Abstract
The strain imposed by mechanical loading on bone tissue normally stimulates a response by bone cells that results in an adjustment of bone architecture that enables the bone to withstand reasonable loads. But it is unclear why this process should become less effective in some 50 per cent of postmenopausal women, who suffer fractures as a result1. Here we show that bone in vivo undergoes an adaptive response to loading that is less effective in the absence of the α-form of the oestrogen receptor (ER-α) and that osteoblast-like cells require ER-α to proliferate in response to mechanical strain in vitro. As ER-α expression in osteoblasts and osteocytes depends on oestrogen concentration2,3,4,5, a failure to maintain bone strength after the menopause might be due to a reduction in the activity of ER-α in bone cells, thereby limiting their anabolic response to mechanical loading and allowing a loss of bone tissue comparable to that associated with disuse.
Similar content being viewed by others
Main
We investigated bone's osteogenic response to loading in mice lacking functional ER-α. The normal peak locomotor strains (0.0026; length change as a proportion of original length) and strain rates (0.1 s−1) in the mouse ulna shaft were determined from surgically implanted strain gauges. We then loaded the ulnae of 20–24-week-old skeletally mature female mice through their olecranon and flexed carpus for three days a week for two weeks6. Each loading session comprised 40 repetitions of a 3.4-newton axial compressive load, which engendered peak strains (0.0028) and maximum strain rates (0.1 s−1) within the high physiological range.
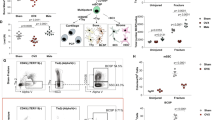
In mice with normal ER-α function (ERα+/+ mice), this loading regimen stimulates sufficient new bone formation on the periosteal and endosteal surfaces to increase the cortical area at the midshaft by 8 ± 0.8% (P < 0.001; Fig. 1a). In their ERα−/− littermates, however, this response was diminished threefold (2.4 ± 0.4%; P < 0.001; Fig. 1b, c).
a, b, Transverse sections from control (left) and loaded (right) ulnae from ERα+/+ (n = 7) (a) and ERα−/− (n = 8) (b) mice. New bone formation is labelled with fluorochrome calcein (shown in white), given on days 3 and 12 of the 2-week experiment. Scale bars, 100 μm. c, In ulnae from ERα−/− mice, the adaptive increase in cortical bone area in response to loading was only one-third of that in ulnae from ERα+/+ littermates. d, Mechanical strain stimulates proliferation in osteoblast-like cells derived from ERα+/+ mice but not from ERα−/− mice. Transfecting osteoblast-like cells derived from ERα−/− mice with competent human ER-α confers strain-induced proliferative responsiveness in comparison with non-strained controls (asterisk denotes P ≤ 0.05; in five experiments with three cultures per experiment, transfection efficiency was 20 ± 2% using 'Effectene' (Qiagen), as assessed by β-galactosidase expression in 1,057 cells).
A single period of dynamic strain applied to monolayer cultures of osteoblast-like cells stimulates their proliferation, a response that is inhibited by ER blockers and increased by transfection with additional ER-α (ref. 7). We derived separate primary cultures of osteoblast-like cells from the ulnae of ERα−/− and ERα+/+ littermates and exposed them to a single 10-min period of mechanical strain (600 cycles, 1 Hz, 0.0034). Over the next 24 h, the number of ERα+/+ cells increased by 58 ± 34%, P = 0.050, whereas the number of ERα−/− cells did not increase. ERα−/− mice have fully functional oestrogen receptors of the β-form, indicating that ER-β does not compensate for incompetent ER-α in this response. However, a proliferation response to strain was conferred on ERα−/− cells by transfecting them with a functional human wild-type ER-α expression vector (pRST7-ER; ref. 8) (Fig. 1d).
These results obtained in vivo and in vitro indicate that strain-related responses by differentiated cells of the osteoblast lineage require ER-α activity. This might explain why postmenopausal women no longer maintain adequate bone mass — their bone cells are less responsive to mechanical stimulation owing to decreased ER-α activity.
The oestrogen receptor is the ancestral steroid receptor9, with one of its possible early reproductive functions being to induce skeletal remodelling to release calcium for egg-laying or embryonic development. The strain-sensitive mechanisms that now enable mammalian and avian skeletons to adapt to load-bearing might have developed later, exploiting this receptor's ability to influence bone (re)modelling activity.
References
Riggs, B. L., Khosla, S. & Melton, L. J. Endocr. Rev. 23, 279–302 (2002).
Sutherland, M. K., Hui, D. U., Rao, L. G., Wylie, J. N. & Murray, T. M. Bone 18, 361–369 (1996).
Hoyland, J. A. et al. J. Pathol. 188, 294–303 (1999).
Lim, S. K., Won, Y. J., Lee, H. C., Huh, K. B. & Park, Y. S. J. Bone Miner. Res. 14, 1189–1196 (1999).
Ankrom, M. A. et al. Biochem. J. 333, 787–794 (1998).
Lee, K. C. L., Maxwell, A. & Lanyon, L. E. Bone 31, 407–412 (2002).
Zaman, G., Cheng, M. Z., Jessop, H. L., White, R. & Lanyon, L. E. Bone 27, 233–239 (2000).
Tzukerman, M. T. et al. Mol. Endocrinol. 8, 21–30 (1994).
Thornton, J. W. Proc. Natl Acad. Sci. USA 98, 5671–5676 (2001).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Lee, K., Jessop, H., Suswillo, R. et al. Bone adaptation requires oestrogen receptor-α. Nature 424, 389 (2003). https://doi.org/10.1038/424389a
Issue Date:
DOI: https://doi.org/10.1038/424389a
This article is cited by
-
Mind Gaps and Bone Snaps: Exploring the Connection Between Alzheimer’s Disease and Osteoporosis
Current Osteoporosis Reports (2024)
-
Antiosteoporosis effect and possible mechanisms of the ingredients of Radix Achyranthis Bidentatae in animal models of osteoporosis: systematic review and meta-analysis of in vivo studies
Journal of Orthopaedic Surgery and Research (2023)
-
The potential anti-osteoporotic effect of exercise—induced increased preptin level in ovariectomized rats
Anatomical Science International (2023)
-
Relationships between longitudinal changes in body composition and bone mineral density in middle-to-older aged Australians
Osteoporosis International (2023)
-
Metabolic activities affect femur and lumbar vertebrae remodeling, and anti-resorptive risedronate disturbs femoral cortical bone remodeling
Experimental & Molecular Medicine (2021)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.