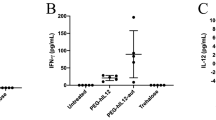
Abstract
The lung is one target organ to which solid tumors frequently metastasize. Given the systemic adverse effects of currently available treatments, developing effective strategies of drug/gene delivery directly to the lungs is therefore needed. Aerosol delivery is a non-invasive gene transfer approach to target the airways. Here, we sought to evaluate the potential to deliver a fractalkine (FKN)-encoding plasmid formulated with the tetrafunctional amphiphilic block copolymer 704 through aerosolization in two models of pulmonary metastases. FKN is a chemokine recently described as a good candidate to stimulate a strong antitumor immune response in various forms of cancers. Here, we have assessed the effect of single and repeated aerosolizations of FKN-encoding plasmid formulated with 704 on the development of experimental lung metastases of mouse colon carcinoma and osteosarcoma. For this purpose, we have designed FKN-Fc sequences encoding an optimized version of the chemokine. Repeated intratracheal administrations of 704/FKN-Fc markedly inhibited growth of experimental lung metastases of CT-26 and K7M2 cells. Our results showed that tetrafunctional amphiphilic block copolymer 704 is a highly efficient synthetic vector for mediating local and safe gene transfer into the lung. In addition, FKN-Fc gene therapy of pulmonary nodules may provide a promising immunotherapeutic approach.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Abbreviations
- FKN:
-
fractalkine
- PEI:
-
polyethyleneimine
- CAT:
-
chloramphenicol acetyl transferase
- IL-6:
-
interleukin-6
- NK cell:
-
natural killer cell
References
Jemal A, Siegel R, Ward E, Hao Y, Xu J, Thun MJ . Cancer statistics, 2009. CA Cancer J Clin 2009; 59: 225–249.
Homey B, Muller A, Zlotnik A . Chemokines: agents for the immunotherapy of cancer? Nat Rev Immunol 2002; 2: 175–184.
Rosenblatt JD, Shin SU, Nechustan H, Yi KH, Tolba K . Potential role of chemokines in immune therapy of cancer. Isr Med Assoc J 2002; 4: 1054–1059.
Flanagan K, Kaufman HL . Chemokines in tumor immunotherapy. Front Biosci 2006; 11: 1024–1030.
Gao JQ, Okada N, Mayumi T, Nakagawa S . Immune cell recruitment and cell-based system for cancer therapy. Pharm Res 2008; 25: 752–768.
Ohta M, Tanaka F, Yamaguchi H, Sadanaga N, Inoue H, Mori M . The high expression of fractalkine results in a better prognosis for colorectal cancer patients. Int J Oncol 2005; 26: 41–47.
Mlecnik B, Tosolini M, Charoentong P, Kirilovsky A, Bindea G, Berger A et al. Biomolecular network reconstruction identifies T-cell homing factors associated with survival in colorectal cancer. Gastroenterology 2010; 138: 1429–1440.
Lavergne E, Combadiere B, Bonduelle O, Iga M, Gao JL, Maho M et al. Fractalkine mediates natural killer-dependent antitumor responses in vivo. Cancer Res 2003; 63: 7468–7474.
Guo J, Zhang M, Wang B, Yuan Z, Guo Z, Chen T et al. Fractalkine transgene induces T-cell-dependent antitumor immunity through chemoattraction and activation of dendritic cells. Int J Cancer 2003; 103: 212–220.
Zeng Y, Jiang J, Huebener N, Wenkel J, Gaedicke G, Xiang R et al. Fractalkine gene therapy for neuroblastoma is more effective in combination with targeted IL-2. Cancer Lett 2005; 228: 187–193.
Xin H, Kikuchi T, Andarini S, Ohkouchi S, Suzuki T, Nukiwa T et al. Antitumor immune response by CX3CL1 fractalkine gene transfer depends on both NK and T cells. Eur J Immunol 2005; 35: 1371–1380.
Nukiwa M, Andarini S, Zaini J, Xin H, Kanehira M, Suzuki T et al. Dendritic cells modified to express fractalkine/CX3CL1 in the treatment of preexisting tumors. Eur J Immunol 2006; 36: 1019–1027.
Vitale S, Cambien B, Karimdjee BF, Barthel R, Staccini P, Luci C et al. Tissue-specific differential antitumour effect of molecular forms of fractalkine in a mouse model of metastatic colon cancer. Gut 2007; 56: 365–372.
Brueckmann M, Borggrefe M . Therapeutic potential of fractalkine: a novel approach to metastatic colon cancer. Gut 2007; 56: 314–316.
Xin H, Sun R, Kanehira M, Takahata T, Itoh J, Mizuguchi H et al. Intratracheal delivery of CX3CL1-expressing mesenchymal stem cells to multiple lung tumors. Mol Med 2009; 15: 321–327.
Momin EN, Mohyeldin A, Zaidi HA, Vela G, Quinones-Hinojosa A . Mesenchymal stem cells: new approaches for the treatment of neurological diseases. Curr Stem Cell Res Ther 2010; 5: 326–344.
Bar J, Herbst RS, Onn A . Targeted drug delivery strategies to treat lung metastasis. Expert Opin Drug Deliv 2009; 6: 1003–1016.
Densmore CL, Orson FM, Xu B, Kinsey BM, Waldrep JC, Hua P et al. Aerosol delivery of robust polyethyleneimine-DNA complexes for gene therapy and genetic immunization. Mol Ther 2000; 1: 180–188.
Goula D, Becker N, Lemkine GF, Normandie P, Rodrigues J, Mantero S et al. Rapid crossing of the pulmonary endothelial barrier by polyethylenimine/DNA complexes. Gene Ther 2000; 7: 499–504.
Desigaux L, Gourden C, Bello-Roufaï M, Richard P, Oudrhiri N, Lehn P et al. Nonionic amphiphilic block copolymers promote gene transfer to the lung. Hum Gene Ther 2005; 16: 821–829.
Pitard B, Bello-Roufaï M, Lambert O, Richard P, Desigaux L, Fernandes S et al. Negatively charged self-assembling DNA/poloxamine nanospheres for in vivo gene transfer. Nucleic Acids Res 2004; 32: e159.
Ferrari S, Moro E, Pettenazzo A, Behr JP, Zacchello F, Scarpa M . ExGen 500 is an efficient vector for gene delivery to lung epithelial cells in vitro and in vivo. Gene Ther 1997; 4: 1100–1106.
McIlroy D, Barteau B, Cany J, Richard P, Gourden C, Conchon S et al. DNA/amphiphilic block copolymer nanospheres promote low-dose DNA vaccination. Mol Ther 2009; 17: 1473–1481.
Pollard H, Remy JS, Loussouarn G, Demolombe S, Behr JP, Escande D . Polyethylenimine but not cationic lipids promotes transgene delivery to the nucleus in mammalian cells. J Biol Chem 1998; 273: 7507–7511.
Piron J, Quang KL, Briec F, Amirault JC, Leoni AL, Desigaux L et al. Biological pacemaker engineered by nonviral gene transfer in a mouse model of complete atrioventricular block. Mol Ther 2008; 16: 1937–1943.
Ziady AG, Gedeon CR, Miller T, Quan W, Payne JM, Hyatt SL et al. Transfection of airway epithelium by stable PEGylated poly-L-lysine DNA nanoparticles in vivo. Mol Ther 2003; 8: 936–947.
Loebinger MR, Janes SM . Stem cells as vectors for antitumour therapy. Thorax 2010; 65: 362–369.
Yang Y, Li Q, Ertl HC, Wilson JM . Cellular and humoral immune responses to viral antigens create barriers to lung-directed gene therapy with recombinant adenoviruses. J Virol 1995; 69: 2004–2015.
Van Ginkel FW, Liu C, Simecka JW, Dong JY, Greenway T, Frizzell RA et al. Intratracheal gene delivery with adenoviral vector induces elevated systemic IgG and mucosal IgA antibodies to adenovirus and beta-galactosidase. Hum Gene Ther 1995; 6: 895–903.
Guo J, Chen T, Wang B, Zhang M, An H, Guo Z et al. Chemoattraction, adhesion and activation of natural killer cells are involved in the antitumor immune response induced by fractalkine/CX3CL1. Immunol Lett 2003; 89: 1–7.
Tang DW, Li LF, Yu YM, Liu XY, Su XD, Zhao X et al. Preparation, crystallization and preliminary X-ray analysis of threonine synthase from Streptococcus mutans. Protein Pept Lett 2007; 14: 836–838.
Kobayashi M, Fitz L, Ryan M, Hewick RM, Clark SC, Chan S et al. Identification and purification of natural killer cell stimulatory factor (NKSF), a cytokine with multiple biologic effects on human lymphocytes. J Exp Med 1989; 170: 827–845.
Gately MK, Wolitzky AG, Quinn PM, Chizzonite R . Regulation of human cytolytic lymphocyte responses by interleukin-12. Cell Immunol 1992; 143: 127–142.
Manetti R, Parronchi P, Giudizi MG, Piccinni MP, Maggi E, Trinchieri G et al. Natural killer cell stimulatory factor (interleukin 12 [IL-12]) induces T helper type 1 (Th1)-specific immune responses and inhibits the development of IL-4-producing Th cells. J Exp Med 1993; 177: 1199–1204.
Hsieh CS, Macatonia SE, Tripp CS, Wolf SF, O’Garra A, Murphy KM . Development of TH1 CD4+ T cells through IL-12 produced by Listeria-induced macrophages. Science 1993; 260: 547–549.
Acknowledgements
We thank Violette Breittmayer for excellent technical assistance, Clothilde Gourden for excellent technical expertise in in vivo experiments, In-Cell-Art for providing tetrafunctional amphiphilic block copolymers and the Animal Facility of the Centre Méditerranéen de Médecine Moléculaire (Institut National de la Santé et de la Recherche Médicale, Unit 895). This research was supported in part by the Institut National de la Santé et de la Recherche Médicale, by the Centre National de la Recherche Scientifique, by the Association pour la Recherche sur le Cancer (ARC, Grant 3707), by Vaincre La Mucoviscidose (Paris, France) and the Association Française contre les Myopathies (Evry, France). PR-F was the recipient of a fellowship from the ARC.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest, except B Pitard who owns stocks in In-Cell-Art Co., which commercializes amphiphilic block copolymers.
Rights and permissions
About this article
Cite this article
Richard-Fiardo, P., Cambien, B., Pradelli, E. et al. Effect of fractalkine-Fc delivery in experimental lung metastasis using DNA/704 nanospheres. Cancer Gene Ther 18, 761–772 (2011). https://doi.org/10.1038/cgt.2011.42
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/cgt.2011.42