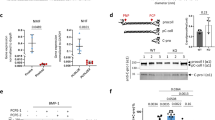
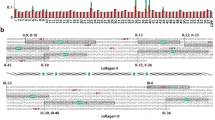
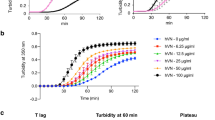
Abstract
Collagen fibres form the stable architecture of connective tissues and their breakdown is a key irreversible step in many pathological conditions1,2. The destruction of collagen is usually initiated by proteinases, the best known of which is the metalloproteinase collagenase (EC 3.4.24)3. Collagenase and related metalloproteinases are regulated at the level of their synthesis and secretion, through the action of specific stimuli such as hormones and cytokines, and also at the level of their extracellular activity through the action of a specific inhibitor, TIMP (tissue inhibitor of metalloproteinases)4–6, which irreversibly forms inactive complexes with metalloproteinases7. Although the mechanisms governing the production of TIMP are unknown, immunologically identical forms of this glycoprotein have been detected in a wide variety of human body fluids and cell and tissue culture media5,8. We therefore suggested5 that under physiological conditions this ubiquitous inhibitor predominates over active metalloproteinases and that tissue destruction may arise when any perturbation of this controlling excess arises. However, further progress towards testing this theory has been hindered by a lack of knowledge about the structure of TIMP and insufficient material for studying it in model systems. Here we describe the structure of TIMP predicted from its complementary DNA, its synthesis in Escherichia coli and transfected animal cells, and the finding that it is identical to a protein recently reported to have erythroid-potentiating activity (EPA)9.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Murphy, G. & Reynolds, J. J. Bioessays 2, 55–60 (1985).
Harris, E. D., Welgus, H. G. & Krane, S. M. Collagen Relat. Res. 4, 493–512 (1984).
Woolley, D. E. in Extracellular Matrix Biochemistry (eds Piez, K. A. & Reddi, A. H.) 119–157 (Elsevier, New York, 1984).
Sellers, A. & Murphy, G. Int. Rev. Connective Tissue Res. 9, 151–190 (1981).
Reynolds, J. J. et al. in Cellular Interactions (eds Dingle, J. T. & Gordon, J. L.) 205–213 (Elsevier-North-Holland, Amsterdam, 1981).
Bunning, R. A. D. et al. Eur. J. Biochem. 139, 75–80 (1984).
Cawston, T. E. et al. Biochem. J. 211, 313–318 (1983).
Welgus, H. G. & Stricklin, G. P. J. biol. Chem. 258, 12259–12264 (1983).
Gasson, J. C. et al. Nature 315, 768–771 (1985).
Murphy, G., Cawston, T. E. & Reynolds, J. J. J. Biochem. 195, 167–170 (1981).
Stricklin, G. P. & Welgus, H. G. J. biol. Chem. 258, 12252–12258 (1983).
Lathe, R. J. molec. Biol. 183, 1–12 (1985).
Grantham, R., Gautier, C., Couy, M., Jacobzone, M. & Mercier, R. Nucleic Acids Res. 9, r43–r47 (1981).
Nussenov, R. J. molec. Biol. 149, 125–131 (1981).
Watson, M. E. E. Nucleic Acids Res. 12, 5145–5153 (1984).
Murphy, G. & Werb, Z. Biochim. biophys. Acta 839, 214–218 (1985).
Kornfeld, S. in The Glycoconjugates Vol. 3A (ed. Horowitz, M. I.) 3–23 (Academic, New York, 1982).
Proudfoot, N. J. & Brownlee, G. G. Nature 252, 359–362 (1981).
Rovera, G., Santoli, D. & Damsky, C. Proc. natn. Acad. Sci. U.S.A. 76, 2779–2783 (1979).
Hembry, R. M., Murphy, G. & Reynolds, J. J. J. Cell Sci. 73, 105–119 (1985).
Harris, T. J. R. in Genetic Engineering Vol. 4 (ed. Williamson, R.) 128–175 (Academic, London, 1983).
Pavalatis, G. N. & Hamer, D. H. Proc. natn. Acad. Sci. U.S.A. 80, 397–401 (1983).
Kishi, J. & Hayakawa, T. J. Biochem., Tokyo 96, 395–404 (1984).
Murphy, G., Reynolds, J. J. & Werb, Z. J. biol. Chem. 260, 3079–3083 (1985).
Patel, T. P. et al. Nucleic Acids Res. 10, 5605–5619 (1982).
Maniatis, T., Fritsch, E. F. & Sambrook, J. Molecular Cloning, A Laboratory Manual (Cold Spring Harbor Laboratory, New York, 1982).
Aviv, H. & Leder, P. Proc. natn. Acad. Sci. U.S.A. 69, 1408–1412 (1972).
Gubler, U. & Hoffman, B. J. Gene 25, 263–269 (1983).
Michelson, A. M. & Orkin, S. H. J. biol. Chem. 257, 14773–14782 (1982).
Hanahan, D. J. molec. Biol. 166, 557–580 (1983).
Vieira, J. & Messing, J. Gene 19, 259–268 (1982).
Sanger, F., Nicklen, S. & Coulson, A. R. Proc. natn. Acad. Sci. U.S.A. 74, 5463–5467 (1977).
Messing, J. & Vieira, J. Gene 19, 269–276 (1982).
Laemmli, U. K. Nature 227, 680–685 (1970).
Burnette, W. N. Analyt. Biochem. 112, 195–203 (1981).
Lowy, D. R. et al. J. Virol. 26, 291–298 (1978).
Graham, F. L. & van der Eb, A. J. Virology 52, 456–461 (1983).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Docherty, A., Lyons, A., Smith, B. et al. Sequence of human tissue inhibitor of metalloproteinases and its identity to erythroid-potentiating activity. Nature 318, 66–69 (1985). https://doi.org/10.1038/318066a0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/318066a0
This article is cited by
-
The human brain NGF metabolic pathway is impaired in the pre-clinical and clinical continuum of Alzheimers disease
Molecular Psychiatry (2021)
-
Establishment of Structure-Function Relationship of Tissue Inhibitor of Metalloproteinase-1 for Its Interaction with CD63: Implication for Cancer Therapy
Scientific Reports (2020)
-
Impact of Elastin-Derived Peptide VGVAPG on Matrix Metalloprotease-2 and -9 and the Tissue Inhibitor of Metalloproteinase-1, -2, -3 and -4 mRNA Expression in Mouse Cortical Glial Cells In Vitro
Neurotoxicity Research (2019)
-
Functional disparities within the TIMP family in cancer: hints from molecular divergence
Cancer and Metastasis Reviews (2019)
-
TIMPs: versatile extracellular regulators in cancer
Nature Reviews Cancer (2017)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.