Abstract
Objective
To report the occurrence of uveitis in patients with prior history of dengue fever.
Methods
A case series of patients with uveitis, presenting with ocular symptoms 3–5 months after contracting dengue fever during the dengue epidemic without any other attributable cause for uveitis. The dengue fever had subsided without any complication. None of the patients had any ocular complication during the acute dengue infection. They were treated on the lines of uveitis with local steroids, cycloplegic, and ocular hypotensive medications, when required, and oral steroid in case of posterior segment involvement. Patients’ follow-up varied from 3–5 months.
Results
Six patients with serological evidence of prior dengue fever had presented with uveitis not attributable to any other disease. Seven eyes of six patients were affected. Anterior uveitis without any evidence of posterior segment involvement was present in six eyes of five patients whereas one patient had severe vitritis also. The most common symptom was progressive decrease in vision and only three out of seven eyes had evidence of ciliary congestion. All patients promptly responded to the treatment and visual acuity returned to 6/9 or better in five eyes, while 6/24 and 6/18 in other two eyes due to the presence of cataract. None of these patients had recurrence during the follow-up period.
Conclusion
Uveitis in dengue infection is rare and ophthalmologists should be aware of these delayed ophthalmic complications. The prognosis of post-dengue uveitis is good.
Similar content being viewed by others
Introduction
Dengue is a mosquito-borne infection found in tropical and subtropical regions in more than 100 countries worldwide.1 Dengue virus belongs to the genus Flavivirus (family: Flaviviridae). Aedes aegypti is the major vector for human transmission in urban areas and Aedes albopictus is the major vector in the suburban and the rural areas.2, 3 There are four serologically distinct, but closely related viruses that cause dengue fever. Recovery from one infection provides lifelong immunity against that serotype but confers only transient and partial protection against heterologous infections,2 and sequential infections may increase the risk of more serious systemic as well as ocular disease.3
Approximately 100 million cases of dengue fever (DF) and 450 thousands cases of dengue hemorrhagic fever (DHF) and dengue shock syndrome (DSS) are estimated to occur annually, whereas approximately 2.5 billion people live in areas at risk of dengue virus transmission.4 A dengue outbreak was witnessed for the first time in September 2003 in Southern India.5 In 2006, India again witnessed an epidemic of dengue and type-3 serotype was found to be the etiological agent for the epidemic.
Ocular manifestations reported to be associated with dengue infection are mostly posterior segment manifestations like macular oedema, vascular occlusions, chorioretinits, vasculitis with related retinal bleeding, or cotton wool spots.6, 7, 8 Anterior segment manifestation has mostly been reported in the form of subconjunctival hemorrhage, which is related to thrombocytopenia.9
Anterior uveitis has been rarely described in the acute dengue infection.8, 10 Ocular manifestations are described mostly during the acute stage of the disease. We, hereby, report six cases of uveitis with history of prior dengue infection.
Methods
We describe a prospective case series of six patients who were seen at the Jawaharlal Institute of Postgraduate Medical Education and Research (Southern India) over 5 months from January 2007 to May 2007 and had uveitis with a prior history of dengue infection, within the previous 3 to 5 months. All of these patients had presented during the acute phase of the disease to our hospital and were on follow-up with the infectious disease physician before the ocular symptoms developed. The diagnosis of dengue infection during the acute phase was made on the basis of characteristic constellation of signs and symptoms and dengue serology (IgM). All the patients had undergone routine ophthalmic examination during the acute phase but had no ophthalmic complications during the acute phase.
At the time of presentation with ophthalmic complaints, detailed history and clinical examination was carried out to rule out any other systemic cause for uveitis. All patients had visual acuity measured with a Snellen acuity chart. All underwent slit-lamp anterior segment examination as well as dilated fundus examination with slit-lamp biomicroscopy. Fluorescein angiography (FAG) was carried out in all these patients.
Investigations on the lines of uveitis and vasculitis, like serology for angiotensin-converting enzyme levels (ACE), HLA-B27 typing, and 24-h urinary calcium (for sarcoidosis), were carried out. Also, investigations to rule out infectious etiology, like syphilis, pulmonary tuberculosis (chest X-ray and mantoux test), and viral (serology for Varicella zoster and Herpes simplex virus-1 DNA), were carried out. Additional investigations, like antinuclear antibodies (ANA), antineutrophilic cytoplasmic antibodies (ANCA), and syphilis serology, were done in the sixth patient as she had retinal vasculitis. Rheumatoid factor (RF) was estimated in the third patient, as he had a history of arthritis over the previous 4–5 months.
These patients were treated on the line of standard treatment for uveitis and their follow-up varied from 3–6 months depending upon the merit of the individual cases. Dengue IgM antibodies were estimated using ELISA (Rapid signal dengue IgM/IgG strips by Orgenics).
Results
Six patients were enrolled in the study, and the male/female ratio was 2 : 4. The age distribution was from 41 to 65 years. All the patients had negative serology for ANA, ANCA, ACE, HLA-B27, syphilis, and herpes/varicella. Urinary calcium profile and chest X-ray were also normal.
The most common presenting symptom was the progressive decrease in the vision, which was painless in four patients. Five of them had uveitis only in one eye and only one patient (case no. 3) had bilateral uveitis. Inflammation was limited to the anterior segment in all the cases except in one patient (case no. 6), who also had vitritis and retinal vasculitis. This patient had persistent symptoms over the previous 1 month but had neglected the symptoms. The cause of decrease of vision was media haze and poor corneal clarity. Visual acuity at presentation varied from finger counting to 6/12, with 4 patients having visual acuity less than 6/24. All of these patients (except patient nos. 1 and 5) also had other contributable causes for the decrease in the vision; however, with the subsidence of uveitis, visual acuity improved, despite the persistence of ocular comorbid conditions, suggesting that uveitis was also contributing to the decrease in the visual acuity.
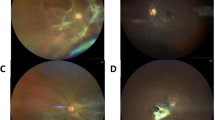
One peculiar feature of the uveitis in these patients was the presence of minimal or no ciliary congestion even in the presence of severe anterior segment inflammation. The keratitic precipitates varied in size from fine to medium to large in size and were distributed all over the cornea in almost all the cases. FAG did not reveal any evidence of posterior segment abnormality in the first five patients (case no. 1–5). However, the last patient (case no. 6) had evidence of vasculitis and retinal hemorrhages in the perimacular region, which became more evident as the ocular media cleared with the institution of treatment. Ocular coherence tomography revealed evidence of macular edema (dengue maculopathy) in the sixth patient. None of the patients had undergone any electrodiagnostic testing.
The treatment protocol included local steroids, like prednisolone eyedrops, subconjunctival dexamethosone, or subtenon triamcinolone, depending upon the severity of inflammation and the extent of the involvement of the posterior segment. Oral prednisolone (1 mg/kg body weight) was given only in the sixth patient for 2 weeks and was tapered over the next 2 weeks. Ocular hypotensive medications used were timolol eyedrops and oral acetazolamide. The clinical details of the patients have been in detailed in Table 1.
Discussion
Dengue infection is known to cause fever, headaches, myalgia, thrombocytopenia, and also hypotension especially in DSS/DHS causing high morbidity and mortality. Ophthalmic symptoms due to dengue infection are now being reported more commonly in the medical literature. Ocular complications reported by Lim et al7 were mainly confined to the macula. However, in the study by Chan DP et al8 various ophthalmic complications of dengue infections included involvement of both the posterior segment (peripheral retina) and the anterior segment (anterior uveitis in only one case), suggesting widespread inflammatory processes in the eye.
Dengue fever can impair the vision ranging from mild blurring of vision to catastrophic and severe blindness. In the largest series of ophthalmic complications of acute dengue infection, intraretinal hemorrhages (45%) were the most common feature and were usually seen in association with vasculitis.10 In the same study, anterior uveitis was seen in 17%, vitritis was seen in 31%, and both anterior uveitis and vitritis in 11%.10 The incidence of dengue maculopathy has been reported to be 10% in acute dengue infections in a recent report.11
Dengue maculopathy is the presence of macular swelling, hemorrhages, and yellow spots at macula due to the affect on retinal or choroidal vasculature. Some patients recover spontaneously to near normal levels of vision, whereas others do not respond to treatment at all.10, 11
Precise mechanisms responsible for various ocular complications in dengue are not known. The ocular features peak in acute dengue infection corresponds to the nadir of level of platelets and, thus, a common immune-mediated pathogenesis has been suggested for these manifestations.8, 10, 11 Low complement C3 and C4 in dengue patients further suggests immune-mediated mechanism.11 The possibility of specific autoantibodies production against various tissues of eye has been suggested.7, 12 This includes the possibility of production of specific autoantibodies against retina, retinal pigment epithelium, and even choroid.7, 13, 14 However, the precise mechanism responsible for the various ocular tissues alteration, leading to sensitization in dengue and there by, causing uveitis, is still unknown.9
The release of cytokines with vasoactive and procoagulant properties in response to immunological activation might explain the occurrences of retinal vascular occlusion seen in patients with dengue infection.6, 7 In addition, these inflammatory mediators cause capillary leakage and breakdown of the blood aqueous barrier, resulting in anterior uveitis.15, 16
All of the patients in this case series presented with an acute infection between October, 2006 and January, 2007. All of them had DF and not DHF/DSS. During the same period, 1686 patients of DF and 33 patients of DHF/DSS were seen by the infectious disease specialist in our hospital. Ocular manifestations were present in 119 (7.1%) out of 1686 of DF patients and 18 (54.5%) out of 33 patients of DHF/DSS. Most commonly ocular manifestation seen was subconjunctival hemorrhage, and other common manifestations seen were retinal hemorrhages, hard exudates, and soft exudates. However, none of our patients had any ocular manifestation during the acute phase and there was no significant difference during acute presentation between the patients who developed uveitis and those who did not develop.
A remarkable distinction between our series and the other published reports is that ocular complication in the form of uveitis did not occur at the time of infection, but rather occurred 3–4 months after the infection had completely resolved. We should be aware of the delayed ophthalmic complications of dengue infection like uveitis, which might occur even after complete recovery from the systemic disease.
To conclude, dengue virus results in a spectrum of ocular manifestations, ranging from nonspecific symptoms to symptomatic retinal hemorrhages and from nonimmunological to immunological manifestations like uveitis. The etiopathogenic relationship of the dengue virus and ocular complications merits further investigation.
References
WHO. Dengue and Dengue Haemorrhagic Fever, Fact Sheet No. 117 WHO: Geneva, 2002. http://www.who.int/mediacentre/factsheets/f117/en/print.html.
Gibbons RV, Vaughn DW . Dengue: an escalating problem. Br Med J 2002; 324: 1563–1566.
Nimmannitya S . Clinical manifestations of dengue/dengue haemorrhagic fever. In: Thongcharoen P (ed). Monograph on Dengue/Dengue Haemorrhagic Fever. Regional Publication No. 22. World Health Organization, Regional Office for South-East Asia: New Delhi, 1993 pp 48–54.
Gubler DJ, Clark GG . Dengue/dengue hemorrhagic fever: the emergence of a global health problem. Emerg Infect Dis 1995; 1: 55–57.
Hoti SL, Soundravally R, Rajendran G . Dengue and dengue haemorrhagic fever outbreak in Pondicherry, South India, during 2003–2004: emergence of DENV-3. Dengue Bull 2006; 30: 42–50.
Cruz-Villegas V, Berrocal AM, Davis JL . Bilateral choroidal effusions associated with dengue fever. Retina 2003; 23: 576–578.
Lim WK, Mathur R, Koh A, Yeoh R, Chee SP . Ocular manifestations of dengue fever. Ophthalmology 2004; 111: 2057–2064.
Chan DP, Teoh SC, Tan CS, Nah GK, Rajagopalan R, Prabhakaragupta MK et al. Ophthalmic complications of dengue. Emerg Infect Dis 2006; 12: 285–289.
Kapoor HK, Bhai S, John M, Xavier J . Ocular manifestations of dengue fever in an East Indian epidemic. Can J Ophthalmol 2006; 41: 741–746.
Bacsal KE, Chee SP, Cheng CL, Flores JV . Dengue-associated maculopathy. Arch Ophthalmol 2007; 125: 501–510.
Su DH, Bacsal K, Chee SP, Flores JV, Lim WK, Cheng BC et al. Dengue maculopathy study group. Prevalence of dengue maculopathy in patients hospitalized for dengue fever. Ophthalmology 2007; 114: 1743–1747.
Halstead SB . Immunological parameters of togaviruses disease syndromes. In: Schlesinger RW (ed). The Togaviruses. Biology, Structure, Replication, Academic Press, New York, 1980, pp 107–174.
Gamble CN, Aronson SB, Brescia FB . Experimental uveitis.1. The pathogenesis of recurrent immunologic (Auer) uveitis and its relationship to increased uveal vascular permeability. Arch Ophthalmol 1970; 84: 321–330.
Wong VG, Anderson RR, McMaster PR . Endogenous immune uveitis. The role of serum sickness. Arch Ophthalmol 1971; 85: 93–102.
Kurane I, Ennis FE . Immunity and immunopatholgy in dengue virus infections. Semin Immunol 1992; 4: 121–127.
Kurane I, Innis BL, Nimmannitya S, Nisalak A, Meager A, Janus J et al. Activation of T lymphocytes in dengue virus infections. High levels of soluble interleukin 2 receptor, soluble CD4, soluble CD8, interleukin2 and interferon-γ in sera of children with dengue. J Clin Invest 1991; 88: 1473–1480.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Gupta, A., Srinivasan, R., Setia, S. et al. Uveitis following dengue fever. Eye 23, 873–876 (2009). https://doi.org/10.1038/eye.2008.124
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/eye.2008.124
Keywords
This article is cited by
-
Ophthalmic complications during the dengue epidemic in Reunion Island in 2020: a case series and review of the literature
BMC Infectious Diseases (2023)
-
Ocular manifestations of emerging viral diseases
Eye (2021)
-
Clinical profile and response to steroids in post-fever retinitis: a nine-year experience from a referral institute in the rural hinterland of Central India
International Ophthalmology (2021)
-
The eye in dengue fever, a rarely appreciated aspect of dengue expanded syndrome: a case report
Journal of Medical Case Reports (2019)
-
Arboviruses and the eye
International Journal of Retina and Vitreous (2017)