Abstract
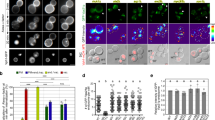
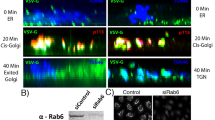
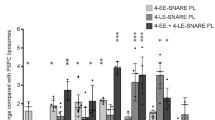
EUKARYOTIC cells seem to use GTP hydrolysis to regulate vesicular traffic in exocytosis and endocytosis. The best evidence for this comes from studies on the yeast Saccharomyces cerevisiae that have identified two small Ras-related GTP-binding proteins, Sec4p and Yptlp, which control distinct stages of the secretory pathway1–6. In mammalian cells the effects of a non-hydrolysable GTP analogue, GTP-γS, on different transport events have suggested that they also have proteins functionally related to yeast Sec4p and Yptlp7–10. The rab genes have recently been cloned and sequenced for rat and human and their proteins have highly conserved domains in common with Sec4p and Yptlp (including a putative effector binding site). They are therefore good candidates for GTP-binding proteins involved in intracellular transport in mammalian cells11,12. One of the Rab proteins (Rab1p) is the mammalian counterpart of Ypt1p (ref. 13). Here we report the localization of the protein Rab6p to the Golgi apparatus in several cell types. By immunolabelling and electron microscopy, Rab6p appears to be concentrated predominantly on the medial and trans cisternae and distributed over their entire surface.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Salminen, A. & Novick, P. Cell 49, 527–538 (1987).
Goud, B., Salminen, A., Walworth, N. & Novick, P. Cell 53, 753–768 (1988).
Walworth, N., Goud, B., Kabcenell, A. & Novick, P. EMBO J. 8, 1685–1693 (1989).
Schmitt, H. D., Wagner, P., Pfaff, E. & Gallwitz, D. Cell 47, 401–412 (1986).
Segev, N., Mulholland, J. & Botstein, D. Cell 52, 915–924 (1988).
Bacon, R. A., Salminen, A., Ruohola, H., Novick, P. & Ferro-Novick, S. J. Cell Biol. 109, 1015–1022 (1989).
Beckers, C. J. M. & Balch, W. E. J. Cell Biol. 108, 1245–1256 (1989).
Melancon, P. et al. Cell 51, 1053–1062 (1987).
Mayorga, L., Diaz, R. & Stahl, P. Science 244, 1475–1477 (1989).
Gomperts, B. Trends Biol. Sci. 11, 290–292 (1986).
Touchot, N., Chardin, P. & Tavitian, A. Proc. natn. Acad. Sci. U.S.A. 84, 8210–8214 (1987).
Zahraoui, A., Touchot, N., Chardin, P. & Tavitian, A. J. biol. Chem. 264, 12394–12401 (1989).
Haubruck, H., Prange, R., Vorgias, C. & Gallwitz, D. EMBO J. 8, 1427–1432 (1989).
Burgoyne, R. D. Trends Biol. Sci. 14, 394–396 (1989).
Barbacid, M. A. Rev. Biochem. 56, 779–827 (1987).
Molenaar, C. M. T., Prange, R. & Gallwitz, D. EMBO J. 7, 971–976 (1988).
Hancock, J. F., Magee, A. I., Childs, J. E. & Marshall, C. J. Cell 57, 1167–1177 (1989).
Jasmin, B. J., Cartaud, J., Bornens, M. & Changeux, J.-P. Proc. natn. Acad. Sci. U.S.A. 86, 7218–7222 (1989).
Rambourg, A., Clermont, Y. & Hermo, L. Am. J. Anat. 183, 187–199 (1988).
Saraste, J., Palade, G. E. & Farquhar, M. G. J. Cell Biol. 105, 2021–2029 (1987).
Bourne, H. Cell 53, 669–671 (1988).
Farquhar, M. G. & Palade, G. E. J. Cell Biol. 91, 77s–103s (1981).
Dunphy, W. G. & Rothman, J. E. Cell 42, 13–21 (1985).
Louvard, D., Reggio, H. & Warren, G. J. Cell Biol. 92, 92–107 (1982).
Bradford, M. M. Analyt. Biochem. 72, 248–254 (1976).
Laemmli, U. K. Nature 227, 680–685 (1970).
Burnette, W. N. Analyt. Biochem. 112, 195–201 (1981).
Talian, J. C., Olmsted, J. B. & Goldman, R. D. J. Cell Biol. 97, 1277–1282 (1983).
McLean, I. W. & Nakane, P. K. J. Histochem. Cytochem. 22, 1077–1083 (1974).
Brown, W. J. & Farquhar, M. G. in Methods in Cell Biology Vol. 31 (ed. Tartakoff, A. M.) 553–569 (Academic Press, San Diego, 1989).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Goud, B., Zahraoui, A., Tavitian, A. et al. Small GTP-binding protein associated with Golgi cisternae. Nature 345, 553–556 (1990). https://doi.org/10.1038/345553a0
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/345553a0
This article is cited by
-
Rab6 is required for rapid, cisternal-specific, intra-Golgi cargo transport
Scientific Reports (2020)
-
A GFP-tagged version of the pseudorabies virus protein UL56 localizes to the Golgi and trans-Golgi network through a predicted C-terminal leucine-rich helix in transfected cells
Virology Journal (2019)
-
Rab6 regulates cell migration and invasion by recruiting Cdc42 and modulating its activity
Cellular and Molecular Life Sciences (2019)
-
Coupling fission and exit of RAB6 vesicles at Golgi hotspots through kinesin-myosin interactions
Nature Communications (2017)
-
Comparative modeling of Rab6 proteins: identification of key residues and their interactions with guanine nucleotides
Journal of Molecular Modeling (2013)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.