113 pediatric patients with mitochondrial disease. Pediatrics 2004; 114: 925–931.
Lee WS, Sokol RJ . Mitochondrial hepatopathies: advances in genetics and pathogenesis. Hepatology 2007; 45: 1555–1565.
Vallance HD, Jeven G, Wallace DC, Brown MD . A case of sporadic infantile histiocytoid cardiomyopathy caused by the A8344G (MERRF) mitochondrial DNA mutation. Pediatr Cardiol 2004; 25: 538–540.
Wallace DC, Brown MD, Lott MT . Mitochondrial DNA variation in human evolution and disease. Gene 1999; 238: 211–230.
Chomyn A, Martinuzzi A, Yoneda M, Daga A, Hurko O, Johns D et al. MELAS mutation in mtDNA binding site for transcription termination factor causes defects in protein synthesis and in respiration but no change in levels of upstream and downstream mature transcripts. Proc Natl Acad Sci USA 1992; 89: 4221–4225.
Hanna MG, Nelson IP, Morgan-Hughes JA, Harding AE . Impaired mitochondrial translation in human myoblasts harbouring the mitochondrial DNA tRNA lysine 8344 A-->G (MERRF) mutation: relationship to proportion of mutant mitochondrial DNA. J Neurol Sci 1995; 130: 154–160.
Bayona-Bafaluy MP, Blits B, Battersby BJ, Shoubridge EA, Moraes CT . Rapid directional shift of mitochondrial DNA heteroplasmy in animal tissues by a mitochondrially targeted restriction endonuclease. Proc Natl Acad Sci USA 2005; 102: 14392–14397.
Tanaka M, Borgeld HJ, Zhang J, Muramatsu S, Gong JS, Yoneda M et al. Gene therapy for mitochondrial disease by delivering restriction endonuclease SmaI into mitochondria. J Biomed Sci 2002; 9: 534–541.
Alexeyev MF, Venediktova N, Pastukh V, Shokolenko I, Bonilla G, Wilson GL . Selective elimination of mutant mitochondrial genomes as therapeutic strategy for the treatment of NARP and MILS syndromes. Gene Ther 2008; 15: 516–523.
Bacman SR, Williams SL, Hernandez D, Moraes CT . Modulating mtDNA heteroplasmy by mitochondria-targeted restriction endonucleases in a ‘differential multiple cleavage-site’ model. Gene Ther 2007; 14: 1309–1318.
Palomeque J, Chemaly ER, Colosi P, Wellman JA, Zhou S, Del Monte F et al. Efficiency of eight different AAV serotypes in transducing rat myocardium in vivo. Gene Ther 2007; 14: 989–997.
Zincarelli C, Soltys S, Rengo G, Rabinowitz JE . Analysis of AAV serotypes 1–9 mediated gene expression and tropism in mice after systemic injection. Mol Ther 2008; 16: 1073–1080.
Huard J, Lochmuller H, Acsadi G, Jani A, Massie B, Karpati G . The route of administration is a major determinant of the transduction efficiency of rat tissues by adenoviral recombinants. Gene Ther 1995; 2: 107–115.
Jenuth JP, Peterson AC, Shoubridge EA . Tissue-specific selection for different mtDNA genotypes in heteroplasmic mice. Nat Genet 1997; 16: 93–95.
Satoh M, Kuroiwa T . Organization of multiple nucleoids and DNA molecules in mitochondria of a human cell. Exp Cell Res 1991; 196: 137–140.
Koene S, Smeitink J . Mitochondrial medicine: entering the era of treatment. J Intern Med 2009; 265: 193–209.
Srivastava S, Moraes CT . Manipulating mitochondrial DNA heteroplasmy by a mitochondrially targeted restriction endonuclease. Hum Mol Genet 2001; 10: 3093–3099.
Gardner JL, Craven L, Turnbull DM, Taylor RW . Experimental strategies towards treating mitochondrial DNA disorders. Biosci Rep 2007; 27: 139–150.
Baker AH, McVey JH, Waddington SN, Di Paolo NC, Shayakhmetov DM . The influence of blood on in vivo adenovirus bio-distribution and transduction. Mol Ther 2007; 15: 1410–1416.
Sorensen L, Ekstrand M, Silva JP, Lindqvist E, Xu B, Rustin P et al. Late-onset corticohippocampal neurodepletion attributable to catastrophic failure of oxidative phosphorylation in MILON mice. J Neurosci 2001; 21: 8082–8090.
Srivastava S, Moraes CT . Double-strand breaks of mouse muscle mtDNA promote large deletions similar to multiple mtDNA deletions in humans. Hum Mol Genet 2005; 14: 893–902.
Fukui H, Moraes CT . Mechanisms of formation and accumulation of mitochondrial DNA deletions in aging neurons. Hum Mol Genet 2009; 18: 1028–1036.
Bacman SR, Williams SL, Moraes CT . Intra- and inter-molecular recombination of mitochondrial DNA after in vivo induction of multiple double-strand breaks. Nucleic Acids Res 2009; 37: 4218–4226.
Yang L, Jiang J, Drouin LM, Agbandje-McKenna M, Chen C, Qiao C et al. A myocardium tropic adeno-associated virus (AAV) evolved by DNA shuffling and in vivo selection. Proc Natl Acad Sci USA 2009; 106: 3946–3951.
Blankinship MJ, Gregorevic P, Allen JM, Harper SQ, Harper H, Halbert CL et al. Efficient transduction of skeletal muscle using vectors based on adeno-associated virus serotype 6. Mol Ther 2004; 10: 671–678.
Gregorevic P, Blankinship MJ, Allen JM, Crawford RW, Meuse L, Miller DG et al. Systemic delivery of genes to striated muscles using adeno-associated viral vectors. Nat Med 2004; 10: 828–834.
Everett RS, Hodges BL, Ding EY, Xu F, Serra D, Amalfitano A . Liver toxicities typically induced by first-generation adenoviral vectors can be reduced by use of E1, E2b-deleted adenoviral vectors. Hum Gene Ther 2003; 14: 1715–1726.
Nicklin SA, Wu E, Nemerow GR, Baker AH . The influence of adenovirus fiber structure and function on vector development for gene therapy. Mol Ther 2005; 12: 384–393.
Kishi Y, Kuba K, Nakamura T, Wen J, Suzuki Y, Mizuno S et al. Systemic NK4 gene therapy inhibits tumor growth and metastasis of melanoma and lung carcinoma in syngeneic mouse tumor models. Cancer Sci 2009; 100: 1351–1358.
Sullivan DE, Dash S, Du H, Hiramatsu N, Aydin F, Kolls J et al. Liver-directed gene transfer in non-human primates. Hum Gene Ther 1997; 8: 1195–1206.
Shayakhmetov DM, Li ZY, Ni S, Lieber A . Analysis of adenovirus sequestration in the liver, transduction of hepatic cells, and innate toxicity after injection of fiber-modified vectors. J Virol 2004; 78: 5368–5381.
Boquet MP, Wonganan P, Dekker JD, Croyle MA . Influence of method of systemic administration of adenovirus on virus-mediated toxicity: focus on mortality, virus distribution, and drug metabolism. J Pharmacol Toxicol Methods 2008; 58: 222–232.
Minczuk M, Papworth MA, Miller JC, Murphy MP, Klug A . Development of a single-chain, quasi-dimeric zinc-finger nuclease for the selective degradation of mutated human mitochondrial DNA. Nucleic Acids Res 2008; 36: 3926–3938.
Segura MM, Monfar M, Puig M, Mennechet F, Ibanes S, Chillon M . A real-time PCR assay for quantification of canine adenoviral vectors. J Virol Methods 2010; 163: 129–136.
Sommer JM, Smith PH, Parthasarathy S, Isaacs J, Vijay S, Kieran J et al. Quantification of adeno-associated virus particles and empty capsids by optical density measurement. Mol Ther 2003; 7: 122–128.
Moraes CT, Ricci E, Petruzzella V, Shanske S, DiMauro S, Schon EA et al. Molecular analysis of the muscle pathology associated with mitochondrial DNA deletions. Nat Genet 1992; 1: 359–367.
Diaz F, Thomas CK, Garcia S, Hernandez D, Moraes CT . Mice lacking COX10 in skeletal muscle recapitulate the phenotype of progressive mitochondrial myopathies associated with cytochrome c oxidase deficiency. Hum Mol Genet 2005; 14: 2737–2748.
Acknowledgements
We are grateful to James M Allen and Jeffrey S Chamberlain for help preparing high titers of AAV6 and advice. This work was supported by PHS grant EY10804, NS041777, CA85700 and the Muscular Dystrophy Association to CTM. This project had the support of the core grant 5P30EY014801-07.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies the paper on Gene Therapy website
Supplementary information
Rights and permissions
About this article
Cite this article
Bacman, S., Williams, S., Garcia, S. et al. Organ-specific shifts in mtDNA heteroplasmy following systemic delivery of a mitochondria-targeted restriction endonuclease. Gene Ther 17, 713–720 (2010). https://doi.org/10.1038/gt.2010.25
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/gt.2010.25