Abstract
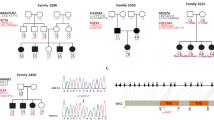
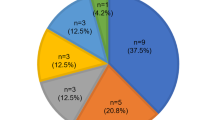
Severe deafness or hearing impairment is the most prevalent inherited sensory disorder, affecting about I in 1,000 children1. Most deafness results from peripheral auditory defects that occur as a consequence of either conductive (outer or middle ear) or sensorineuronal. (cochlea) abnormalities. Although a number of mutant genes have been identified that are responsible for syndromic (multiple phenotypic disease) deafness such as Waarden 2-4 burg syndrome and Usher 1B syndrome2–4, little is known about the genetic basis of non-syndromic (single phenotypic disease) deafness. Here we study a pedigree containing cases of autosomal dominant deafness and have identified a mutation in the gene encoding the gap-junction protein connexin 26 (Cx26) that segregates with the profound deafness in the family. Cx26 mutations resulting in premature stop codons were also found in three autosomal recessive non-syndromic sensorineuronal deafness pedigrees, genetically linked to chromosome 13q11-12 (DFNB1), where the Cx26 gene is localized. Immunohistochemical staining of human cochlear cells for Cx26 demonstrated high levels of expression. To our knowledge, this is the first nonsyndromic sensorineural autosomal deafness susceptibility gene to be identified, which implicates Cx26 as an important component of the human cochlea.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Morton, N. E. Genetic epidemiology of hearing impairment. Ann. N.Y. Acad. Sci. 630, 16–31 (1991).
Tassabehji, M. et al. Waardenburg's syndrome patients have mutations in the human homologue of the Pax-3 paired box gene. Nature 355, 635–636 (1992).
Tassabehji, M. et al. Mutations in the PAX3 gene causing Waardenburg syndrome type 1 and type 2. Nature Genet. 3, 26–30 (1993).
Weil, D. et al. Defective myosin VIIA gene responsible for Usher syndrome 1B. Nature 374, 60–61 (1995).
Verbov, J. Palmoplantar keratoderma, deafness and atopy. Br. J. Dermatol. 116, 881–882 (1987).
Fitzgerald, D. A. & Verbov, J. L. Hereditary palmoplantar keratoderma with deafness. Br. J. Dermatol. 134, 939–942 (1996).
Guilford, P. et al. A human gene responsible for neurosensory, non-syndromic recessive deafness is a candidate homologue of the mouse sh-1 gene. Hum. Mol. Genet. 3, 989–993 (1994).
Chaib, H. et al. A gene responsible for a dominant form of neurosensory non-syndromic deafness maps to NSRD1 recessive deafness gene interval. Hum. Mol. Genet. 3, 2219–2222 (1994).
Kibar, Z. et al. The gene responsible for Clouston hidrotic ectodermal dysplasia maps to the pericentromeric region of chromosome 13q. Hum. Mol. Genet. 5, 543–547 (1996).
Mignon, C. et al. Assignment of connexin 26 (GJB2) and 46 (GJA3) genes to human chromosome 13q11–q12. Cytogenet. Cell Genet. 72, 185–186 (1996).
Kumar, N. M. & Gilula, N. B. The gap junction communication channel. Cell 84, 381–388(1996).
Beyer, E. C. Gap junction. Int. Rev Cytol. 137, 231–242 (1993).
Kumar, N. M. & Gilula, N. B. Molecular biology and genetics of gap junction channels . Semin. Cell Biol. 3, 3–16 (1992).
Risek, B., Klier, F. G. & Gilula, N. B. Multiple gap junction genes are utilized during rat skin and hair development. Development 116, 639–651 (1992).
Goliger, J. A. & Paul, D. L. Expression of gap junction proteins Cx26, Cx31.1, Cx37, and Cx43 in developing and mature epidermis. Dev. Dyn. 200, 1–13 (1994).
Yeager, M. Nicholson, B. J. Structure of gap junction intercellular channels. Curr. Opin. Struct. Biol. 6, 183–192 (1996).
Markey, A. C., Lane, E. B., MacDonald, D. M. & Leigh, I. M. Keratin expression in basal cell carcinomas. Br J. Dermatol. 126, 154–160 (1992).
Kikuchu, T. Kimar, R. S. Paul, D. L. & Adams, J. C. Gap junctions in the rat cochlea: immunohistochemical and ultrastructural analysis. Anat. Embryol. 191, 101–118 (1995).
Brown, K. A. et al. Linkage studies in non-syndromic recessive deafness (NSRD) in a family originating from the Mirpur region of Pakistan maps DFNB I centromeric to D13S175. Hum. W. Genet. 5, 169–173 (1996).
Petit, C. Genes responsible for human hereditary deafness: a symphony of a thousand. Nature Genet. 14 385–391 (1996).
Bergoffen, J. et al. Connexin mutations in X-linked Charcot-Marie-Tooth Disease. Science 262, 2039–2041 (1993).
Britz-Cunningham, S. H., Shah, M. M., Zuppan, C. W. & Fletcher, W. H/Mutations of the connexin-43 gap function gene in patients with heart malformations and defects of laterality. N. Engl. J. Med. 332, 1323–1329 (1995).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Kelsell, D., Dunlop, J., Stevens, H. et al. Connexin 26 mutations in hereditary non-syndromic sensorineural deafness. Nature 387, 80–83 (1997). https://doi.org/10.1038/387080a0
Issue Date:
DOI: https://doi.org/10.1038/387080a0
This article is cited by
-
The congenital hearing phenotype in GJB2 in Queensland, Australia: V37I and mild hearing loss predominates
European Journal of Human Genetics (2024)
-
A novel method for detecting nine hotspot mutations of deafness genes in one tube
Scientific Reports (2024)
-
Burden of Mendelian disorders in a large Middle Eastern biobank
Genome Medicine (2024)
-
A Descriptive Observational Study of GJB2 and GJB6 Mutations in Familial Autosomal Recessive Non-syndromic Hearing Impairment
Indian Journal of Otolaryngology and Head & Neck Surgery (2023)
-
Biallelic mutations in pakistani families with autosomal recessive prelingual nonsyndromic hearing loss
Genes & Genomics (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.