Key Points
-
Lasers have been advocated to address the clinical preparation of restorative cavities and also the pain associated with conservative dentistry.
-
All oral hard tissues can be ablated with laser energy.
-
Precision, preservation of healthy tissue and control of temperature rise are hallmarks of correct laser use on hard tissue.
-
The development of interceptive techniques to address early carious lesions may herald a more widespread use of lasers in restorative dentistry.
Key Points
Lasers in dentistry
-
1
Introduction, history of lasers and laser light production
-
2
Laser-tissue interaction
-
3
Low-level laser use in dentistry
-
4
Lasers and soft tissue: 'loose' soft tissue surgery
-
5
Lasers and soft tissue: 'fixed' soft tissue surgery
-
6
Lasers and soft tissue: periodontal therapy
-
7
Surgical laser use in implantology and endodontics
-
8
Surgical lasers and hard dental tissue
-
9
Laser regulation and safety in general dental practice
Abstract
The cutting of dental hard tissue during restorative procedures presents considerable demands on the ability to selectively remove diseased carious tissue, obtain outline and retention form and maintain the integrity of supporting tooth tissue without structural weakening. In addition, the requirement to preserve healthy tissue and prevent further breakdown of the restoration places the choice of instrumentation and clinical technique as prime factors for the dental surgeon. The quest for an alternative treatment modality to the conventional dental turbine has been, essentially, patient-driven and has led to the development of various mechanical and chemical devices. The review of the literature has endorsed the beneficial effects of current laser machines. However utopian, there is additional evidence to support the development of ultra-short (nano- and femto-second) pulsed lasers that are stable in use and commercially viable, to deliver more efficient hard tissue ablation with less risk of collateral thermal damage. This paper explores the interaction of laser energy with dental hard tissues and bone and the integration of current laser wavelengths into restorative and surgical dentistry.
Similar content being viewed by others
The development of the 'laser drill'
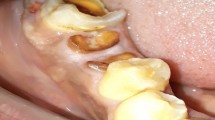
The fundamental concept of laser energy with oral tissue components has been explored in earlier papers in this series. The first laser wavelengths to be made available for general dental practice use were the Nd:YAG (1,064 nm) laser together with the carbon dioxide (10,600 nm) laser, which had already found acceptance in oral and maxillofacial surgery. Both laser wavelengths, in their emission mode configuration, had been in use for some time in their capacity to ablate soft tissue. Regrettably, the Nd:YAG 'dental laser' (free-running pulsed – 150 μs pulse width) was marketed as being suitable in tooth cavity preparation – a claim that was quickly deemed to be erroneous for clinical relevance. Early research into this claim supported the ablative effect of the 1,064 nm wavelength on accessible pigmented carious lesions,1,2,3 but whenever healthy enamel and dentine was exposed to the laser energy, the comparatively long pulse width and associated heat transfer, combined with the lack of water spray, resulted in thermal cracking and melting of hydroxyapatite (Figs 1 and 2), together with high intra-pulpal temperatures, as investigated by a number of workers.4,5,6,7,8,9,10,11 It is pertinent to note that thermal cracking is also evident with rotary instrumentation (Fig. 3).
Interestingly, it was discovered that the reformed, amorphous hydroxyapatite in post-laser sites was more resistant to acid dissolution and some studies were published advocating the use of the Nd:YAG laser in a quasi-fissure sealant technique for erupted posterior teeth.12,13,14,15,16
The carbon dioxide wavelength and emission mode (continuous or gated continuous wave) of commercially available lasers also made it impractical for restorative procedures (Figs 4,5,6). Although there is a high absorption peak of this wavelength (in the region of 10,000 nm) by carbonated hydroxyapatite (CHA), the continuous wave emission of laser energy and lack of axial water coolant resulted in carbonisation, cracking and melting of tooth tissue.17,18,19 During laser light generation, the slow decay from the energised state in the CO2 active medium results, potentially, in a number of individual wavelengths from this laser (9,300, 9,600, 10,300 and 10,600 nm). If one of the shorter wavelengths other than the usual 10,600 nm is selected, the absorption coefficient of CHA (carbonate group) increases greatly.20,21 Featherstone and co-workers at UCSF (California, USA) have investigated extensively with an experimental ultra-pulsed 9,300 and 9,600 nm laser, the ablation rates of enamel and dentine, with much success. In addition, the removal, through ablation, of the carbonate group from the CHA molecule results in a greatly increased acid-resistant compound.22,23,24,25,26
The development of the Er:YAG and Er,Cr:YSGG lasers and investigations of their action on dental tissues shifted the emphasis from 'fringe' beneficial action of laser light towards a true ablation, that did not cause thermal or mechanical damage to the tooth or pulp and was sufficiently fast to be acceptable in a clinical setting.27,28,29 The prime chromophore of the erbium YAG wavelength is water and the free-running micro-pulse emission mode results in rapid and expansive vaporisation (Fig. 7). When exposed to this wavelength, the small amounts of water contained in enamel and dentine are vaporised, causing an explosive dislocation of the gross structure (Figs 8,9,10).
Clinically, this is seen as ejection of micro-fragments of tooth tissue within the laser plume and the change in pressure in the immediately surrounding air results in an audible 'popping' sound. In target tissue that has greater water content (caries > dentine > enamel), the popping sound is louder. With experience, this can aid the clinician in selectively ablating carious vs non-carious tissue.30 Compared to near-infrared wavelengths, the explosive outward effect of erbium laser energy results in minimal thermal diffusion through the tooth structure.
Co-axial with this laser is a water spray, to aid in dispersing ablation products and to provide cooling of the target site. This is vitally important as, in a situation of laser incident energy commensurate with tooth cutting, the ablation front will be in advance of the thermal front. Insufficient water coolant and consequent dispersal of ablation products during cavity preparation can lead to a build-up of eschar, which can become super-heated (Fig. 11). Conduction of this thermal energy to surrounding tooth tissue will lead to morphological cracking and melting, pain and possible pulpal damage.31 An additional factor may be the shielding effect of the ejected debris and laser plume products into the line of the incident beam (Fig. 12).32,33 The development of ultra-short pulse laser emissions of the erbium group of wavelengths appears promising in reducing the conductive heat potential, whilst increasing the rates of tissue ablation.
The other major laser wavelength that is applicable to tooth and bone ablation is the Er,Cr:YSGG (2,780 nm). This laser is similar to the Er:YAG in that it is absorbed by water and both wavelengths ablate through vaporisation of interstitial water,34 although the absorption coefficient is slightly lower (4,000 cm−1 vs 13,000 cm−1 for Er:YAG). When one examines the absorption curve of CHA (enamel), there is a peak, coincident with 2,700 nm, representing absorption by the hydroxyl group (OH−) contained in the mineral molecule (Fig. 13).
Absorption curve of carbonated hydroxyapatite (CHA) interaction with Er,Cr:YSGG, Er:YAG and CO2 laser wavelengths. Peaks at 2,700 nm and 2,900 nm correspond to OH− group and free water; a small peak at approx 7,000 nm is coincident with (CO3)2− group absorption; absorption at 9,600 nm is coincident with the phosphate group in the molecule; water absorption is shown as a dotted line. (Produced in conjunction with Prof. J. Featherstone, UCSF, California, USA)
It is thought that the simultaneous ablation of this radical, with concomitant rapid heating of the mineral together with some direct vaporisation of whole water in hard tissue, contributes to the explosive dislocation of the target tissue when using this laser.33 Claims have been made as to the involvement of the atomised water spray, used with the erbium YSGG and referred to as a hydro-kinetic effect.35 The hypothesis draws on the postulation that water droplets axial to the laser beam absorb kinetic energy and are accelerated to aid hard tissue ablation. Investigation into this effect has questioned the validity of such claims and in addition, with comparable incident energies, suggested that the ablation rate of the erbium YSGG with enamel is slightly slower than that of erbium YAG.34 As mentioned above, this might be explained by the absorption dynamics of the YSGG and transfer of conductive heat from apatite to free water, although Walsh and Cummings have suggested that absorption of mid-infrared radiation by water is a function of temperature and pressure, both of which rise rapidly during an ablative laser pulse.36 Apel et al. further confirmed this slight difference in their investigation into the ablation threshold of enamel with these lasers, finding values of 9-11 J cm−2 for Er:YAG and 10-14 J cm−2 for Er,Cr:YSGG.37 Nonetheless, both laser wavelengths allow cavity preparation within acceptable clinical parameters.38
Early erbium lasers had rudimentary hand-pieces which were comparatively heavy. In addition, the delivery of laser energy through a non-contact sapphire window proved inaccurate in delivery of precise cutting action. Newer developments have resulted in balanced waveguides or low-OH− fibres, together with hand-pieces that are similar to turbines and use contact tips (Figs 14,15,16,17). Tissue ablation results from end-on emission of laser energy from the tip, which should be moved gently over the tooth surface to develop the cavity.
Numerous studies have been carried out to investigate the ablation rates of both wavelengths with enamel, dentine and caries, together with non-metallic or non-thermo-fused ceramic restoratives such as direct composite resin and glass-ionomer cements (Figs 18,19,20). Under normal operating parameters, pulpal temperature has been shown to rise minimally (<5°C) during laser-assisted cavity preparation. Comparisons have been advantageously made between laser use and rotary instrumentation, although speed comparisons fall below that obtained with an airotor.
The gross and micro-appearance of a 'laser' cavity in tooth tissue is essentially a crater form (Fig. 21), which is markedly different to the 'classical' cavity form obtained with rotary instrumentation and consistent with the production of stable amalgam restorations.39 However, the micro-dislocation of mineral at the cavity edges, visually evident as 'etched' in appearance, can be beneficially employed in aiding the bonding of composite resin materials (Fig. 22).
Studies carried out into the marginal integrity of such restorations reflect a poor stability, partly explained by a post-ablation weakness in marginal enamel.40,41,42 Interestingly, one study comparing laser and bur preparation but both without acid etching, found that marginal integrity was greater following laser preparation.43 However, when the cut surface is further treated with conventional acid-etch techniques, this improves the longevity of the restoration, with some enhanced bond-strength.44,45 This facility can be employed as an adjunct where restorative procedures requiring facial or incisal bonding of direct composite resin materials is required, or in the placement of orthodontic brackets (Figs 23,24,25,26).
Pain perception during laser-assisted cavity preparation
The avoidance of pain during restorative and dental surgical procedures remains a strong factor in promoting patient acceptance of treatment and many studies have been carried out to evaluate this.46,47,48,49,50,51 The use of the Nd:YAG laser in developing pulpal analgesia, possibly through interference with the 'gate theory' of neural stimulus propagation, has been suggested, although investigation into the subjectivity or placebo effect has rendered its application inconsistent.52,53,54
Perhaps of greater significance in addressing claims of pain avoidance during laser-assisted tooth preparation is the lack of tactile and thermal stimulation compared to rotary instrumentation. In addition, there is the patient-centred factor of previous experience of turbine use together with other emotional and conditioning states. Many studies into the use of erbium lasers in restorative dentistry include reports of such treatment being less painful;55,56,57,58,59,60,61 Keller and Hibst investigated 103 patients with 206 preparations distributed amongst 194 teeth. Only 6% requested local anaesthesia during laser application. Eighty percent of the patients rated laser treatment more comfortable than bur preparation and 82% of the patients indicated that they would prefer the Er:YAG laser preparation for further caries treatment.62 Similar results were obtained by Matsumoto et al., reporting a 6% request for anaesthesia, although pain was reported in 32% of cavities prepared.63 Chaiyavej et al. found that Er:YAG, like bur cutting of tooth tissue, caused neural response in both A and C intradental fibres.64 Overall, the anecdotal reporting of prowess in delivering 'pain free' laser cavity preparation continues to provoke much debate – possibly to the detriment of the true capability of both Er:YAG and Er,Cr:YSGG in delivery of a genuine alternative to conventional rotary instrumentation.
'The aim of the wise is not to secure pleasure, but to avoid pain' (Aristotle). There remains the duty of the practitioner in showing professional responsibility towards the individual patient, their management and their comfort.
Erbium lasers and cavity preparation
i) Enamel
Enamel is composed, by volume, of 85% mineral (predominately carbonated hydroxyapatite), 12% water and 3% organic proteins. The majority of free water exists within the peri-prismatic protein matrix. Of the major hard tissues, enamel exhibits greatest resistance to laser ablation and this is seen most in healthy, fluoridated, occlusal sites, where ablation rate is approximately 20% of that achieved with a turbine. Fluoridated enamel presents a greater resistance, due to the combined effects of a harder fluorapatite (Ca10(PO4)6F2) mineral and the replacement of the hydroxyl group by fluoride (Fig. 27).
In Class III, IV and V cavity sites and certainly where prismatic density is less (as in deciduous teeth), the ablation rate is comparable to rotary instrumentation.65,66,67 Anecdotally, the speed of laser cutting is maximised if the incident beam is directed parallel to the prismatic structure and this is supportive of addressing the wavelength to the prime chromophore and its location within the tissue structure. In early research into the use of Er:YAG and enamel, it was shown that laser power parameters of approximately 350 mJ/2-4 pps (average power 0.7-1.4 W) would initiate enamel ablation in human teeth.68 With the development of better co-axial coolant and shorter pulses, fast and efficient cavity preparation can be achieved with power levels of 400-700 mJ/10-20 pps (average power range 4-8 W) which, with adequate water cooling, does not cause pulpal damage. Clinical experience would suggest that with 'harder' occlusal enamel, the use of higher energy-per-pulse and lower repetition rates provides for easier ablation. Where an etch-bonding technique is required, lower power levels (350-500 mJ/5-10 pps – average power 1.75-3.5 W) should be employed. The concept of average power is more important in those lasers where the pulse rate is fixed.
ii) Dentine
Dentine has a higher water content and less mineral density than enamel, being 47% by volume mineral (carbonated hydroxyapatite), 33% protein (mostly collagen) and 20% water. Consequently, ablation rates are faster than for enamel and power parameters can be correspondingly lower (average power range 3-5 W) and it is essential to use co-axial water spray (Fig. 28).
The sound resonance is greater, reflecting the greater water content. With carious dentine there is a potential in gross caries for the laser beam to quickly pass through the surface layer, thus leading to dehydration in deeper layers. Where gross caries is present it is advisable to use an excavator to remove bulk volume, both to prevent heat damage and to expedite cavity preparation. Both erbium lasers will leave a cut surface without a smear layer and it is advisable to use a dentine protector on open tubules exposed by the ablation process (Figs 29,30,31).
Erbium lasers and bone ablation
Early study into the effect of the Er:YAG laser on bone showed that, as with enamel and dentine ablation, tissue cutting is a thermally induced explosive process.69,70 As with other hard tissue interaction, it is essential to maintain a co-axial water spray to prevent heat damage which would delay healing. Studies into the rate of thermal denaturation of collagen, a major component of bone tissue, show that above a critical temperature (74°C), the rate of collagen denaturation rapidly increases causing coagulation of tissue. Other forms of thermal damage and tissue necrosis follow similar kinetics.71,72 At temperatures above 100-300°C there is an ascending dehydration, followed by carbonisation of proteins and lipids. The poor haemostatic effect of current mid-infrared lasers with adjunctive water spray, can be used to advantage in the ablation of bone, in ensuring blood perfusion of the surgical site (Figs 32 and 33).
However, the ablation process results in a considerable splatter of blood, and precautions (eye protection and mask) are recommended. An additional risk may be the creation of an air embolism in the tissue due to the air-induced water spray, although a review of the literature has not revealed any association. The acoustic level of bone ablation (100-120 dB) is much higher than with tooth tissue ablation.73
The use of erbium lasers in dento-alveolar surgery represents a less traumatic experience for the patient, when compared to the intense vibration of the slow-speed surgical bur. Ablation threshold measurements of 10-30 J cm−2 have been recorded for bone of varying density74 and clinically, with maxillary alveolar bone, the speed of laser cutting is comparable with that of a bur and is slightly slower in the mandible, reflecting the greater cortical bone composition. It is considered important that excessive power parameters are avoided, to reduce the 'stall-out' effect of debris and minimise blood spatter. Laser power values of 350-500 mJ/10-20 pps (average power range 3.5-7.0 W) with maximal water spray appear to effect good ablation rates.
The micro-analysis of the cut surface (Figs 34,35,36) reveals little evidence of thermal damage and any char layer appears to be restricted to a minimal zone of 20-30 μm in depth.74,75 Studies into the healing of lased bone would support the contention that the reduced physical trauma, reduced heating effects and reduced bacterial contamination, together with some claims to an osteogenic potential, lead to uncomplicated healing processes when compared to conventional use of a surgical bur.76,77,78,79,80
Conclusion
It is unquestioned that the patient avoidance of restorative dentistry is based upon the perceived association of such procedures with pain. Local anaesthetic drugs and techniques can address the demands on delivery of care, but the emergence of many innovative mechanical and chemical modalities for tooth cavity preparation and caries control during the last 15 years bears testimony to a desire to find a clinically acceptable replacement to the dental turbine. This is not just a cosmetic exercise, as the risks of pulpal temperature rise and iatrogenic healthy tooth tissue damage, together with the sensory effects of sound and vibration associated with the dental turbine, have been investigated and acknowledged for many years. With this in mind, it is of little comfort that the rates of tissue ablation with rotary instrumentation remain faster than the alternatives. The wide application of current commercially-available laser wavelengths that have been shown to be safe within correct power parameters, endorses their incorporation into the armamentarium of the restorative and surgical dentist. With the rapidly-growing concept of early intervention of caries, together with the general move away from direct metal restorative material and an embracing of 'non-classical' micro-retentive tooth cavities, there is a strong argument that laser-assisted cavity preparation, caries control and bonding techniques will find growing acceptance.
References
Birardi V, Bossi L, Dinoi C . Use of the Nd:YAG laser in the treatment of early childhood caries. Eur J Paediatr Dent 2004; 5: 98–101.
Bassi G, Chawla S, Patel M . The Nd:YAG laser in caries removal. Br Dent J 1994; 177: 248–250.
Harris D M, White J M, Goodis H et al. Selective ablation of surface enamel caries with a pulsed Nd:YAG dental laser. Lasers Surg Med 2002; 30: 342–350.
Cox C J, Pearson G J, Palmer G . Preliminary in vitro investigation of the effects of pulsed Nd:YAG laser radiation on enamel and dentine. Biomaterials 1994; 15: 1145–1151.
Yamada M K, Uo M, Ohkawa S, Akasaka T, Watari F . Three-dimensional topographic scanning electron microscope and Raman spectroscopic analyses of the irradiation effect on teeth by Nd:YAG, Er:YAG, and CO2 lasers. J Biomed Mater Res B Appl Biomater 2004; 71: 7–15.
Yamada M K, Watari F . Imaging and non-contact profile analysis of Nd:YAG laser-irradiated teeth by scanning electron microscopy and confocal laser scanning microscopy. Dent Mater J 2003; 22: 556–568.
Srimaneepong V, Palamara J E, Wilson P R . Pulpal space pressure and temperature changes from Nd:YAG laser irradiation of dentin. J Dent 2002; 30: 291–296.
Lan W H, Chen K W, Jeng J H, Lin C P, Lin S K . A comparison of the morphological changes after Nd-YAG and CO2 laser irradiation of dentin surfaces. J Endod 2000; 26: 450–453.
McDonald A, Claffey N, Pearson G, Blau W, Setchell D . The effect of Nd:YAG pulse duration on dentine crater depth. J Dent 2001; 29: 43–53.
Goodis H E, White J M, Marshall G W Jr et al. Effects of Nd: and Ho:yttrium-aluminium-garnet lasers on human dentine fluid flow and dental pulp-chamber temperature in vitro. Arch Oral Biol 1997; 42: 845–854.
Seka W, Fried D, Featherstone J D, Borzillary S F . Light deposition in dental hard tissue and simulated thermal response. J Dent Res 1995; 74: 1086–1092.
Kwon Y H, Kwon O W, Kim H I, Kim K H . Nd:YAG laser ablation and acid resistance of enamel. Dent Mater J 2003 22: 404–411.
Tsai C L, Lin Y T, Huang S T, Chang H W . In vitro acid resistance of CO2 and Nd-YAG laser-treated human tooth enamel. Caries Res 2002; 36: 423–429.
Harazaki M, Hayakawa K, Fukui T, Isshiki Y, Powell L G . The Nd-YAG laser is useful in prevention of dental caries during orthodontic treatment. Bull Tokyo Dent Coll 2001; 42: 79–86.
Hossain M, Nakamura Y, Kimura Y, Yamada Y, Kawanaka T, Matsumoto K . Effect of pulsed Nd:YAG laser irradiation on acid demineralization of enamel and dentin. J Clin Laser Med Surg 2001; 19: 105–108.
Myaki S I, Watanabe I S, Eduardo C de P, Issao M . Nd:YAG laser effects on the occlusal surface of premolars. Am J Dent 1998; 11: 103–105.
Malmstrom H S, McCormack S M, Fried D, Featherstone J D . Effect of CO2 laser on pulpal temperature and surface morphology: an in vitro study. J Dent 2001; 29: 521–529.
Watanabe I, Lopes R A, Brugnera A, Katayama A Y, Gardini A E . Effect of CO2 laser on class V cavities of human molar teeth under a scanning electron microscope. Braz Dent J 1996; 7: 27–31.
Friedman S, Liu M, Izawa T, Moynihan M, Dorscher-Kim J, Kim S . Effects of CO2 laser irradiation on pulpal blood flow. Proc Finn Dent Soc 1992; 88 (Suppl 1): 167–171.
Konishi N, Fried D, Staninec M, Featherstone J D . Artificial caries removal and inhibition of artificial secondary caries by pulsed CO2 laser irradiation. Am J Dent 1999; 12: 213–216.
Mullejans R, Eyrich G, Raab W H, Frentzen M . Cavity preparation using a superpulsed 9.6-microm CO2 laser – a histological investigation. Lasers Surg Med 2002; 30: 331–336.
Featherstone J D, Barrett-Vespone N A, Fried D, Kantorowitz Z, Seka W . CO2 laser inhibitor of artificial caries-like lesion progression in dental enamel. J Dent Res 1998; 77: 1397–1403.
Kantorowitz Z, Featherstone J D, Fried D . Caries prevention by CO2 laser treatment: dependency on the number of pulses used. J Am Dent Assoc 1998; 129: 585–591.
Goodis H E, Fried D, Gansky S, Rechmann P, Featherstone J D . Pulpal safety of 9.6 microm TEA CO2 laser used for caries prevention. Lasers Surg Med 2004; 35: 104–110.
McCormack S M, Fried D, Featherstone J D, Glena R E, Seka W . Scanning electron microscope observations of CO2 laser effects on dental enamel. J Dent Res 1995; 74: 1702–1708.
Tepper S A, Zehnder M, Pajarola G F, Schmidlin P R . Increased fluoride uptake and acid resistance by CO2 laser-irradiation through topically applied fluoride on human enamel in vitro. J Dent 2004; 32: 635–641.
Takamori K, Furukawa H, Morikawa Y, Katayama T, Watanabe S . Basic study on vibrations during tooth preparations caused by high-speed drilling and Er:YAG laser irradiation. Lasers Surg Med 2003; 32: 25–31.
Glockner K, Rumpler J, Ebeleseder K, Stadtler P . Intrapulpal temperature during preparation with the Er:YAG laser compared to the conventional burr: an in vitro study. J Clin Laser Med Surg 1998; 16: 153–157.
Pelagalli J, Gimbel C B, Hansen R T, Swett A, Winn DW 2nd . Investigational study of the use of Er:YAG laser versus dental drill for caries removal and cavity preparation – phase I. J Clin Laser Med Surg 1997; 15: 109–115.
Clark J, Symons A L, Diklic S, Walsh L J . Effectiveness of diagnosing residual caries with various methods during cavity preparation using conventional methods, chemo-mechanical caries removal, and Er:YAG laser. Aust Dent J 2001; 46 (Suppl): S20.
Lee B S, Lin C P, Hung Y L, Lan W H . Structural changes of Er:YAG laser irradiated human dentin. Photomed Laser Surg 2004; 22: 330–334.
Murray A K, Dickinson M R . High-speed photography of plasma during excimer laser-tissue interaction. Phys Med Biol 2004; 49: 3325–3340.
Fried D. IR laser ablation of dental enamel. Proc SPIE 2000; 3910: 136–148.
Freiberg R J, Cozean C D . Pulsed erbium laser ablation of hard dental tissue: the effects of atomized water spray versus water surface film. Proc SPIE 2002; 4610: 74–84.
Riziou I, Kimmel A. Atomized fluid particles for electromagnetically induced cutting. US Patent 5,741,247. 1998.
Walsh J T Jr, Cummings J P. Effect of the dynamic optical properties of water on midinfrared laser ablation. Lasers Surg Med 1994; 15: 295–305.
Apel C, Meister J, Ioana R S, Franzen R, Hering P, Gutknecht N . The ablation threshold of Er:YAG and Er:YSGG laser radiation in dental enamel. Lasers Med Sci 2002; 17: 246–252.
Harashima T, Kinoshita J, Kimura Y et al. Morphological comparative study on ablation of dental hard tissues at cavity preparation by Er:YAG and Er,Cr:YSGG lasers. Photomed Laser Surg 2005; 23: 52–55.
Boyde A. Enamel structure and cavity margins. Oper Dent 1976; 1: 13–28.
Chinelatti M A, Ramos R P, Chimello D T, Borsatto M C, Pecora J D, Palma-Dibb R G. Influence of the use of Er:YAG laser for cavity preparation and surface treatment in microleakage of resin-modified glass ionomer restorations. Oper Dent 2004; 29: 430–436.
Corona S A, Borsatto M C, Pecora JD, De SA Rocha R A, Ramos T S, Palma-Dibb R G . Assessing microleakage of different class V restorations after Er:YAG laser and bur preparation. J Oral Rehabil 2003; 30: 1008–1014.
Corona S A, Borsatto M, Dibb R G, Ramos R P, Brugnera A, Pecora J D. Microleakage of class V resin composite restorations after bur, air-abrasion or Er:YAG laser preparation. Oper Dent 2001; 26: 491–497.
Kohara E K, Hossain M, Kimura Y, Matsumoto K, Inoue M, Sasa R . Morphological and microleakage studies of the cavities prepared by Er:YAG laser irradiation in primary teeth. J Clin Laser Med Surg 2002; 20: 141–147.
Niu W, Eto J N, Kimura Y, Takeda F H, Matsumoto K . A study on microleakage after resin filling of class V cavities prepared by Er:YAG laser. J Clin Laser Med Surg 1998; 16: 227–231.
Gutknecht N, Apel C, Schafer C, Lampert F . Microleakage of composite fillings in Er,Cr:YSGG laser-prepared class II cavities. Lasers Surg Med 2001; 28: 371–374.
Malamed S F. Pain and anxiety control in dentistry. J Calif Dent Assoc 1993; 21: 35–38, 40–41.
Penfold C N. Pain-free oral surgery. Dent Update 1993; 20: 421–426.
Maskell R. Pain-free dental treatment is changing dentistry's image. Probe (Lond) 1991; 33(9): 36–37.
Arora R. Influence of pain-free dentistry and convenience of dental office on the choice of a dental practitioner: an experimental investigation. Health Mark Q 1999; 16(3): 43–54.
Blechman A M. Pain-free and mobility-free orthodontics? Am J Orthod Dentofacial Orthop 1998; 113: 379–383.
Delfino J. Public attitudes toward oral surgery: results of a Gallup poll. J Oral Maxillofac Surg 1997; 55: 564–567.
Whitters C J, Hall A, Creanor S L et al. A clinical study of pulsed Nd: YAG laser-induced pulpal analgesia. J Dent 1995; 23: 145–150.
Orchardson R, Whitters C J . Effect of HeNe and pulsed Nd:YAG laser irradiation on intradental nerve responses to mechanical stimulation of dentine. Lasers Surg Med 2000; 26: 241–249.
Orchardson R, Peacock J M, Whitters C J . Effect of pulsed Nd:YAG laser radiation on action potential conduction in isolated mammalian spinal nerves. Lasers Surg Med 1997; 21: 142–148.
Hubbard L G. Smile improvement: the laser way. Dent Today 2000; 19(2): 94–95.
Smith T A, Thompson J A, Lee W E . Assessing patient pain during dental laser treatment. J Am Dent Assoc 1993; 124: 90–95.
Kato J, Moriya K, Jayawardena J A, Wijeyeweera R L . Clinical application of Er:YAG laser for cavity preparation in children. J Clin Laser Med Surg 2003; 21: 151–155.
Hadley J, Young D A, Eversole L R, Gornbein J A . A laser-powered hydrokinetic system for caries removal and cavity preparation. J Am Dent Assoc 2000; 131: 777–785.
Dostalova T, Jelinkova H, Kucerova H et al. Noncontact Er:YAG laser ablation: clinical evaluation. J Clin Laser Med Surg 1998; 16: 273–282.
Matsumoto K, Nakamura Y, Mazeki K, Kimura Y . Clinical dental applHication of Er:YAG laser for class V cavity preparation. J Clin Laser Med Surg 1996; 14: 123–127.
Keller U, Hibst R . Effects of Er:YAG laser in caries treatment: a clinical pilot study. Lasers Surg Med 1997; 20: 32–38.
Keller U, Hibst R, Geurtsen W et al. Erbium:YAG laser application in caries therapy. Evaluation of patient perception and acceptance. J Dent 1998; 26: 649–656.
Matsumoto K, Hossain M, Hossain M M, Kawano H, Kimura Y . Clinical assessment of Er,Cr:YSGG laser application for cavity preparation. J Clin Laser Med Surg 2002; 20: 17–21.
Chaiyavej S, Yamamoto H, Takeda A, Suda H . Response of feline intradental nerve fibers to tooth cutting by Er:YAG laser. Lasers Surg Med 2000; 27: 341–349.
Stock K, Hibst R, Keller U . Comparison of Er:YAG and Er:YSGG laser ablation of dental hard tissues. Proc SPIE 2000; 3192: 88–95.
Belikov A V, Erofeev A V, Shumilin V V, Tkachuk A M. Comparative study of the 3um laser action on different hard tissue samples using free running pulsed Er-doped YAG, YSGG, YAP and YLF lasers. Proc SPIE 1993; 2080: 60–67.
Mercer C, Anderson P, Davis G . Sequential 3D X-ray microtomographic measurement of enamel and dentine ablation by an Er:YAG laser. Br Dent J 2003; 194: 99–104.
Hibst R, Keller U . Mechanism of Er:YAG laser-induced ablation of dental hard substances. Proc SPIE 1993; 1880: 156–162.
Hibst R. Mechanical effects of erbium:YAG laser bone ablation. Lasers Surg Med 1992; 12: 125–130.
Peavy G M, Reinisch L, Payne J T, Venugopalan V . Comparison of cortical bone ablations by using infrared laser wavelengths 2.9 to 9.2 microm. Lasers Surg Med 1999; 25: 421–434.
Thomsen S. Pathologic analysis of photothermal and photomechanical effects of laser-tissue interactions. Photochem Photobiol 1991; 53: 825–835.
Thomsen S, Cheong W, Pearce J . Changes in collagen birefringence: a quantitative histologic marker of thermal damage in skin. Proc SPIE 1992; 1422: 32–42.
Li Z Z, Reinisch L, Van de Merwe W P . Bone ablation with Er:YAG and CO2 laser: study of thermal and acoustic effects. Lasers Surg Med 1992; 12: 79–85.
Fried N M, Fried D . Comparison of Er:YAG and 9.6-microm TE CO2 lasers for ablation of skull tissue. Lasers Surg Med 2001; 28: 335–343.
Sasaki K M, Aoki A, Ichinose S, Ishikawa I . Ultrastructural analysis of bone tissue irradiated by Er:YAG laser. Lasers Surg Med 2002; 31: 322–332.
Wang X, Zhang C, Matsumoto K . In vivo study of the healing processes that occur in the jaws of rabbits following perforation by an Er,Cr:YSGG laser. Lasers Med Sci 2005; 20: 21–27.
Walsh J T Jr, Deutsch T F. Er:YAG laser ablation of tissue: measurement of ablation rates. Lasers Surg Med 1989; 9: 327–337.
Wang X, Ishizaki N T, Suzuki N, Kimura Y, Matsumoto K . Morphological changes of bovine mandibular bone irradiated by Er,Cr:YSGG laser: an in vitro study. J Clin Laser Med Surg 2002; 20: 245–250.
Pourzarandian A, Watanabe H, Aoki A et al. Histological and TEM examination of early stages of bone healing after Er:YAG laser irradiation. Photomed Laser Surg 2004; 22: 342–350.
O'Donnell R J, Deutsch T F, Flotte R J et al. Effect of Er:YAG laser holes on osteoinduction in demineralized rat calvarial allografts. J Orthop Res 1996; 14: 108–113.
Acknowledgements
The assistance provided by Professor J. Featherstone, UCSF, California, USA in the design of Figure 13 (absorption curve of CHA) is acknowledged.
Author information
Authors and Affiliations
Corresponding author
Additional information
Refereed paper
Rights and permissions
About this article
Cite this article
Parker, S. Surgical lasers and hard dental tissue. Br Dent J 202, 445–454 (2007). https://doi.org/10.1038/bdj.2007.294
Published:
Issue Date:
DOI: https://doi.org/10.1038/bdj.2007.294
This article is cited by
-
The impact of Er:YAG laser combined with fluoride treatment on the supragingival plaque microbiome in children with multiple caries: a dynamic study
BMC Oral Health (2022)
-
Time-resolved whole field investigation of plasma plume-induced shock wave in liquid media of different densities
Applied Physics B (2017)
-
Evaluation of micromorphological changes in tooth enamel after mechanical and ultrafast laser preparation of surface cavities
Lasers in Medical Science (2013)
-
Influence of etching with erbium, chromium:yttrium–scandium–gallium–garnet laser on microleakage of class V restoration
Lasers in Medical Science (2010)
-
Methodological quality of systematic reviews analyzing the use of laser therapy in restorative dentistry
Lasers in Medical Science (2010)