Abstract
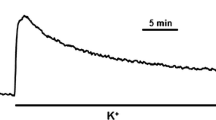
Microtubules are ubiquitous in eukaryotic cells and play key roles in many cellular activities. The purpose of this study was to investigate the influence of microtubules on vascular smooth muscle contraction. Quantitative immunocytochemical analysis of rat aortic tissue revealed that, relative to the control group, colchicine (15 μM, 90 min) and nocodazole (15 μM, 90 min) decreased the microtubule density by 40–50% while taxol (10 μM, 90 min) increased the microtubule density by 33%. Isometric contraction studies demonstrated that both colchicine and nocodazole caused an upward shift in the phenylephrine (10−8 to 10−5 M) dose–response curve while taxol caused no significant change when compared to the control group. Potassium chloride (30 mM) induced 55 ± 5% P 0 contraction in DMSO treated vessel rings. The active tension increased to 73 ± 5% P 0 and 71 ± 6% P 0 after pretreatment of the aortic rings with colchicine or nocodazole, respectively. Taxol did not cause a significant change in the active tension (56 ± 7% P 0). These results indicate that microtubule depolymerization enhances isometric contraction of vascular smooth muscle and this enhanced contraction is not receptor dependent. Pretreatment of the aortic rings with an inhibitor of nitric oxide synthase (NOS) (N ω-nitro-L-arginine) did not change the increased contractile response to phenylephrine due to microtubule depolymerization suggesting that this phenomenon is not mediated by endothelium dependent relaxation.
Similar content being viewed by others
References
Battistella-Patterson AS, Wang S and Wright GL (1997) Effect of disruption of the cytoskeleton on smooth muscle contraction. Can J Physiol Pharmacol 75(12): 1287–1299.
Bulinski JC (1994) MAP4. In: Hyams JS and Lloyd CW (eds) Microtubules. (pp. 167–182) Wiley, New York.
Driska S and Porter R (1986) Isolation of smooth muscle cells from swine carotid artery by digestion with papain. Am J Physiol 251(20): C474-C481.
Fay FS, Fujiwara K, Rees DD and Fogarty KE (1983) Distribution of alpha-actinin in single isolated smooth muscle cells. J Cell Biol 96(3): 783–795.
Fleming I and Busse R (1999) NO: the primary EDRF. J Mol Cell Cardiol 31(1): 5–14.
Gundersen GG and Cook TA (1999) Microtubules and signal transduction. Curr Opin Cell Biol 11(1): 81–94.
Harris DE and Warshaw DM (1990) Slowing of velocity during isotonic shortening in single isolated smooth muscle cells. Evidence for an internal load. J Gen Physiol 96(3): 581–601.
Horwitz SB, Parness J, Schiff PB and Manfredi JJ (1982) Taxol: a new probe for studying the structure and function of microtubules. Cold Spring Harb Symp Quant Biol 46(Pt 1): 219–226.
Ishibashi Y, Tsutsui H, Yamamoto S, Takahashi M, Imanaka-Yoshida M and Takeshita A (1996) Role of microtubules in myocyte contractile dysfunction during cardiac hypertrophy in the rat. Am J Physiol 271(5 Pt 2): H1978-H1987.
Knudsen HL and Frangos JA (1997) Role of cytoskeleton in shear stress-induced endothelial nitric oxide production. Am J Physiol 273(1 Pt 2): H347-H355.
Kolodney MS and Elson EL (1995) Contraction due to microtubule disruption is associated with increased phosphorylation of myosin regulatory light chain. Proc Natl Acad Sci USA 92(22): 10252–10256.
Leite R and Webb RC (1998) Microtubule disruption potentiates phenylephrine-induced vasoconstriction in rat mesenteric arterial bed. Eur J Pharmacol 351(1): R1-R3.
Liu BP, Chrzanowska-Wodnicka M and Burridge K (1998) Microtubule depolymerization induces stress fibers, focal adhesions, and DNA synthesis via the GTP-binding protein Rho. Cell Adhes Commun 5(4): 249–255.
Marczin N, Jilling T, Papapetropoulos A, Go C and Catravas JD (1996) Cytoskeleton-dependent activation of the inducible nitric oxide synthase in cultured aortic smooth muscle cells. Br J Pharmacol 118(5): 1085–1094.
Noda M, Yasuda-Fukazawa C, Moriishi K, Kato T, Okuda T, Kurokawa K and Takuwa Y (1995) Involvement of rho in GTP gamma S-induced enhancement of phosphorylation of 20 kDa myosin light chain in vascular smooth muscle cells: inhibition of phosphatase activity. FEBS Lett 367(3): 246–250.
Platts SH, Falcone JC, Holton WT, Hill MA and Meininger GA (1999) Alteration of microtubule polymerization modulates arteriolar vasomotor tone. Am J Physiol 277(1): H100-H106.
Ross R (1971) The smooth muscle cell. II. Growth of smooth muscle in culture and formation of elastic fibers. J Cell Biol 50(1): 172–186.
Sheridan BC, McIntyre Jr RC, Meldrum DR, Agrafojo J, Banerjee A, Harken AH and Fullerton DA (1996) Microtubules regulate pulmonary vascular smooth muscle contraction. J Surg Res 62(2): 284–287.
Swartz DR, Moss RL and Greaser ML (1997) Characteristics of troponin C binding to the myofibrillar thin filament: extraction of troponin C is not random along the length of the thin filament. Biophys J 73(1): 293–305.
Swartz DR, Moss RL and Greaser ML (1996) Calcium alone does not fully activate the thin filament for S1 binding to rigor myofibrils. Biophys J 71(4): 1891–1904.
Tagawa H, Koide M, Sato H, Zile MR, Carabello BA and Cooper G (1998) Cytoskeletal role in the transition from compensated to decompensated hypertrophy during adult canine left ventricular pressure overloading. Circ Res 82(7): 751–761.
Tsutsui H, Ishihara K and Cooper G (1993) Cytoskeleton role in the contractile dysfunction of hypertropied myocardium. Science 260(5108): 682–687.
Wang N, Butler JP and Ingber DE (1993) Mechanotransduction across the cell surface and through the cytoskeleton [see comments]. Science 260(5111): 1124–1127.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Zhang, D., Jin, N., Rhoades, R.A. et al. Influence of microtubules on vascular smooth muscle contraction. J Muscle Res Cell Motil 21, 293–300 (2000). https://doi.org/10.1023/A:1005600118157
Issue Date:
DOI: https://doi.org/10.1023/A:1005600118157