Key Points
-
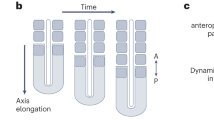
During somitogenesis, presomitic mesoderm (PSM) can be divided into at least two distinct regions: region I (posterior PSM) and region II (anterior PSM), which correspond to the two distinct cellular states, state I and state II.
-
No signs of segment specification can be detected at a molecular or cellular level in region I, but PSM cells acquire rostrocaudal polarity and become competent to segment once they have reached region II.
-
Cyclical gene expression that reflects an underlying segmentation clock is translated into Notch activity that keeps the oscillations of neighbouring PSM cells synchronized in region I.
-
Expression of delta-like 1 (Dll1) in region II, which is regulated by mesoderm posterior 2 (Mesp2) through the Notch signalling pathway, determines the rostrocaudal polarity of somites.
-
Notch signalling can be either presenilin 1 (Psen1) dependent or independent; the former is involved in inducing Dll1 expression and the latter in inhibiting Dll1 expression. Mesp2 might stimulate the inhibitory pathway and suppress the induction pathway.
-
The transition from state I to state II is controlled by the level of fibroblast growth factor (Fgf) signalling. A high level of Fgf activation in the posterior PSM maintains PSM cells in an immature state, whereas low Fgf levels accelerate the maturation process of PSM cells in the anterior PSM.
-
Because the Fgf activation domain retreats as somitogenesis proceeds, the wavefront, which is the interphase between region I and region II, gradually moves back. So, periodic interactions between the wavefront and the oscillation wave create a regularly spaced somite border.
Abstract
The reiterated structures of the vertebrate axial skeleton, spinal nervous system and body muscle are based on the metameric structure of somites, which are formed in a dynamic morphogenetic process. Somite segmentation requires the activity of a biochemical oscillator known as the somite-segmentation clock. Although the molecular identity of the clock remains unknown, genetic and experimental evidence has accumulated that indicates how the periodicity of somite formation is generated, how the positions of segment borders are determined, and how the rostrocaudal polarity within somite primordia is generated.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$189.00 per year
only $15.75 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Gossler, A. & Hrabe de Angelis, M. Somitogenesis. Curr. Top. Dev. Biol. 38, 225–287 (1998).An excellent overview of vertebrate somitogenesis.
Palmeirim, I., Henrique, D., Ish-Horowicz, D. & Pourquie, O. Avian hairy gene expression identifies a molecular clock linked to vertebrate segmentation and somitogenesis. Cell 91, 639–648 (1997).This article provided the first molecular evidence that presomitic mesoderm cells show oscillatory behaviour by the expression of c-hairy1.
Artavanis-Tsakonas, S., Rand, M. D. & Lake, R. J. Notch signaling: cell fate control and signal integration in development. Science 284, 770–776 (1999).
Joyner, A. L. & Martin, G. R. En-1 and En-2, two mouse genes with sequence homology to the Drosophila engrailed gene: expression during embryogenesis. Genes Dev. 1, 29–38 (1987).
Marti, E., Takada, R., Bumcrot, D. A., Sasaki, H. & McMahon, A. P. Distribution of Sonic hedgehog peptides in the developing chick and mouse embryo. Development 121, 2537–2547 (1995).
Gavin, B. J., McMahon, J. A. & McMahon, A. P. Expression of multiple novel Wnt-1/int-1-related genes during fetal and adult mouse development. Genes Dev. 4, 2319–2332 (1990).
Christ, B. & Ordahl, C. P. Early stages of chick somite development. Anat. Embryol. 191, 381–396 (1995).
Aoyama, H. & Asamoto, K. Determination of somite cells: independence of cell differentiation and morphogenesis. Development 104, 15–28 (1988).
Stern, C. D. & Keynes, R. J. Interactions between somite cells: the formation and maintenance of segment boundaries in the chick embryo. Development 99, 261–272 (1987).
Conlon, R. A., Reaume, A. G. & Rossant, J. Notch1 is required for the coordinate segmentation of somites. Development 121, 1533–1545 (1995).
Hrabe de Angelis, M., McIntyre, J. & Gossler, A. Maintenance of somite borders in mice requires the Delta homologue DII1. Nature 386, 717–721 (1997).
Kusumi, K. et al. The mouse pudgy mutation disrupts Delta homologue Dll3 and initiation of early somite boundaries. Nature Genet. 19, 274–278 (1998).
Wong, P. C. et al. Presenilin 1 is required for Notch1 and DII1 expression in the paraxial mesoderm. Nature 387, 288–292 (1997).
Zhang, N. & Gridley, T. Defects in somite formation in lunatic fringe-deficient mice. Nature 394, 374–377 (1998).
Evrard, Y. A., Lun, Y., Aulehla, A., Gan, L. & Johnson, R. L. Lunatic fringe is an essential mediator of somite segmentation and patterning. Nature 394, 377–381 (1998).
Oka, C. et al. Disruption of the mouse RBP-Jκ gene results in early embryonic death. Development 121, 3291–3301 (1995).
Saga, Y., Hata, N., Koseki, H. & Taketo, M. M. Mesp2: a novel mouse gene expressed in the presegmented mesoderm and essential for segmentation initiation. Genes Dev. 11, 1827–1839 (1997).Shows that the transcription factor Mesp2 is involved in establishing the rostrocaudal polarity of somites.
Van Eeden, F. J. et al. Mutations affecting somite formation and patterning in the zebrafish, Danio rerio. Development 123, 153–164 (1996).
Haddon, C., Jiang, Y. J., Smithers, L. & Lewis, J. Delta–Notch signalling and the patterning of sensory cell differentiation in the zebrafish ear: evidence from the mind bomb mutant. Development 125, 4637–4644 (1998).
Jiang, Y. J. et al. Notch signalling and the synchronization of the somite segmentation clock. Nature 408, 475–479 (2000).Proposes that the function of Notch signalling is to keep the oscillations of neighbouring PSM cells synchronized.
Holley, S. A., Geisler, R. & Nusslein-Volhard, C. Control of her1 expression during zebrafish somitogenesis by a Delta-dependent oscillator and an independent wave-front activity. Genes Dev. 14, 1678–1690 (2000).This report shows that aei is deltaD in zebrafish.
Bettenhausen, B., Hrabe de Angelis, M., Simon, D., Guenet, J. L. & Gossler, A. Transient and restricted expression during mouse embryogenesis of Dll1, a murine gene closely related to Drosophila Delta. Development 121, 2407–2418 (1995).
Takahashi, Y. et al. Mesp2 initiates somite segmentation through the Notch signalling pathway. Nature Genet. 25, 390–396 (2000).A genetic study that shows how Mesp2 works in the Notch signalling pathway.
Aulehla, A. & Johnson, R. L. Dynamic expression of lunatic fringe suggests a link between notch signaling and an autonomous cellular oscillator driving somite segmentation. Dev. Biol. 207, 49–61 (1999).
Sawada, A. et al. Zebrafish Mesp family genes, mesp-a and mesp-b are segmentally expressed in the presomitic mesoderm, and Mesp-b confers the anterior identity to the developing somites. Development 127, 1691–1702 (2000).
Jen, W. C., Gawantka, V., Pollet, N., Niehrs, C. & Kintner, C. Periodic repression of Notch pathway genes governs the segmentation of Xenopus embryos. Genes Dev. 13, 1486–1499 (1999).
Yoon, J. K., Moon, R. T. & Wold, B. The bHLH class protein pMesogenin1 can specify paraxial mesoderm phenotypes. Dev. Biol. 222, 376–391 (2000).
Duband, J. L. et al. Adhesion molecules during somitogenesis in the avian embryo. J. Cell Biol. 104, 1361–1374 (1987).
Dubrulle, J., McGrew, M. J. & Pourquie, O. FGF signaling controls somite boundary position and regulates segmentation clock control of spatiotemporal Hox gene activation. Cell 106, 219–232 (2001).A crucial paper indicating that FGF functions to keep PSM cells in an immature state in the chick.
McGrew, M. J., Dale, J. K., Fraboulet, S. & Pourquie, O. The lunatic fringe gene is a target of the molecular clock linked to somite segmentation in avian embryos. Curr. Biol. 8, 979–982 (1998).
Jouve, C. et al. Notch signalling is required for cyclic expression of the hairy-like gene HES1 in the presomitic mesoderm. Development 127, 1421–1429 (2000).
Leimeister, C. et al. Oscillating expression of c-Hey2 in the presomitic mesoderm suggests that the segmentation clock may use combinatorial signaling through multiple interacting bHLH factors. Dev. Biol. 227, 91–103 (2000).
Kokubo, H., Lun, Y. & Johnson, R. L. Identification and expression of a novel family of bHLH cDNAs related to Drosophila hairy and enhancer of split. Biochem. Biophys. Res. Commun. 260, 459–465 (1999).
Schnell, S. & Maini, P. K. Clock and induction model for somitogenesis. Dev. Dyn. 217, 415–420 (2000).
Blair, S. S. Notch signaling: Fringe really is a glycosyltransferase. Curr. Biol. 10, R608–R612 (2000).
Aoyama, H. & Asamoto, K. The developmental fate of the rostral/caudal half of a somite for vertebra and rib formation: experimental confirmation of the resegmentation theory using chick-quail chimeras. Mech. Dev. 99, 71–82 (2000).
Mansouri, A., Voss, A. K., Thomas, T., Yokota, Y. & Gruss, P. Uncx4.1 is required for the formation of the pedicles and proximal ribs and acts upstream of Pax9. Development 127, 2251–2258 (2000).
Leitges, M., Neidhardt, L., Haenig, B., Herrmann, B. G. & Kispert, A. The paired homeobox gene Uncx4.1 specifies pedicles, transverse processes and proximal ribs of the vertebral column. Development 127, 2259–2267 (2000).
Barrantes, I. B. et al. Interaction between Notch signalling and Lunatic fringe during somite boundary formation in the mouse. Curr. Biol. 9, 470–480 (1999).
Koizumi, K. et al. The role of presenilin 1 during somite segmentation. Development 128, 1391–1402 (2001).
Kim, S. H., Jen, W. C., De Robertis, E. M. & Kintner, C. The protocadherin PAPC establishes segmental boundaries during somitogenesis in Xenopus embryos. Curr. Biol. 13, 821–830 (2000).
Sawada, A. et al. Fgf/MAPK signaling is a crucial positional cue in somite boundary formation. Development (in the press).Indicates that Fgf functions to keep PSM cells in an immature state in zebrafish.
Yamaguchi, T. P., Conlon, R. A. & Rossant, J. Expression of the fibroblast growth factor receptor FGFR-1/flg during gastrulation and segmentation in the mouse embryo. Dev. Biol. 152, 75–88 (1992).
Yamaguchi, T. P., Harpal, K., Henkemeyer, M. & Rossant, J. Fgfr-1 is required for embryonic growth and mesodermal patterning during mouse gastrulation. Genes Dev. 8, 3032–3044 (1994).
Sun, X., Meyers, E. N., Lewandoski, M. & Martin, G. R. Targeted disruption of Fgf8 causes failure of cell migration in the gastrulating mouse embryo. Genes Dev. 13, 1834–1846 (1999).
Reifers, F. et al. Fgf8 is mutated in acerebellar (ace) mutants and is required for maintenance of midbrain–hindbrain boundary development and somitogenesis. Development 125, 2381–2395 (1998).
Gotoh, Y. & Nishida, E. Signals for mesoderm induction. Roles of fibroblast growth factor (FGF)/mitogen-activated protein kinase (MAPK) pathway. Biochim. Biophys. Acta 1288, F1–F7 (1996).
Bagnall, K. M., Higgins, S. J. & Sanders, E. J. The contribution made by a single somite to the vertebral column: experimental evidence in support of resegmentation using the chick–quail chimaera model. Development 103, 69–85 (1988).The first demonstration of resegmenation in which half-somites are implicated.
Goldstein, R. S. & Kalcheim, C. Determination of epithelial half-somites in skeletal morphogenesis. Development 116, 441–445 (1992).
Stickney, H. L., Barresi, M. J. & Devoto, S. H. Somite development in zebrafish. Dev. Dyn. 219, 287–303 (2000).
Morin-Kensicki, E. M. & Eisen, J. S. Sclerotome development and peripheral nervous system segmentation in embryonic zebrafish. Development 124, 159–167 (1997).
Jen, W. C., Wettstein, D., Turner, D., Chitnis, A. & Kintner, C. The Notch ligand, X-Delta-2, mediates segmentation of the paraxial mesoderm in Xenopus embryos. Development 124, 1169–1178 (1997).
Cooke, J. & Zeeman, E. C. A clock and wavefront model for control of the number of repeated structures during animal morphogenesis. J. Theor. Biol. 58, 455–476 (1976).
Meinhardt, H. in Somites in Developing Embryos (eds Bellairs, R., Ede, D. A. & Lash, J. W.) 179–191 (Plenum Publ. Corpl., USA, 1986).
Primmett, D. R., Stern, C. D. & Keynes, R. J. Heat shock causes repeated segmental anomalies in the chick embryo. Development 104, 331–339 (1988).
Primmett, D. R., Norris, W. E., Carlson, G. J., Keynes, R. J. & Stern, C. D. Periodic segmental anomalies induced by heat shock in the chick embryo are associated with the cell cycle. Development 105, 119–130 (1989).
Collier, J. R. et al. A cell cycle model for somitogenesis: mathematical formulation and numerical simulation. J. Theor. Biol. 207, 305–316 (2000).
Flint, O. P., Ede, D. A., Wilby, O. K. & Proctor, J. Control of somite number in normal and amputated mutant mouse embryos: an experimental and a theoretical analysis. J. Embryol. Exp. Morphol. 45, 189–202 (1978).
Polezhaev, A. A. A mathematical model of the mechanism of vertebrate somitic segmentation. J. Theor. Biol. 156, 169–181 (1992).
Kerszberg, M. & Wolpert, L. A clock and trail model for somite formation, specialization and polarization. J. Theor. Biol. 205, 505–510 (2000).
Logeat, F. et al. The Notch1 receptor is cleaved constitutively by a furin-like convertase. Proc. Natl Acad. Sci. USA 95, 8108–8112 (1998).
Struhl, G. & Greenwald, I. Presenilin-mediated transmembrane cleavage is required for Notch signal transduction in Drosophila. Proc. Natl Acad. Sci. USA 98, 229–234 (2001).
Berezovska, O. et al. Rapid Notch1 nuclear translocation after ligand binding depends on presenilin-associated γ-secretase activity. Ann. NY Acad. Sci. 920, 223–226 (2000).
Schroeter, E. H., Kisslinger, J. A. & Kopan, R. Notch-1 signalling requires ligand-induced proteolytic release of intracellular domain. Nature 393, 382–386 (1998).
Qi, H. et al. Processing of the Notch ligand Delta by the metalloprotease Kuzbanian. Science 283, 91–94 (1999).
Bruckner, K., Perez, L., Clausen, H. & Cohen, S. Glycosyltransferase activity of Fringe modulates Notch–Delta interactions. Nature 406, 411–415 (2000).
Moloney, D. J. et al. Fringe is a glycosyltransferase that modifies Notch. Nature 406, 369–375 (2000).
Yoon, J. K. & Wold, B. The bHLH regulator pMesogenin1 is required for maturation and segmentation of paraxial mesoderm. Genes Dev. 14, 3204–3214 (2000).
Author information
Authors and Affiliations
Corresponding author
Related links
Related links
DATABASES
Glossary
- PARAXIAL MESODERM
-
A subpopulation of mesoderm that lies on both sides of the neural tube, which gives rise to somites.
- CEPHALOCHORDATE
-
A subphylum of chordates that includes the amphioxus Branchiostoma, which has a notochord, dorsal nervous system and segmented trunk, but lacks characters such as complex paired sensory organs and a true brain.
- PRESOMITIC MESODERM
-
Precursor unsegmented mesoderm, which generates somites on segmentation.
- CYCLING GENE
-
A gene, the expression of which oscillates rostrocaudally in the presomitic mesoderm.
- ROSTROCAUDAL POLARITY
-
The difference between the rostral (anterior) and caudal (posterior) halves of a somite that underlies a difference in future developmental fates.
- SEGMENT-POLARITY GENE
-
A gene originally identified in Drosophila early development, the expression of which divides the embryo into units that are 14 segments wide.
- SCLEROTOME
-
Mesenchymal cell mass located in the medial region of a somite, from which the axial skeleton derives
- PRIMITIVE STREAK
-
A longitudinal cleft formed on the surface of the amniote early embryo by a convergence of cells. At the onset of gastrulation, epiblast cells migrate towards and into the streak, and in so doing acquire mesodermal cell fate.
- TAILBUD
-
The caudal end of the tail region at which gastrulation continues to generate precursors for the paraxial mesoderm and the neural tube.
- EPITHELIAL SOMITE
-
Spherical epithelial structure made up of epithelial cells that differentiate from mesenchymal cells on segmentation.
- AMNIOTE
-
Animal, such as reptile, bird or mammal, whose eggs contain an amnion — a membrane that surrounds the embryo and helps retain fluids.
- SURFACE ECTODERM
-
The outermost germ layer of the embryo that develops during gastrulation. Also the cell layer that covers the paraxial mesoderm, from which several diffusible factors are secreted that induce somitic cells to take on the dermomyotome fate.
- DERMOMYOTOME
-
Epithelial cell layer in the dorsolateral region of the somite that faces the ectoderm and further differentiates into the most dorsal dermatome, which later differentiates into dermis and myotome — future skeletal muscles.
- NEURONAL HYPERPLASIA
-
Excessive formation of neuronal tissues due to transdifferentiation as a result of defects in Notch signalling.
- HOMOTYPIC INTERACTION
-
Protein interaction between molecules of the same type.
Rights and permissions
About this article
Cite this article
Saga, Y., Takeda, H. The making of the somite: molecular events in vertebrate segmentation. Nat Rev Genet 2, 835–845 (2001). https://doi.org/10.1038/35098552
Issue Date:
DOI: https://doi.org/10.1038/35098552
This article is cited by
-
Tetraparesis following thoracic spine surgery in a patient with Klippel–Feil syndrome and ABCB4 mutation: a case report
Journal of Medical Case Reports (2023)
-
Reconstituting human somitogenesis in vitro
Nature (2023)
-
Cyclical dermal micro-niche switching governs the morphological infradian rhythm of mouse zigzag hair
Nature Communications (2023)
-
Natterin-like depletion by CRISPR/Cas9 impairs zebrafish (Danio rerio) embryonic development
BMC Genomics (2022)
-
Periodic formation of epithelial somites from human pluripotent stem cells
Nature Communications (2022)